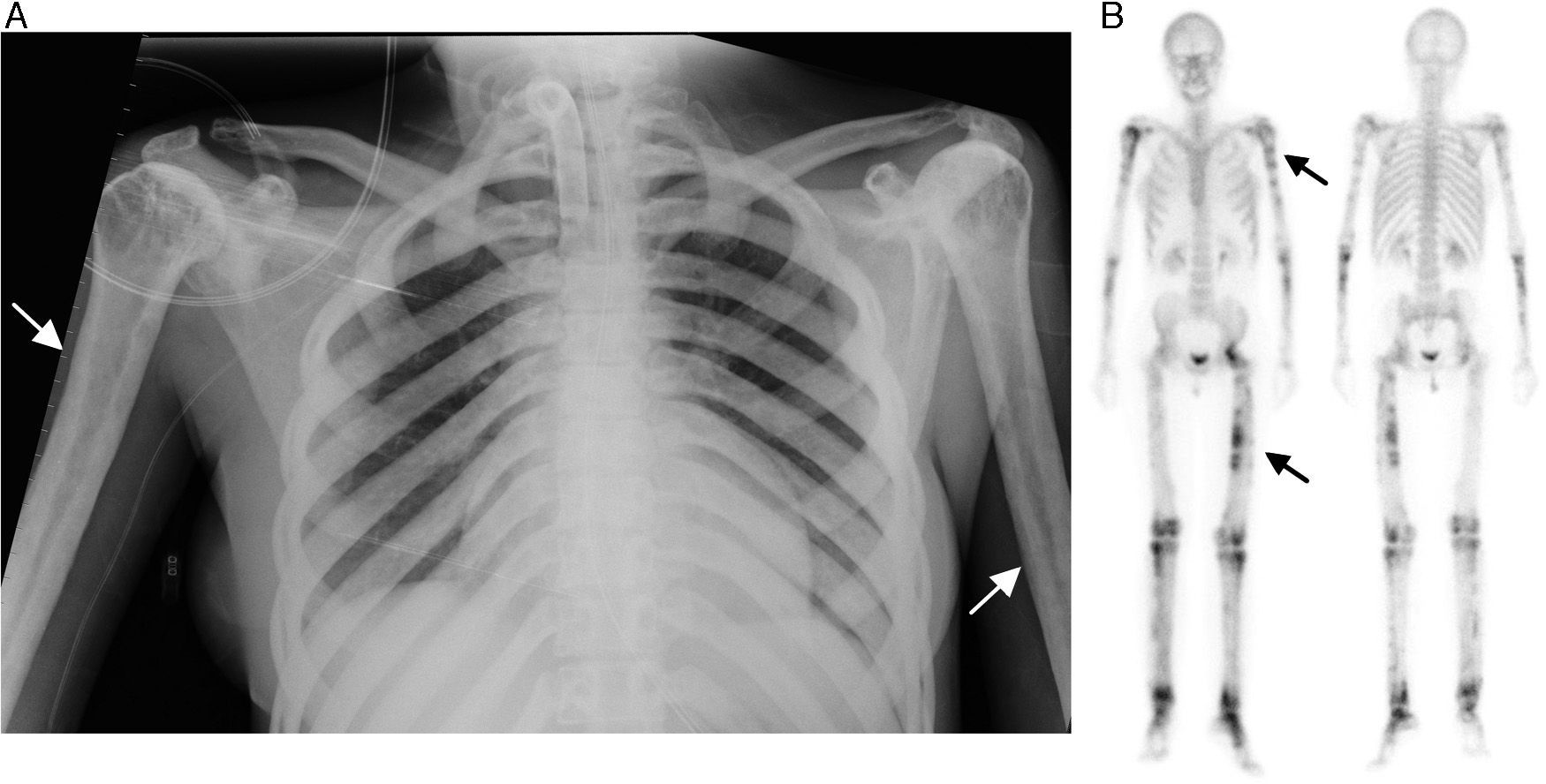
The patient was a 37-year-old woman, with a personal history of muscle weakness, pain in lower limbs and a waddling gait with claudication since she had been 18 months old. In a chest radiography carried out for dyspnea, it was incidentally found that she had generalized demineralization, with thickening of the diaphyseal cortical bone of the long bones (Fig. 1A, arrows). Given these findings, she underwent whole body bone scintigraphy, to determine the extension of the involvement (Fig. 1B, arrows), which revealed large areas with an increase in osteoblastic activity in the diaphysis of the long bones of her upper and lower limbs, as well as involvement of left hip, knees and tarsus, with different degrees of activity. Molecular genetic testing resulted in the definitive diagnosis of Camurati-Engelmann disease. The genetic diagnosis was subsequently reached in 4 first-degree members of her family with a history of pain in lower limbs and proximal muscle weakness.
(A) Anteroposterior chest radiography showing the irregular thickening of the diaphyseal cortical bone of the long bones, especially in both humeri. This bilateral and symmetrical cortical thickening is also evident in the clavicles, greater in the medial area. (B) Whole body bone scintigraphy in anterior and posterior images, showing multiple abnormal uptakes of 99mTc-diphosphonates with different uptake intensity, in relation to the osteoblastic activity, which is more evident in diaphyseal regions of the long bones and left hip.
Camurati-Engelmann disease is an autosomal dominant disease, described for the first time in 1929,1 characterized by a progressive diaphyseal dysplasia. To date there have been approximately 300 cases reported in the literature, and the clinical course and progression rates are defined as being highly variable. This rare disabling disease is expressed as the thickening of the endosteum and the periosteum of the diaphyseal cortical bone of the long bones, whereas the epiphysis and metaphysis appear to be conserved. In some cases, the skullcap or hips can be affected.2,3 Within the clinical variability, we stress the pain in the limbs, the broad-based waddling gait, muscle weakness, clumsiness for precise movements, and its ability to remain underdiagnosed for years. Bone scintigraphy enables the detection of the generalized bone involvement, with extensive areas showing an increase in the osteoblastic activity in up to 74% of the patients studied.4 The disease is determined by mutations in the transforming growth factor β1 gene (TGF-β1), an important stimulator of bone regeneration, located in chromosome 19q13.1-q13.3.5,6 With respect to treatment, the symptoms can be managed with corticosteroids, nonsteroidal anti-inflammatory drugs, proton pump inhibitors and rehabilitation programs, when they are tolerated.
Informative MessageBone scintigraphy is a simple, noninvasive technique that enables the study of the whole body, showing the areas with the greatest involvement in patients with generalized bone disease. The incidental finding of cortical thickening on imaging studies should be examined by an extensive bone scan to improve staging and its classification.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflicts of InterestThe authors declare they have no conflicts of interest.
Please cite this article as: de Bonilla Damiá Á, García Gómez FJ. Enfermedad de Camurati-Engelmann. Reumatol Clin. 2017;13:48–49.