To determine abnormal plain radiograph findings of the distal phalanx tuft of the hand (DPTH) associated with systemic sclerosis in adults.
MethodsA systematic review was developed following the parameters of the PRISMA guidelines in databases: MEDLINE, EMBASE, BIREME, Scielo, Google Scholar and others including as primary outcomes alterations of DPTH (erosions, resorption, sclerosis and proliferation) detected by simple radiography in subjects with systemic sclerosis. The prevalence of radiographic findings was synthesized using the fixed effects model. The statistical associations were expressed in terms of relative risk or odds ratio with their respective confidence intervals and p values.
ResultsTwenty-two observational studies were included; the prevalence of DPTH resorption was 28.3% (95% CI: 0.256–0.312; p<.001); I2=80.4%, the prevalence of calcinosis was 15.6% (95% CI: 0.113–0.210; p<.001); I2=0%. No study reported proliferation or erosions and only one study described sclerosis of DPTH in 5 individuals.
ConclusionsResorption and calcinosis of DPTH are the characteristic radiographic findings in patients with systemic sclerosis. However, new studies with greater methodological strength are needed to establish associations between these phenomena and their presence in other connective tissue diseases.
Determinar en radiografía simple los hallazgos anormales del penacho de la falange distal de los dedos de la mano (PFDM) asociados a la esclerosis sistémica (ES) en adultos.
MétodosSe desarrolló una revisión sistemática siguiendo los parámetros de las guías PRISMA en las bases de datos: MEDLINE, EMBASE, BIREME, Scielo, Google Scholar entre otras incluyendo como resultados primarios las alteraciones del PFDM (erosiones, resorción, esclerosis y proliferación) detectadas por radiografía simple en sujetos con ES. La prevalencia de los hallazgos radiográficos fue sintetizada usando el modelo de efectos fijos. Las asociaciones estadísticas (según el tipo de estudio) se expresaron en términos de riesgo relativo u odds ratio con sus respectivos intervalos de confianza y valores p.
ResultadosVeintidós estudios observacionales fueron incluidos, la prevalencia de resorción del PFDM fue de 28,3% (IC 95%: 0,256–0,312; p<0,001) y un valor I2=80,4%, la prevalencia de calcinosis fue de 15,6% (IC 95%: 0,113–0,210; p<0,001) y un valor I2=0%. Ningún estudio reportó proliferación o erosiones y solo un estudio describió esclerosis del PFDM en 5 pacientes.
ConclusionesLa resorción y calcinosis del PFDM son los hallazgos radiográficos característicos en pacientes con ES, sin embargo hacen falta estudios con una mayor solidez metodológica que permitan establecer con certeza las asociaciones entre estos fenómenos y la presencia de otras enfermedades del tejido conectivo.
Systemic sclerosis (SSc) is the result of a combination of autoimmune and vascular phenomena in association with fibrosis that triggers changes in the skin, soft tissue, joints and bones.1 It mostly affects women and has a variable clinical course. It is a rapidly progressive condition and, in some cases, has a high risk of mortality.
An early manifestation of SSc is the involvement of the hands,2 which occurs in between 46% and 97% of the cases.3 Despite the low sensitivity and specificity for the detection of early disease-related changes,4 plain radiography of the hands is useful for the follow-up of patients, with calcinosis and acro-osteolysis being the characteristic radiographic findings.5,6
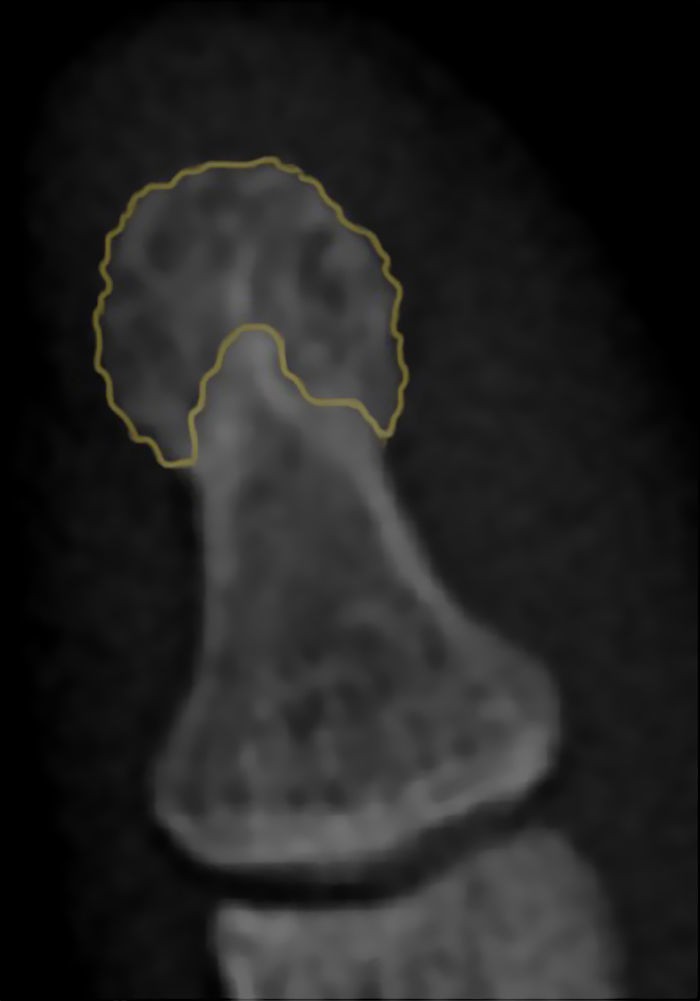
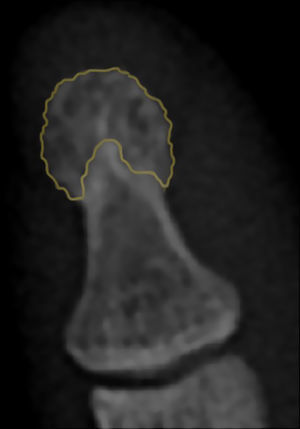
The term acro-osteolysis refers to the resorption of the terminal digital tufts.7 We should point out that the distal phalangeal tuft does not have a universal radiographic definition. However, it can be considered the region of the distal phalanx in which the cortical bone changes and, rather than being completely smooth, acquires a tapered aspect8 (Fig. 1).
The radiographic changes in the phalangeal tuft of the fingers (PTF) in patients with SSc is not exclusive to that disorder. Acro-osteolysis has also been described in individuals with mixed connective tissue diseases (MCTD).6,9 On the other hand, in 1929, Edeiken10 reported 3 cases of SSc with sclerosis of the distal phalanx, a finding that was subsequently characterized in patients with rheumatoid arthritis (RA).11
As a outcome of the discrepancy between evidence concerning the radiological involvement of PTF in musculoskeletal diseases, the description of these structures is overlooked in the interpretation of a plain radiograph of the hands in clinical practice.
The objective of this review is to establish the abnormal findings in the PTF observed in plain radiographs in adults with SSc.
MethodsThis review was carried out following the parameters of the PRISMA guidelines.12 The protocol was registered (registration no. CRD42015025185) in the PROSPERO database of systematic reviews of the University of York, which can be consulted in the following link: http://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID=CRD42015025185.
Search Strategy and EligibilitySearch Method for Identifying Potential Studies- •
We conducted a systematic search up to December 2015, with no restrictions on language or publication date, in the following databases: MEDLINE, EMBASE, BIREME (Biblioteca Regional de Medicina), LILACS (Literatura Latinoamericana y del Caribe en Ciencias de la Salud), SciELO, EBMR (Evidence-Based Medicine Reviews), Cochrane Central Register of Controlled Trials, Clinical Trials and Google Scholar.
- •
The search in MEDLINE was carried out via PubMed, utilizing the MeSH terms “Scleroderma, Systemic”, “Finger Phalanges” and “Radiography”, linking them with the Boolean operator AND. To increase the sensitivity of the search, we aggregated the respective entry terms for each MeSH term, linking them with the Boolean operator OR.
- •
Given that PTF has no MeSH equivalent, an additional search was performed replacing the MeSH term “Finger Phalanges” by the words “tuft OR plume”.
- •
The search strategy employed in MEDLINE was adapted for BIREME, using the respective DeSC terms “Descriptores en Ciencias de la Salud [Health Sciences Descriptors]”, and for the other databases, we used the equivalent MeSH-DeSC keywords mentioned above. The complete set of search terms can be found in the supplemental material corresponding to the protocol registered in the PROSPERO platform.
- •
Once duplicates had been excluded, 2 authors (LMC and SVG) who were not experts in radiology or rheumatology evaluated the potential articles. Those documents included had at least 1 of the following words in the title or abstract: “erosion”, “resorption”, “sclerosis” or “proliferation” (which were translated to the original language of the publication). Any disagreement was resolved by consensus with a third author (YEI).
- (1)
Type of study: case report, case-control, cross-sectional or cohort study and randomized or nonrandomized clinical trial.
- (2)
Type of population: adults with a diagnosis of SSc in accordance with the accepted diagnostic criteria at the time of the study publication date.
- (3)
Intervention: study evaluating the PTF in plain radiography of the hands in one or more projections.
- (1)
Investigations that included individuals with a concomitant diagnosis of SSc and some other musculoskeletal disease.
The complete texts of the screened articles were acquired from online databases, by searching for the journals in public libraries and, in some cases, by contacting with the publishers or authors to request the original article.
The verification of the inclusion criteria was carried out independently by 3 authors (ECP, LMC and SVG) who examined the complete texts of the screened publications. Any disagreement was resolved by means of discussion and consensus.
Evaluation of the Risk of BiasIndividually, 3 authors (LMC, SVG and FSZ) evaluated the risk of bias of the studies included in the qualitative synthesis. The level of evidence was established in accordance with the 2011 Oxford Centre for Evidence-Based Medicine levels of evidence. Moreover, the risk of selection bias, detection bias and performance bias of each of the articles was based on a group of questions formulated by the authors that can be consulted in the supplemental material corresponding to the protocol registered in the PROSPERO platform.
The risk of bias assessment for each article was recorded on a form designed for that purpose using the categories “high risk of bias”, low risk of bias” and “unclear risk of bias”. The discrepancies in the evaluation of the authors were resolved by common consent.
Measurements Applied to the Results- –
Definition of the Variables: the following definitions were utilized to record the results.
- –
Acro-osteolysis: irregular decrease in the size of the tuft, a finding that was classified according to 4 grades of severity.1,7
- –
Tuft resorption: loss of the tapered pattern of the PTF, which acquires a focal or generalized concave morphology, with or without disruption of the cortical bone.
- –
Sclerosis of the tuft: thickness of the cortical bone of the PTF greater than 1mm with an increase in bone radiopacity.11
- –
Erosions: area of disruption of the PTF cortical bone, definition similar to that provided by OMERACT for erosions on magnetic resonance images.
- –
Calcinosis of the tuft: calcification of soft tissue adjacent to the PTF.1
- –
Bone proliferation affecting the tuft: increase in the bone surface of the external PTF toward normal cortical bone.
- –
Measures of Central Tendency: continuous variables are expressed using the mean and standard deviation as measures of central tendency and dispersion. Prevalence data are shown as the fraction and equivalent percentage. Finally, the statistical associations (depending on the type of study) were calculated as the relative risk (RR) or adds ratio (OR), with the respective confidence intervals (CI) and p values.
For the studies included in the qualitative synthesis, 2 authors (FSZ and SVG) independently extracted the data and recorded them in a form designed specifically for this purpose. In the case of discrepancy, a third author (YEI) resolved the disagreement.
Statistical AnalysisThe software package Review Manager (RevMan) version 5.313 was used to calculate the statistical association of the dichotomous variables expressed in terms of RR or OR with the p value and 95% CI for the articles that did not express their findings employing these statistical terms in the original text.
The heterogeneity of the studies was determined with the I2 statistic, in which we took as cutoff points >75%, between 51% and 74% and <50% for high, moderate and low heterogeneity, respectively.
The prevalence of the radiographic findings was expressed using the fixed-effects model of Mantel-Haenszel with the software package Comprehensive Meta-Analysis version 3.0.14
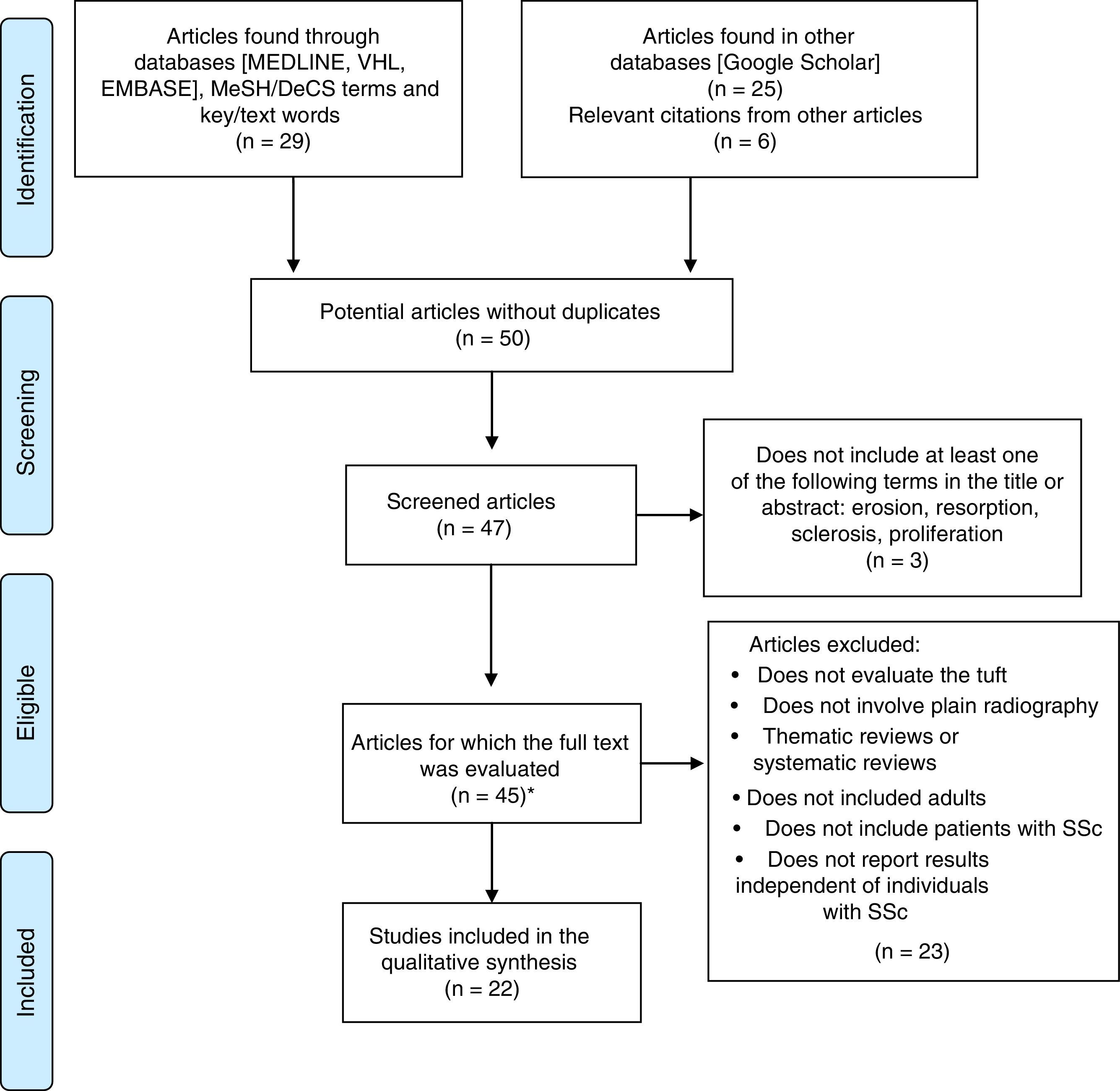
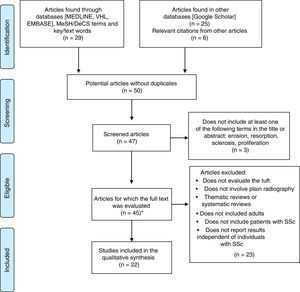
ResultsIn the initial search, we identified 8 articles in MEDLINE, 10 in BIREME, 11 in EMBASE and 25 in Google Scholar. After exclusion of duplicates and screening, we obtained 41 articles eligible for full-text review, in addition to 6 relevant citations in the respective bibliographies. It was impossible to acquire 2 complete texts due to the publication date or to discrepancy in the literature citation provided by the database.
The qualitative synthesis included 3 case reports, 5 case–control studies, 11 cross-sectional studies and 3 cohort studies. The flow chart of the systematic review and the articles included in the analysis is shown in Fig. 2.
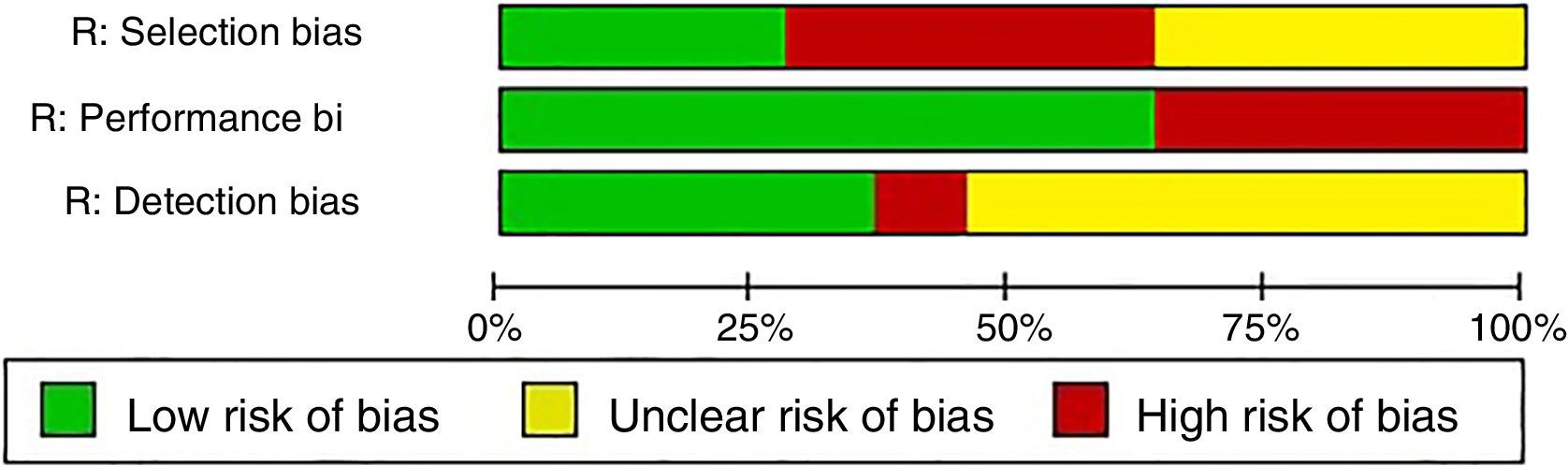
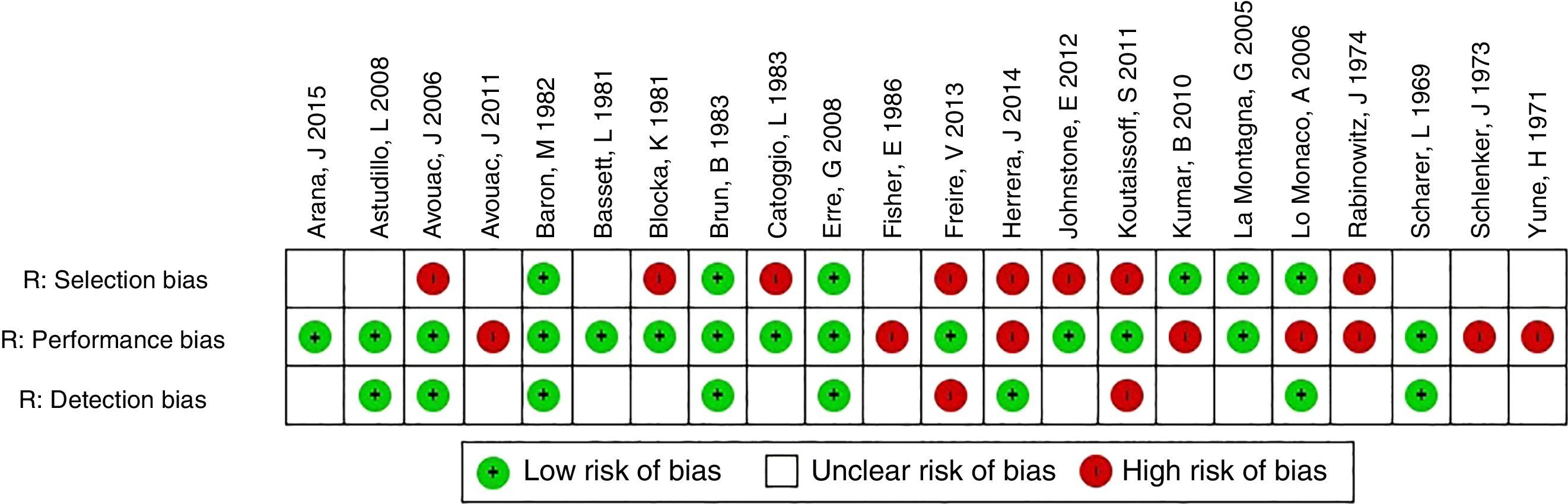
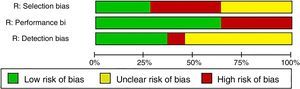
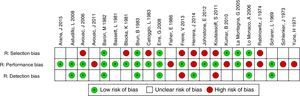
In relation to the risk of bias, only the studies of Baron et al.3 and La Montagna et al.15 were qualified for the 3 items as being “low risk”. The selection risk was more often rated as “high” because a large number of the individuals included had a history of traumatic injury to the fingers. More than half of the studies were designated as showing an “unclear” risk of detection bias since the authors did not specify whether the radiological evaluation was blinded or did not establish the interobserver variability. The results of the evaluation of risk of bias are summarized in Figs. 3 and 4. With respect to the Oxford levels of evidence, 8 articles fell into level 4, 11 into level 3 and this classification was not applied in the remainder as they were case reports.
No study reported proliferation or erosions of the PTF as radiographic findings in patients with SSc and only the study by Bassett et al.16 described sclerosis of the PTF in 5 patients (9.09%).
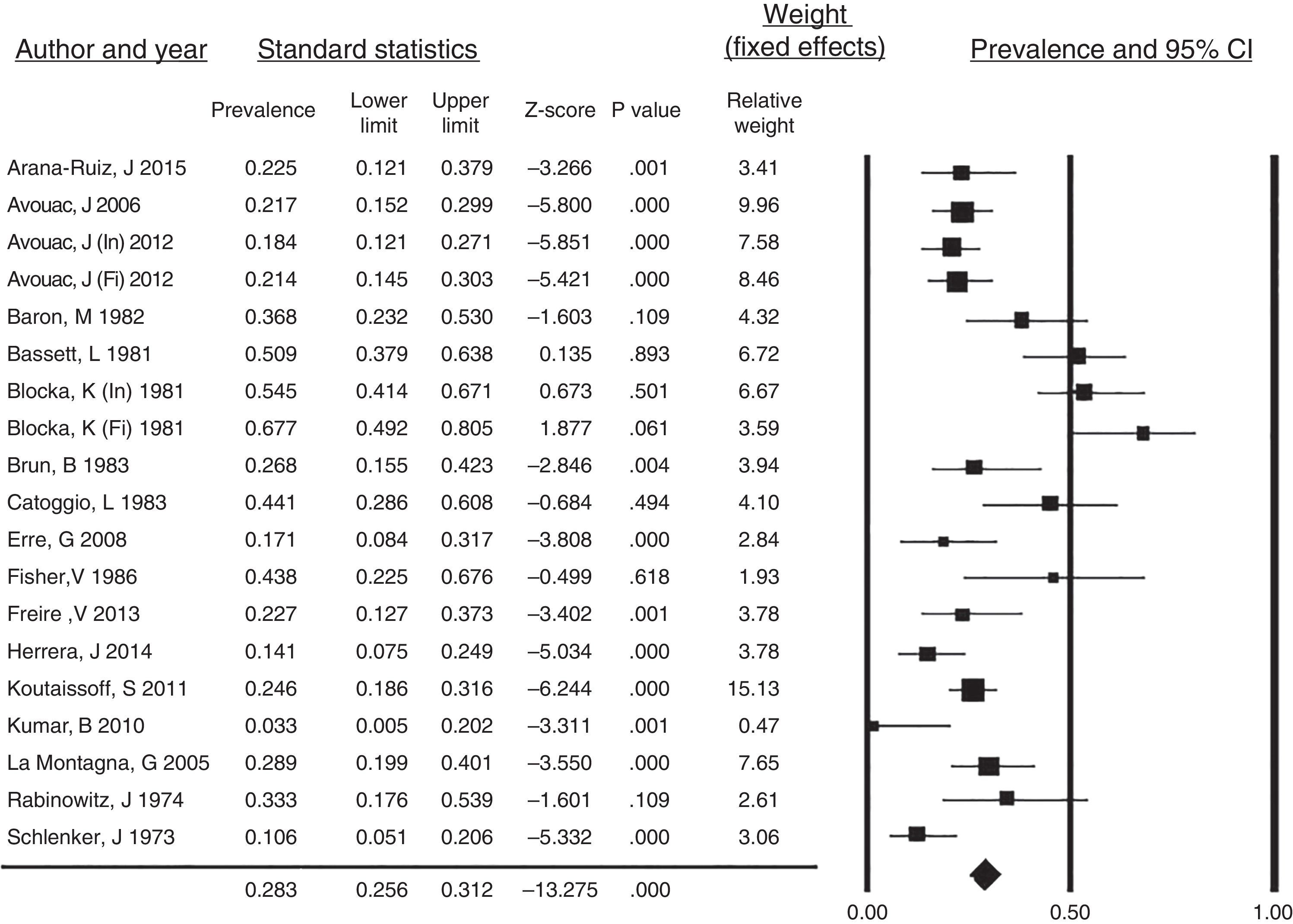
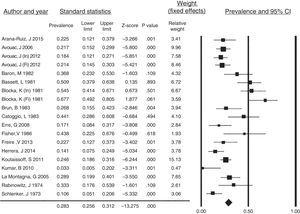
All of the case reports described resorption of the PTF as the only clear finding in the radiographs of the hands of SSc patients. Taking into account the remaining articles, the prevalence of PTF resorption was 28.3% (95% CI: 0.256–0.312; p<.001) and the I2 value=80.4%, indicating a high heterogeneity (Fig. 5). In this analysis, we omitted the investigation by Allali et al.,17 as they did not discriminate the prevalence of tuft resorption in hands and feet and the study of Johnstone et al.7 as, in their results, a grade of acro-osteolysis of 0 involved individuals with and without tuft resorption (grade 0: individual score of each phalange ≤1 or total score ≤8). Fig. 5 corresponds to the forest plot of the prevalence of PTF resorption in SSc patients and the weight recorded for each study established utilizing a fixed-effects model. The data from the initial and final radiographic evaluation of the cohorts studied by Avouac et al.2 and by Blocka et al.6 were included separately.
Prevalence of PTF resorption in systemic sclerosis patients. Forest plot of the prevalence of PTF resorption in the studies included in the systematic review with the weight recorded in each and calculated overall prevalence.
CI, confidence interval; Fi, final cohort evaluation; In, initial cohort evaluation.
The studies that compared individuals with SSc versus (vs) MCTD-CREST (calcinosis, Raynaud's phenomenon, esophageal dysfunction, sclerodactyly and telangiectasia)11,16 found no statistically significant association related to PTF resorption. However, in these subgroups with calcinosis of the PTF, Yune et al.18 found for SSc vs MCTD an OR of 89.85 (95% CI: 5.36–1506.3; p=.002). Koutaissoff et al.1 reported a similar datum in their comparison of individuals with SSc vs “controls without disease”, observing tuft calcinosis with an OR of 60.31 (95% CI: 3.64–999.39; p=.004). Taking these 2 articles into account, the prevalence of tuft calcinosis was 15.6% (95% CI: 0.113–0.210; p<.001) and an I2 value=0%.
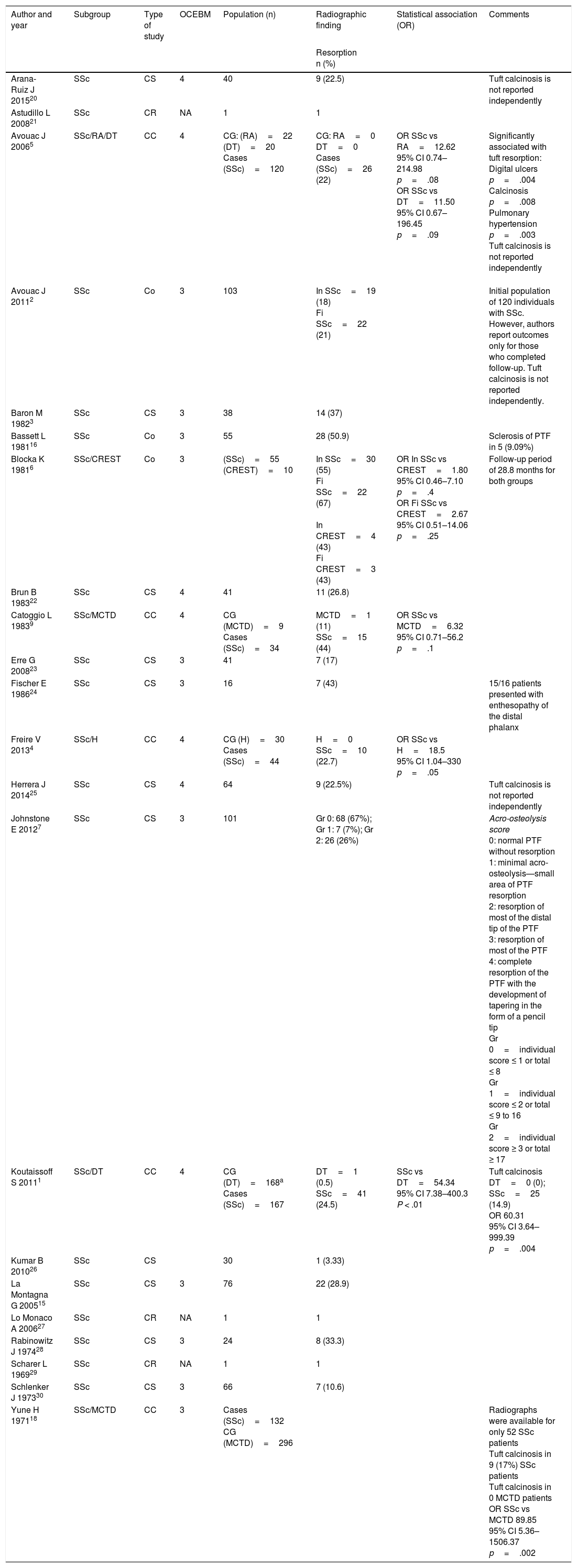
On the other hand, Avouac et al.,5 Freire et al.4 and Koutaissoff et al.1 compared individuals with SSc with “controls without disease”, finding for PTF resorption an OR=11.50 (95% CI: 0.67–196.45; p=.09), OR=18.5 (95% CI: 1.04–330; p=.05) and OR=54.34 (95% CI: 7.38–400.3; p<.01), respectively. Table 1 summarizes in detail the studies included in the synthesis, the populations evaluated and the associations reported.
Characteristics of the Studies Included.
| Author and year | Subgroup | Type of study | OCEBM | Population (n) | Radiographic finding | Statistical association (OR) | Comments |
|---|---|---|---|---|---|---|---|
| Resorption n (%) | |||||||
| Arana-Ruiz J 201520 | SSc | CS | 4 | 40 | 9 (22.5) | Tuft calcinosis is not reported independently | |
| Astudillo L 200821 | SSc | CR | NA | 1 | 1 | ||
| Avouac J 20065 | SSc/RA/DT | CC | 4 | CG: (RA)=22 (DT)=20 Cases (SSc)=120 | CG: RA=0 DT=0 Cases (SSc)=26 (22) | OR SSc vs RA=12.62 95% CI 0.74–214.98 p=.08 OR SSc vs DT=11.50 95% CI 0.67–196.45 p=.09 | Significantly associated with tuft resorption: Digital ulcers p=.004 Calcinosis p=.008 Pulmonary hypertension p=.003 Tuft calcinosis is not reported independently |
| Avouac J 20112 | SSc | Co | 3 | 103 | In SSc=19 (18) Fi SSc=22 (21) | Initial population of 120 individuals with SSc. However, authors report outcomes only for those who completed follow-up. Tuft calcinosis is not reported independently. | |
| Baron M 19823 | SSc | CS | 3 | 38 | 14 (37) | ||
| Bassett L 198116 | SSc | Co | 3 | 55 | 28 (50.9) | Sclerosis of PTF in 5 (9.09%) | |
| Blocka K 19816 | SSc/CREST | Co | 3 | (SSc)=55 (CREST)=10 | In SSc=30 (55) Fi SSc=22 (67) In CREST=4 (43) Fi CREST=3 (43) | OR In SSc vs CREST=1.80 95% CI 0.46–7.10 p=.4 OR Fi SSc vs CREST=2.67 95% CI 0.51–14.06 p=.25 | Follow-up period of 28.8 months for both groups |
| Brun B 198322 | SSc | CS | 4 | 41 | 11 (26.8) | ||
| Catoggio L 19839 | SSc/MCTD | CC | 4 | CG (MCTD)=9 Cases (SSc)=34 | MCTD=1 (11) SSc=15 (44) | OR SSc vs MCTD=6.32 95% CI 0.71–56.2 p=.1 | |
| Erre G 200823 | SSc | CS | 3 | 41 | 7 (17) | ||
| Fischer E 198624 | SSc | CS | 3 | 16 | 7 (43) | 15/16 patients presented with enthesopathy of the distal phalanx | |
| Freire V 20134 | SSc/H | CC | 4 | CG (H)=30 Cases (SSc)=44 | H=0 SSc=10 (22.7) | OR SSc vs H=18.5 95% CI 1.04–330 p=.05 | |
| Herrera J 201425 | SSc | CS | 4 | 64 | 9 (22.5%) | Tuft calcinosis is not reported independently | |
| Johnstone E 20127 | SSc | CS | 3 | 101 | Gr 0: 68 (67%); Gr 1: 7 (7%); Gr 2: 26 (26%) | Acro-osteolysis score 0: normal PTF without resorption 1: minimal acro-osteolysis—small area of PTF resorption 2: resorption of most of the distal tip of the PTF 3: resorption of most of the PTF 4: complete resorption of the PTF with the development of tapering in the form of a pencil tip Gr 0=individual score ≤ 1 or total ≤ 8 Gr 1=individual score ≤ 2 or total ≤ 9 to 16 Gr 2=individual score ≥ 3 or total ≥ 17 | |
| Koutaissoff S 20111 | SSc/DT | CC | 4 | CG (DT)=168a Cases (SSc)=167 | DT=1 (0.5) SSc=41 (24.5) | SSc vs DT=54.34 95% CI 7.38–400.3 P < .01 | Tuft calcinosis DT=0 (0); SSc=25 (14.9) OR 60.31 95% CI 3.64–999.39 p=.004 |
| Kumar B 201026 | SSc | CS | 30 | 1 (3.33) | |||
| La Montagna G 200515 | SSc | CS | 3 | 76 | 22 (28.9) | ||
| Lo Monaco A 200627 | SSc | CR | NA | 1 | 1 | ||
| Rabinowitz J 197428 | SSc | CS | 3 | 24 | 8 (33.3) | ||
| Scharer L 196929 | SSc | CR | NA | 1 | 1 | ||
| Schlenker J 197330 | SSc | CS | 3 | 66 | 7 (10.6) | ||
| Yune H 197118 | SSc/MCTD | CC | 3 | Cases (SSc)=132 CG (MCTD)=296 | Radiographs were available for only 52 SSc patients Tuft calcinosis in 9 (17%) SSc patients Tuft calcinosis in 0 MCTD patients OR SSc vs MCTD 89.85 95% CI 5.36–1506.37 p=.002 |
The description includes the type of study, number of participants, disease evaluated, 2011 Oxford Centre for Evidence-Based Medicine (OCEBM) levels of evidence, the prevalence of PTF resorption and reported or calculated statistical associations.
Cases, SSc patients; CC, case-control; CG, control group; Co, cohort; CR, case report; CS, cross-sectional; DT, digital trauma, Fi, final cohort evaluation; Gr, grade; H, healthy; In, initial cohort evaluation; MCTD, mixed connective tissue diseases; NA, not applicable; RA, rheumatoid arthritis; SSc, systemic sclerosis; vs, versus.
The present study found that PTF resorption was the radiographic finding affecting the tuft associated with a higher rate of SSc in individuals and a prevalence of 28.3%, and a statistically significant difference when compared with controls without disease, with an OR between 11.5 and 54. Only the study of Avouac et al.5 did not observe a significant association, a fact that may be explained by the limited number of controls involved in the report (20 individuals with a history of traumatic injury to the fingers).
Many of the studies selected for the qualitative synthesis included control groups with a history of traumatic injury. This fact could lead to bias due to a confounding factor; the other review, a publication written by our group (the results of which are available in http://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID=CRD42015025185), found that patients with a history of traumatic injury were more likely to develop changes in the PTF than individuals with no previous traumatic injury affecting their hands. Thus, it was decided to employ the concept of “controls without disease” and not utilize the term “healthy controls”.
In the comparison with radiographic findings in the PTF of patients with SSC and with other diseases, the evidence was limited. Three reports involved patients with MCTD-CREST6,9,18 and 1 included controls with RA.5 None of these studies found statistically significant associations in terms of tuft resorption, possibly as a result of the small number of individuals in the control groups.
The study of Yune et al.,18 which had a large control group (296 patients with MCTD), did not evaluate PTF resorption, but did report calcinosis of the PTF as a significant finding in individuals with SSc. This observation agrees with results documented by Koutaissoff et al.,1 who also graded the severity of the calcinosis in accordance with the extent and density of the calcifications. Other studies pointed out the high prevalence of soft tissue calcinosis, but did not differentiate between calcinosis of the tuft (or the pulp of the finger) and calcification of other sites of the hand, and, thus, were not included in the analysis.
It was not possible to perform a meta-analysis because of the high statistical and clinical heterogeneity (only 1 study compared SSc and RA and 4 compared SSc and controls without disease). On the other hand, cross-sectional and cohort studies did not provide data on disease duration or the sex or age of participants and, therefore, it was not possible to carry out a more in-depth analysis.
We mention 2 classifications for grading the severity of acro-osteolysis in SSc patients. Koutaissoff et al.1 classify acro-osteolysis ranging from absence of resorption to complete resorption of the tuft. They present images illustrating each grade and define as abnormal a score greater than 2 for each finger. On the other hand, Johnstone et al.7 propose a score that is specified in the comments on their study provided in Table 1. To date, the performance and reliability of these scores in unknown as the authors did not evaluate their inter- and intraobserver variability.
In accordance with the results of Johnstone et al.,7 individuals with moderate-to-severe acro-osteolysis were more likely to require debridement, amputation or the use of intravenous prostanoids than those with mild acro-osteolysis. This suggests, as was proposed by Avouac et al. in 2006,5 that the grade of acro-osteolysis is proportional to the severity of digital ischemia. Likewise, the 2011 prospective study of Avouac et al.2 found that the patients who developed acro-osteolysis and those in whom it got worse were more likely to ultimately meet severity criteria such as flexion contracture, severe vascular damage and interstitial lung involvement. Thus, this change in the PTF is a marker of the prognosis and the severity of SSc.
Chung et al.19 recently presented a radiographic scoring system to evaluate calcinosis in SSc patients that has an excellent inter- and intraobserver reliability (intraclass correlation coefficient 0.89 and 0.93, respectively). For this calculation, the authors divided the hand into 22 anatomical regions (10 corresponding to the PTF) and estimated the percentage of area covered with calcium and the severity of the calcinosis according to a simple scoring system previously proposed by Koutaissoff et al.1
An important limitation to this study lies in the differences between the diagnostic criteria employed by the authors of the primary studies, which include research from 1969 to 2013. This could explain the high clinical and statistical heterogeneity, such as that found in the prevalence of PTF resorption in this review.
Finally, current evidence suggests that acro-osteolysis is another of the ischemic manifestations of SSc and, thus, its radiographic characterization is useful in establishing the prognosis and severity of the disease. In this respect, the implementation of radiological scores standardizes criteria for the evaluation of these aspects, although further research is necessary to determine their reliability and validity.
In conclusion, resorption and calcinosis of the PTF are the characteristic radiographic findings in patients with SSc. However, studies with a greater methodological soundness are required to enable us to firmly establish the associations between these phenomena and the presence of other connective tissue diseases.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
FundingThis project was performed without outside funding.
Conflicts of InterestThe authors declare they have no conflicts of interest.
The authors wish to thank Óscar Eduardo Guzmán Mendoza, assistant researcher, a medical student of Universidad Nacional de Colombia and member of an incentive program for investigation, Grupo de Investigación en Radiología e Imágenes Diagnósticas (GRID).
Please cite this article as: Izquierdo YE, Calvo Páramo E, Castañeda LM, Gómez SV, Zambrano FS. Cambios radiográficos del penacho de la falange distal de las manos en pacientes con esclerosis sistémica. Revisión sistemática. Reumatol Clin. 2018;14:20–26.