To determine and compare the prevalence of vitamin D insufficiency and deficiency in patients with systemic lupus erythematosus (SLE) with and without disease activity.
Patients and methodsWe made a comparative, observational, cross-sectional, prospective study of 137 women with SLE according to American College of Rheumatology criteria. Patients with chronic kidney disease, cancer, hyperparathyroidism, pregnancy, and lactation were excluded. Disease activity was assessed using the MEX-SLEDAI score: a score of ≥3 was considered as disease activity. Data were collected on diabetes mellitus, the use of corticosteroids, chloroquine, and immunosuppressants, photoprotection and vitamin D supplementation. Vitamin D levels were measured by chemiluminescent immunoassay: insufficiency was defined as serum 25-hydroxyvitamin D <30ng/mL and deficiency as <10ng/mL.
Results137 women with SLE (mean age 45.9±11.6 years, disease duration 7.7±3.4 years) were evaluated. Mean disease activity was 2 (0–8): 106 patients had no disease activity and 31 had active disease (77.4% versus 22.6%). Vitamin D insufficiency and deficiency was found in 122 (89.0%) and 4 (2.9%) patients, respectively. There was no significant difference in vitamin D levels between patients with and without active disease (19.3±4.5 versus 19.7±6.8; P=.75). No correlation between the MEX-SLEDAI score (P=.21), photosensitivity, photoprotection, prednisone or chloroquine use and vitamin D supplementation was found.
ConclusionsWomen with SLE had a high prevalence of vitamin D insufficient. No association between vitamin D levels and disease activity was found.
Determinar la prevalencia de insuficiencia y deficiencia de vitamina D en pacientes con lupus eritematoso sistémico (LES) y compararlas con actividad de la enfermedad.
Pacientes y métodosEstudio comparativo, observacional, transversal y prolectivo. Se incluyeron 137mujeres con LES según los criterios del Colegio Americano de Reumatología. Se excluyeron pacientes con enfermedad renal crónica, cáncer, hiperparatiroidismo, embarazo y lactancia. La actividad fue medida mediante el índice MEX-SLEDAI, considerando actividad ≥3. Se obtuvieron los siguientes datos: diabetes mellitus, uso de glucocorticoides, cloroquina e inmunosupresores, fotoprotección y suplementación con vitamina D. Los niveles de vitamina D se midieron con inmunoanálisis quimioluminiscente considerando insuficiencia a niveles séricos de 25-hidroxivitamina D<30ng/ml y deficiencia<10ng/ml.
ResultadosSe evaluaron 137mujeres con LES (edad promedio 45,9±11,6años, duración de la enfermedad 7,7±3,4 años). La mediana de actividad mediante MEX-SLEDAI fue 2 (0-8),106pacientes en inactividad y 31 con actividad (77,4% versus 22,6%). La insuficiencia y deficiencia de vitamina D se encontró en 122 (89,0%) y 4 (2,9%) pacientes respectivamente. Al comparar los niveles de vitamina D entre pacientes con y sin actividad no existieron diferencias estadísticamente significativas (19,3±4,5 versus 19,7±6,8; p=0,75); tampoco se encontró una correlación con el puntaje MEX-SLEDAI (p=0,21) ni fotosensibilidad, fotoprotección, uso de prednisona, cloroquina ni suplementación con vitamina D.
ConclusionesLas mujeres con LES presentaron elevada prevalencia de insuficiencia de vitamina D. No se encontró asociación de niveles de vitamina D con actividad de la enfermedad.
Vitamin D (vit D) is a fat-soluble prohormone that acts by binding to vitamin D receptor (VDR).1 The discovery of VDR in most of the cells of the immune system (lymphocytes and macrophages) suggests that it participates in the regulation of the immune response, beyond its traditional role in bone health and calcium homeostasis. Vitamin D has been related to several chronic diseases such as cardiovascular disease, cancer and autoimmune diseases like type 1 diabetes mellitus, multiple sclerosis, rheumatoid arthritis and systemic lupus erythematosus (SLE).2,3 The latter is a multisystem autoimmune disease characterized by a diversity of immunological changes. A number of the immunomodulatory activities of vit D deficiency counteract the immunological alterations observed in SLE. This vitamin has been related to inhibition of the production of IgG anti-double-stranded DNA (anti-dsDNA), dendritic cell maturation and interferon alpha (IFN-α) gene expression in patients with SLE.4,5 A recent study also showed that patients with SLE and vit D deficiency had greater IFN-α activity and B-cell activation compared with those who had higher vit D levels. This fact shows that vit D insufficiency can intensify the immunological changes in SLE patients6; this deficiency seems to be multifactorial (lack of sun exposure, use of sunscreens, glucocorticoid therapy and chronic kidney disease).7,8 The epidemiology of vit D insufficiency in SLE patients in tropical countries like Mexico should be analyzed, as we have access to preventive and therapeutic methods. However, the relationship between vit D levels and disease activity is a controversial subject. The objectives of this study are to determine the prevalence of vit D insufficiency and deficiency in Mexican SLE patients and compare the vit D levels in patients with and without disease activity.
Patients and MethodsIn this observational, cross-sectional, prospective study we enrolled Mexican Mestizo women who met the classification criteria for SLE of the American College of Rheumatology (ACR).9,10 The patients were from the research unit for systemic autoimmune diseases of Hospital General Regional N.° 36 of the Mexican Social Security Institute, Puebla, Mexico. All of the participants signed the informed consent form. The local institutional committee approved the study. The sample size was determined in accordance with the difference in vit D in SLE patients with and without disease activity. The calculated minimum sample size was 126patients. The participating women were legally of age (≥18 years), and were excluded if they had chronic kidney disease (creatinine clearance <60mL/min), cancer or hyperparathyroidism, and if they were pregnant or breastfeeding. A structured interview was held to obtain demographic data and the medical history, including age, a history of photosensitivity, the use of sunscreens, disease duration, a diagnosis of diabetes mellitus, glucocorticoid (prednisone) therapy and dose at the time of the study, as well as administration of immunosuppressive agents, chloroquine and vit D supplementation. Physical examination provided weight, height and body mass index.
Disease ActivityWe used the Systemic Lupus Erythematosus Disease Activity Index, validated for the Mexican population (MEX-SLEDAI),11 to evaluate disease activity. This resulted in a group of patients with no activity—MEX-SLEDAI score of 0–2—and those with activity, indicated by a score of ≥3.12
Vitamin D MeasurementsSeveral serum samples were collected to measure serum 25-hydroxyvitamin D (25[OH]D) using an ARCHITECT chemiluminescent microparticle immunoassay (Abbott, Abbott Park, IL, United States). Vitamin D insufficiency was defined as 25(OH)D levels <30ng/mL and deficiency as <10ng/mL.7,13 For the statistical analysis, we considered any level of vit D <30ng/mL to be low.
Statistical AnalysisAll of the statistical estimates were performed using the statistical software package SPSS version 18.0 for Windows (SPSS Inc., Chicago, IL, United States). Normality of the data was established with the Kolmogorov–Smirnov test. Normally distributed variables were expressed using means and standard deviations, and those that were not normally distributed, by median and interquartile range. The prevalence of vit D insufficiency and deficiency was calculated as the proportion between the number of patients with 25(OH)D levels under the cut-off point (30 and 10ng/mL, respectively) and total participants. Univariate comparisons of categorical variables were done with the chi-square test. Comparisons of continuous variables in the two groups were done using Student's t test or the Mann–Whitney U test, depending on the case. In the correlation between two continuous variables, Pearson or Spearman correlation coefficient was utilized, respectively, for normal and non-normal data. We performed linear regression analysis, in the attempt to identify factors that could be associated with 25(OH)D. Statistical significance was considered to be indicated by a P value <.05.
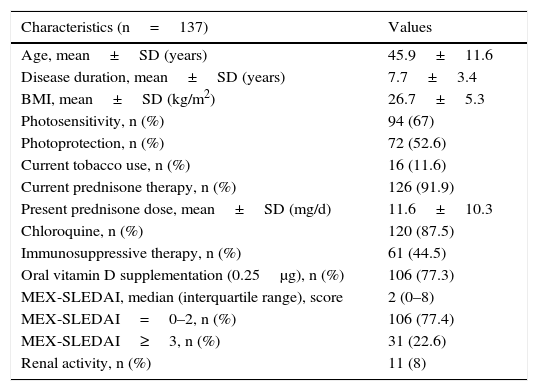
ResultsPatient CharacteristicsThe demographic, clinical and treatment characteristics of the 137 patients included are described in Table 1. The mean age and disease duration were 45.9±11.6 and 7.7±3.4 years, respectively. The majority of the participants had a history of photosensitivity (67%), utilization of sunscreens (52.6%) and oral vit D supplementation (77.3%).
Demographic, Clinical and Therapeutic Characteristics of 137 Women With Systemic Lupus Erythematosus.
| Characteristics (n=137) | Values |
|---|---|
| Age, mean±SD (years) | 45.9±11.6 |
| Disease duration, mean±SD (years) | 7.7±3.4 |
| BMI, mean±SD (kg/m2) | 26.7±5.3 |
| Photosensitivity, n (%) | 94 (67) |
| Photoprotection, n (%) | 72 (52.6) |
| Current tobacco use, n (%) | 16 (11.6) |
| Current prednisone therapy, n (%) | 126 (91.9) |
| Present prednisone dose, mean±SD (mg/d) | 11.6±10.3 |
| Chloroquine, n (%) | 120 (87.5) |
| Immunosuppressive therapy, n (%) | 61 (44.5) |
| Oral vitamin D supplementation (0.25μg), n (%) | 106 (77.3) |
| MEX-SLEDAI, median (interquartile range), score | 2 (0–8) |
| MEX-SLEDAI=0–2, n (%) | 106 (77.4) |
| MEX-SLEDAI≥3, n (%) | 31 (22.6) |
| Renal activity, n (%) | 11 (8) |
BMI, body mass index; MEX-SLEDAI, Mexican Systemic Lupus Erythematosus Disease Activity Index; SD, standard deviation.
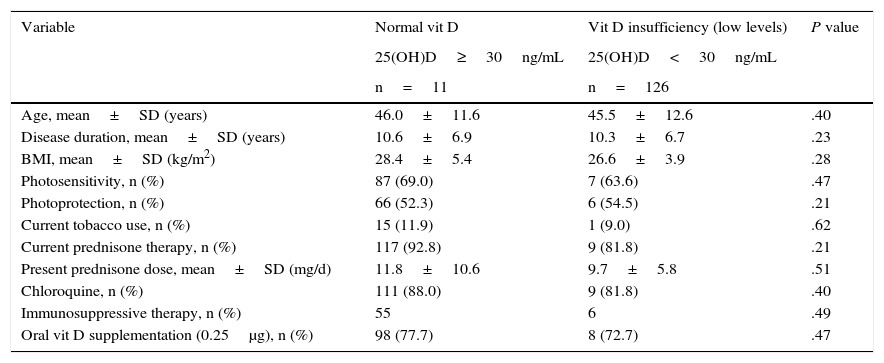
The mean 25(OH)D value was 19.7±6.3ng/mL. The prevalence of vit D insufficiency (<30ng/mL) was 89.0% (122 patients) and that of deficiency (<10ng/mL) was 2.9% (4 patients); thus, 91.9% (126 patients) had low vit D levels. Univariate analysis showed no association between insufficiency (low levels) of vit D and the demographic and clinical variables studied, including photosensitivity, the use of sunscreens, oral vit D supplementation and glucocorticoid therapy (Table 2).
Comparison of Patients With Systemic Lupus Erythematosus With Normal Vitamin D Versus Vitamin D Insufficiency (Low Levels).
| Variable | Normal vit D | Vit D insufficiency (low levels) | P value |
|---|---|---|---|
| 25(OH)D≥30ng/mL | 25(OH)D<30ng/mL | ||
| n=11 | n=126 | ||
| Age, mean±SD (years) | 46.0±11.6 | 45.5±12.6 | .40 |
| Disease duration, mean±SD (years) | 10.6±6.9 | 10.3±6.7 | .23 |
| BMI, mean±SD (kg/m2) | 28.4±5.4 | 26.6±3.9 | .28 |
| Photosensitivity, n (%) | 87 (69.0) | 7 (63.6) | .47 |
| Photoprotection, n (%) | 66 (52.3) | 6 (54.5) | .21 |
| Current tobacco use, n (%) | 15 (11.9) | 1 (9.0) | .62 |
| Current prednisone therapy, n (%) | 117 (92.8) | 9 (81.8) | .21 |
| Present prednisone dose, mean±SD (mg/d) | 11.8±10.6 | 9.7±5.8 | .51 |
| Chloroquine, n (%) | 111 (88.0) | 9 (81.8) | .40 |
| Immunosuppressive therapy, n (%) | 55 | 6 | .49 |
| Oral vit D supplementation (0.25μg), n (%) | 98 (77.7) | 8 (72.7) | .47 |
BMI, body mass index; SD, standard deviation; vit D, vitamin D.
In most SLE patients, the intensity of disease activity evaluated with MEX-SLEDAI was mild to moderate, with a median (interquartile range) score of 2 (0–8). In agreement with the presence and absence of activity, 31 patients (22.6%) were classified as active (20 patients [14.6%] with mild activity and 11 patients [8%] with moderate activity) and 106 patients (77.4%) as inactive. The levels of 25(OH)D were slightly lower in the group of patients with activity, compared to those with no activity; however, this difference was not statistically significant (19.3±4.5ng/mL versus 19.7±6.8ng/mL; P=.75). The 25(OH)D levels were not related to the MEX-SLEDAI values (P=.21). This variable was not a statistically significant predictor in the multiple regression model mentioned above.
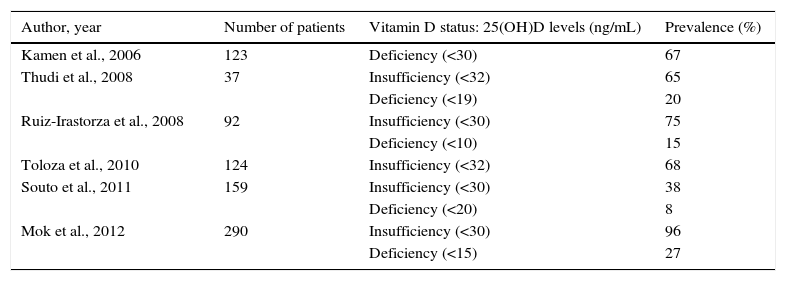
DiscussionThe measurement of serum 25(OH)D levels is considered to be the most accurate method of evaluating an individual's vit D status.14 The results of the present study show that low vit D levels are common among patients with SLE (91.9% with levels <30ng/mL), despite the fact that the population studied lives in Mexico, a country with sunny weather most of the year. To our knowledge, this is the first Mexican study designed to identify the prevalence of vit D insufficiency and deficiency in a population with SLE. A number of studies in other populations have reported these prevalences in SLE, although the 25(OH)D levels considered for the analysis differ from one study to another (Table 3). In a recent report, Mok et al.15 studied 290 SLE patients, finding a prevalence similar to the 96% observed by us, one of the highest, together with that of a Spanish study authored by Cutillas-Marco et al.16 in which, in a population of 55 patients—most of whom (80%) had skin involvement—the authors found a prevalence of insufficiency of 95% and of deficiency of 11%. A Mexican report evaluated the vit D insufficiency in the general population, including 31 women, with a mean age of 34.1±9.7years. The prevalence of vit D insufficiency (<30ng/mL) in this group was 83.9%.17 Clark et al.18 measured serum vit D levels in 585 healthy Mexicans, 54% of whom were women; of the total population, 9.6% had adequate levels (≥30ng/mL), with prevalences of 46.8% and 43.6% for levels of 20–29ng/mL and <20ng/mL respectively. Both studies show a high prevalence of vit D insufficiency in the general population of Mexico. However, our study suggests that the frequency of vit D insufficiency is greater in SLE patients. On the other hand, in the present study, we analyzed the possible risk factors associated with vit D insufficiency in women with SLE, finding no association with age, disease duration, photosensitivity, photoprotection, vit D supplementation or glucocorticoid therapy. The absence of these associations may be due to a sample size that was insufficient for this purpose. With respect to these findings, it is important to point out that vit D plays two important physiological functions in SLE patients: first, it helps to preserve bone homeostasis, maintaining the amount of calcium-phosphate product within a normal range19 and, second, it also modulates the immune system and has immunoregulatory effects. In vitro studies have shown that 1,25-hydroxyvitamin D3 modulates the immune response, both innate and adaptive.20 Vitamin D increases the production of interleukin 10 and the chemokine, CCL22, derived from dendritic cells that promote the development and chemotaxis of T regulatory cells.21,22 It also decreases T lymphocyte activation mediated by dendritic cells through the downregulation of the expression of major histocompatibility complex class II molecules and costimulators.5 Moreover, vit D exerts modulatory effects on the B cells, inhibiting their proliferation, their differentiation into plasma cells and the production of immunoglobulin (Ig) G and IgM.23 Several of the immunoregulatory actions of vit D counteract the immunological changes observed in SLE patients. Studies have demonstrated that 1,25-hydroxyvitamin D and its analogs inhibit the production of IgG anti-dsDNA.24 Likewise, vit D has been found to inhibit dendritic cell maturation and the expression of IFN-α genes in patients with SLE.4 In a more recent study, it was also seen that SLE patients with vit D deficiency had a greater serum IFN-α activity and B cell activation compared with those patients who had higher levels of vit D.6
Summary of Recent Studies on the Prevalence of Vitamin D Insufficiency and Deficiency in Patients With Systemic Lupus Erythematosus.
| Author, year | Number of patients | Vitamin D status: 25(OH)D levels (ng/mL) | Prevalence (%) |
|---|---|---|---|
| Kamen et al., 2006 | 123 | Deficiency (<30) | 67 |
| Thudi et al., 2008 | 37 | Insufficiency (<32) | 65 |
| Deficiency (<19) | 20 | ||
| Ruiz-Irastorza et al., 2008 | 92 | Insufficiency (<30) | 75 |
| Deficiency (<10) | 15 | ||
| Toloza et al., 2010 | 124 | Insufficiency (<32) | 68 |
| Souto et al., 2011 | 159 | Insufficiency (<30) | 38 |
| Deficiency (<20) | 8 | ||
| Mok et al., 2012 | 290 | Insufficiency (<30) | 96 |
| Deficiency (<15) | 27 |
Considering the effects of vit D on immunological tolerance, there is growing interest in the influence of vit D deficiency on disease activity in SLE. It is evident that vit D could physiologically contribute to the regulation of the innate and adaptive immune responses. This concept is compatible with the results of research that takes into account the association between vit D insufficiency and a more marked activity in SLE; however the results are controversial. Ruiz-Irastorza et al.,25 analyzing 92 SLE patients in Spain, concluded that vit D insufficiency was not associated with activity measured with the SLEDAI score, as occurred in the study of Toloza et al.,26 who, in 124 SLE patients, observed no correlation between disease activity and the vit D levels. Souto et al.27 recently studied 159 Brazilian patients with a diagnosis of SLE, and found no association between the score for lupus activity evaluated by SLEDAI-2K and vit D insufficiency. In our study, we failed to find an association between vit D insufficiency and lupus activity measured using the MEX-SLEDAI tool; however, it is important that we consider that most of our patients had mild-to-moderate activity, meaning that these results could not be extrapolated to populations with SLE in whom the disease activity was severe. On the other hand, Amital et al.28 analyzed several cohorts of 378 SLE patients from Israel and Europe, and concluded that the serum vit D concentrations were inversely related to disease activity; however, this correlation was weak (r=−0.12). In a study in Hungary, in which 90 SLE patients were analyzed, the serum 25(OH)D levels correlated inversely with the SLEDAI score, and with the concentrations of anti-dsDNA, of anti-Smith and of immunoglobulins.29 The present study has certain limitations. In the first place, the cross-sectional nature of the study design, with the possible presentation of susceptibility bias. In the second place, the absence of healthy controls with whom to compare prevalences of vit D insufficiency and deficiency. Moreover, in our study, it was not possible to determine anti-dsDNA, C3, C4 or other serum markers in all of the patients. Finally, as we mentioned above, most of the participants in our study had a lupus activity that had been classified as mild-to-moderate, a situation that limits the extrapolation of our results to other SLE populations.
In conclusion, women in whom SLE has been diagnosed have an elevated prevalence of vit D insufficiency, even in a tropical country like Mexico. The prevalence of vit D insufficiency shown by our findings was higher than that of most of the studies conducted in a population with SLE. In this report, it was not possible to identify factors associated with vit D insufficiency, and the 25(OH)D levels were not related to the disease activity. The consequences of vitamin insufficiency can interfere with bone metabolism, but may have no relationship to the lupus activity.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflicts of InterestThe authors declare they have no conflicts of interest.
We wish to thank Abbott Laboratories for donating the kits for determining serum vitamin D levels.
Please cite this article as: García-Carrasco M, Mendoza-Pinto C, Etchegaray-Morales I, Soto-Santillán P, Jiménez-Herrera EA, Robles-Sánchez V, et al. Insuficiencia y deficiencia de vitamina D en pacientes mexicanas con lupus eritematoso sistémico: prevalencia y relación con actividad de la enfermedad. Reumatol Clin. 2017;13:97–101.