Kawasaki disease (KD) is an acute systemic vasculitis affecting medium-sized arteries, particularly the coronary arteries. Classic diagnosis is based in prolonged fever and different clinical features, including acute arthritis. Our objective is to determine the prevalence of arthritis at the moment of the diagnosis, the response to intravenous immunoglobulin infusion and the relation with cardiac findings.
Material and methodsRetrospective study through review of medical records of 42 patients with KD from 1988 to 2013. Demographic, clinical, laboratory variables and treatment were reviewed.
ResultsMale sex was predominant (57%). Fever (100%), exanthema (92.9%), conjunctivitis (78.6%), oropharingeal changes (76.2%), cervical lymphadenopathy (71.4%), edema (52.4%) and peripheral desquamation (46.3%) were reported. Eight patients presented ecocardiography alterations (ectasia and aneurism). Acute articular involvement was reported in 7 (16%) patients, including oligoarticular (57%), monoarticular (29%) and polyarticular (14%) patterns. All patients had elevation of acute phase reactants with neutrophilia (57%) and hypoalbuminemia (71.5%), but showed a good therapeutic response to intravenous immunoglobulin, without sequelaes. Sixteen patients had incomplete KD nine males, with 100% of fever exanthema (75%), conjunctivitis (56%) and 50% of cervical lymphadenopathy. Whereas oropharingeal changes and edemas was described in 44% and 25% of them. Four patients with incomplete KD had coronary artery abnormalities.
ConclusionsAcute arthritis was an uncommon finding (16%) and resulted in no sequelae. Maybe the treatment with intravenous immunoglobulin and aspirin prevents the development of articular abnormalities and then leading to a decrease in its follow-up requirement by reumathologist. The cardiovascular sequelae, mainly incomplete KD, remains determining its prognosis. The presence of articular involvement seems not to have influence over cardiac involvement.
La enfermedad de Kawasaki (EK) es una vasculitis sistémica aguda de arterias de tamaño medio, especialmente las coronarias. Su diagnóstico clásico se basa en la presencia de fiebre prolongada y diversas manifestaciones clínicas. En la era preinmunoglobulinas, la artritis aguda constituía un hallazgo común. Nuestro objetivo es definir la prevalencia de la artritis al diagnóstico, su respuesta a inmunoglobulinas intravenosas y su relación con eventos cardiacos.
Material y métodosEstudio retrospectivo de 42 pacientes con EK desde enero de 1988 a noviembre de 2013. Las variables demográficas, clínicas, laboratorio y tratamiento fueron revisadas.
ResultadosSe encontró predominio de varones (57,1%), fiebre (100%), exantema (92,9%), conjuntivitis (78,6%), afectación oral (76,2%), adenopatía cervical (71,4%), edema (52,4%) y descamación (46,3%). Ocho pacientes presentaron clínica cardiológica y ecocardiograma con alteraciones coronarias. Siete pacientes (16%) presentaron afectación articular aguda. El 57% fue oligoarticular con predominio de tobillos, 29% monoarticular de cadera y 14% poliarticular. El 100% aumentó los reactantes de fase aguda, con neutrofilia (57%) e hipoalbuminemia (71,5%). Todos respondieron a inmunoglobulinas intravenosas, sin afectación cardiaca ni secuelas posteriores. Hubo dieciséis EK incompletos: nueve varones, con fiebre (100%), exantema (75%), conjuntivitis (56%), adenopatía cervical (50%), alteración de mucosa oral (44%) y edemas (25%). Cuatro tuvieron afectación cardiaca (un aneurisma; tres ectasias).
ConclusionesLa artritis aguda fue poco frecuente (16%) y sin secuelas posteriores. El tratamiento con inmunoglobulinas intravenosas y ácido acetilsalicílico posiblemente previene su desarrollo, disminuyendo el seguimiento por Reumatología. La afectación cardiaca, principalmente en EK incompleta, sigue marcando su pronóstico, sin hallar empeoramiento cardíaco en pacientes con artritis.
Kawasaki disease (KD) is a systemic vasculitis affecting medium-sized arteries, mainly in boys under the age of 5 years.1 It is most prevalent in Japan and Korea, especially during the winter and autumn months. It shows a predilection for the coronary arteries, a feature that originates its major complication, as it is accountable for the associated mortality and a more extensive and accelerated atherosclerosis in these children.2 At the present time, it is the main cause of acquired heart disease in pediatric patients in developed countries. Its etiology remains to be determined. An inappropriate immune response to external or infectious agents (staphylococcal and streptococcal toxins) that would act like triggering superantigens in genetically susceptible individuals and in which IgA plasma cells would play an important role.3–5 The diagnosis of KD is mainly based on certain clinical criteria, in which fever is a key, in addition to 4 of the following characteristics: (1) changes in extremities (erythema and edema in palms and soles, desquamation of the fingers and toes); (2) polymorphous exanthema; (3) bilateral nonexudative bulbar conjunctival injection; (4) changes in lips and oral cavity (erythema of the lips or mucosa, cracked lips, strawberry tongue); (5) unilateral cervical lymphadenopathy greater than 1.5cm in diameter.3 However, for the early diagnosis and treatment of KD, incomplete KD is defined as those cases in which less than 4 criteria are met, but the typical changes in the coronary arteries are identified (measured by echocardiogram or arteriography).6
Although cardiac involvement is the most important prognostic factor, arthritis has been considered a common manifestation of KD, having been reported in up to 31% of the patients in different publications from the preimmunoglobulin era.7 Moreover, a close relationship between early acute arthritis and a poorer cardiac outcome has been demonstrated in the literature, including the development of coronary aneurysms in 39% of the patients with arthritis,6 versus an overall average of 19%. The introduction of intravenous immunoglobulins (IVIG) meant an advance in the control of the disease, reducing the fever and systemic inflammatory processes, as well as the incidence of coronary artery involvement. The prognosis marked by the clinical signs of cardiac disease has led to the publication7 of a number of studies focusing on the outcome of this manifestation, and there are few articles devoted to joint involvement. The objective of this report was to define the prevalence of arthritis in KD at the time of diagnosis, its response to IVIG and its relationship to cardiac events.
Material and MethodsThis retrospective descriptive study deals with children with KD admitted to Hospital Universitario Severo Ochoa in Madrid, Spain, between January 1988 and November 2013. This report was approved by the hospital ethics committee.
We reviewed the clinical histories of all the pediatric patients who had been classified under the diagnostic code for “Kawasaki disease” or “mucocutaneous lymph node syndrome” provided by the information system of our records department. In our hospital, those children in whom KD is suspected are assessed and admitted by the pediatric department. They begin to receive rheumatology care during admission, following a request on the part of the pediatricians. The children included in this study were diagnosed with KD in accordance with the criteria proposed by the American Academy Of Pediatrics/American Heart Association.3
The clinical signs were recorded, particularly those concerning the criteria of the diagnostic classification, as were the demographic variables (age, sex, race), the season of the year in which the disease presented, history of infections and the analytical variables at diagnosis (C-reactive protein [CRP], erythrocyte sedimentation rate [ESR], biochemical data, albumin, hemoglobin, platelets, leukocytes and urinalysis results). In the analytical variables collected, any value that was outside the normal range established by the central laboratory services of our center was defined as anomalous. For acute-phase reactants, we considered CRP>10mg/L and ESR>20mm/h.
Cardiac involvement was based on the presence of abnormal findings in the electrocardiogram and/or echocardiogram. Coronary aneurysm was defined as a maximum diameter of the coronary artery greater than 3mm in patients under 5 years of age, and greater than 4mm in those aged 5 years or older.8 Moreover, they were classified by size as small (less than 5mm), medium (5–8mm) or giant (greater than 8mm). In the absence of aneurysms, coronary ectasia or dilatation was considered to be the presence of a coronary artery with a diameter greater than normal.
Joint involvement was based mainly on clinical signs, and on the findings derived from the musculoskeletal ultrasound study (since 2004). It was classified as oligoarticular if the number of affected joints was ≤4 and polyarticular with ≥5 affected joints. In addition, we included in the study the presence of any chronic clinical manifestation (sequelae) of cardiac or articular nature.
The existence of a protocol-guided therapeutic management of KD, adopted in the pediatric department of our hospital, meant that all the children received the same treatment during the first 7–10 days after diagnosis. During the acute phase, according to the protocol, patients should rest, and acetylsalicylic acid (ASA) together with IVIG are prescribed at admission. Initially the ASA dose is anti-inflammatory (80–100mg/kg body weight [bw]/day), up to 48h after the fever and clinical signs have disappeared. At that time, the amount is reduced to the levels of antiplatelet therapy, with a single dose of 3–5mg/kg bw/day.9 Intravenous immunoglobulins are given once, in a single dose of 2g/kg bw, administered slowly (duration of the injection 10h), and the fever usually disappears by the time the administration has finished. If this treatment is followed within 36h by clinical and analytical improvement, but the fever returns, a second dose of IVIG should be administered.
Perhaps ≥10% of cases of KD are resistant to IVIG, and are defined by persistence of fever and acute-phase reactants and/or clinical signs ≥36h after IVIG administration. An increase in the risk of coronary artery aneurysms has also been reported in these patients.10 As second-line therapy, the combination of intravenous corticosteroids (methylprednisolone pulses, 30mg/kg bw, for 1–3 days)9,10 with another dose of IVIG has been indicated. Despite the good clinical response, some studies conclude that corticosteroids do not manage to reduce the incidence of coronary complications.9 If resistance to treatment persists, a third line of therapy could be drugs like infliximab, abciximab, etanercept, cyclophosphamide or methotrexate, which have been found to achieve good results in several published studies.9,10
For this report, the response to the treatment administered was specified. The pediatric follow-up of each patient depended mainly on the presence of cardiac involvement.3 On the other hand, the course of the joint disease was assessed at 1, 2 and 4 weeks.
ResultsA total of 42 patients with a diagnosis of KD was identified, with a predominance of boys (24 boys and 18 girls). The age at diagnosis ranged between 3 months and 16 years, with a mean and standard deviation of 4.3 (±1.4) years. The information collected from the medical record at the time of diagnosis enabled us to classify the patients in terms of complete or incomplete KD depending on whether or not they met the classification criteria. In all, 62% (n=26) of the population was classified as having complete KD, versus 38% with incomplete KD (n=16).
The Caucasian race (n=19) predominated, versus the remaining 20% who were Latin Americans. We observed a higher prevalence with onset occurring during the winter and autumn months. Most of the patients (66.6%) had no known history of infection.
One hundred percent of the children had fever at onset. The most widespread clinical signs were exanthema (92.9%), conjunctivitis (78.6%), oral mucosa involvement (76.2%) and unilateral lymphadenopathy (71.4%), and the least common were edema (52.4%) and subsequent desquamation (46.3%). The analytical findings at diagnosis showed elevated acute-phase reactants; 92.9% had an increase in CRP (88.1±58 [38–125] mg/L) and 78.6% had an increase in ESR (69±30 [47.7–92] mm/h). Most of the children had normal leukocyte counts (83.3%), versus 14.3% with leukocytosis and 2.4% with leukopenia. Another parameter that was a marker of activity like hypoalbuminemia was observed in 42.9% of the patients at diagnosis, whereas a change in urinary sediment was found in 14.3%.
All of the patients followed the initial therapeutic protocol at diagnosis with ASA and IVIG, as we detected no clinical parameters that indicated resistance to IVIG. The major complication observed was cardiac involvement in 8 patients (19%), with evidence in the echocardiographic study of coronary ectasias and aneurysms in 6 and 2 patients, respectively. Of the 42 patients, 92.9% ultimately had no long-term complications or sequelae.
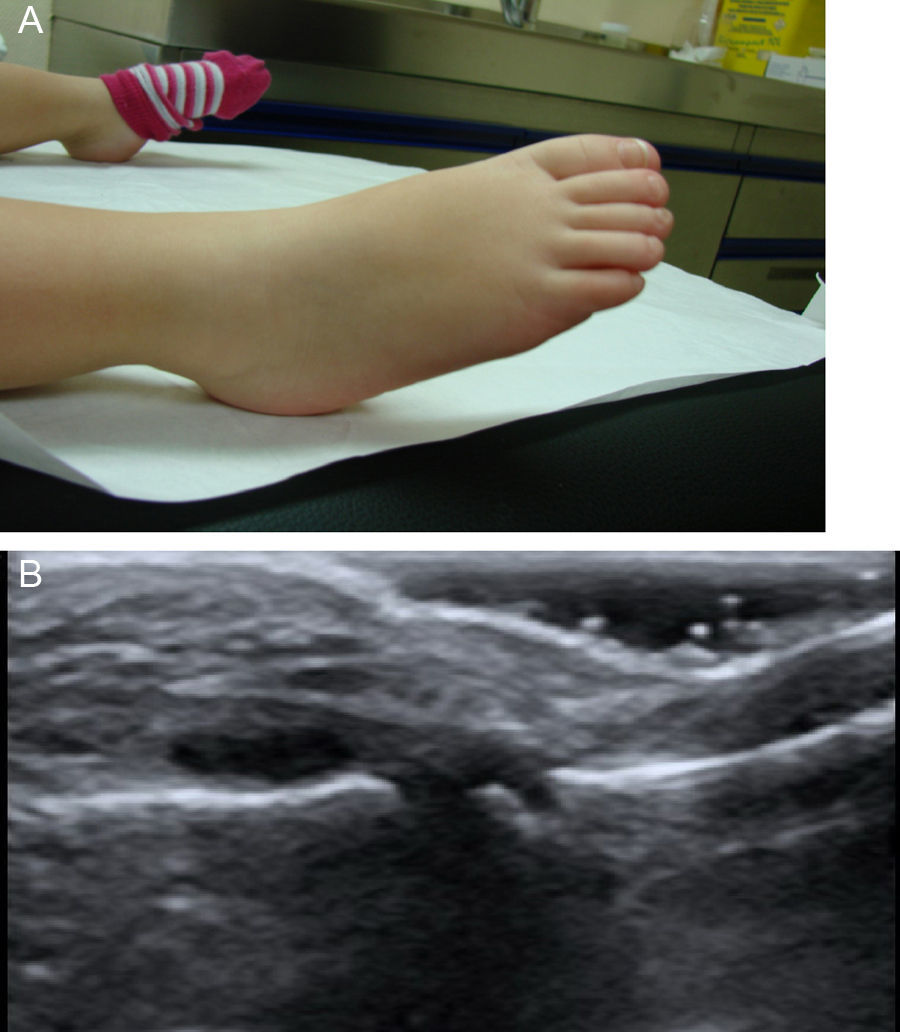
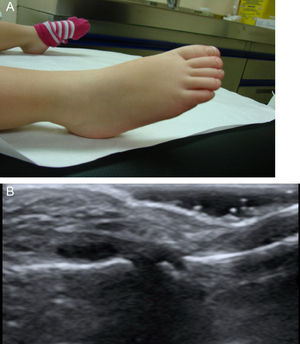
Arthritis in Patients With Kawasaki DiseaseThe presence of joint involvement was detected in only 7 of the 42 children (16%). Oligoarticular disease was predominant (57%), affecting lower limbs, knees and, especially, ankles. The next most common type was monoarticular involvement of the hip (29%), and the ultrasound study accompanying the history showed the presence of distension of the joint capsule due to effusion with no synovial hypertrophy or associated Doppler signal. Finally, only 1 patient (14%) had polyarticular disease, especially in ankles, knees, hands and proximal interphalangeal joint of right foot (Fig. 1). Ultrasound of large joints showed capsular distension provoked by effusion, with no synovial hypertrophy or Doppler signal and accompanied by severe periarticular involvement.
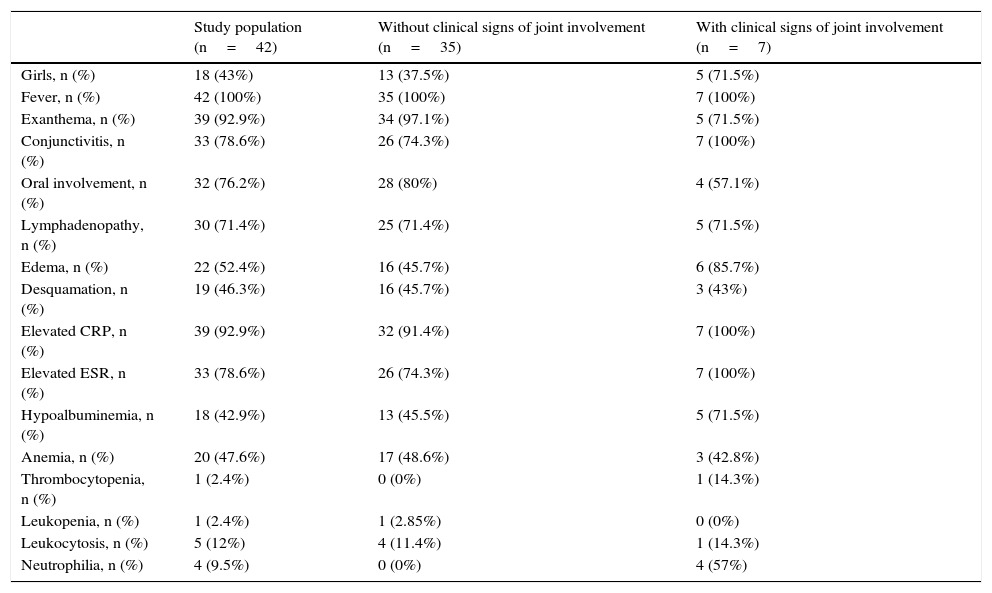
There was a predominance of girls, as can be seen in Table 1. Of the 7 patients, 5 (61%) were diagnosed as having complete KD and 2 had incomplete KD. Conjunctivitis was among the extra-articular manifestations most closely associated with joint disease (Table 1). The analytical data recorded for these patients show an increase in the ESR and hypoalbuminemia in a high proportion of patients (Table 1).
Clinical and Analytical Findings in Children Diagnosed With Kawasaki Disease.
| Study population (n=42) | Without clinical signs of joint involvement (n=35) | With clinical signs of joint involvement (n=7) | |
|---|---|---|---|
| Girls, n (%) | 18 (43%) | 13 (37.5%) | 5 (71.5%) |
| Fever, n (%) | 42 (100%) | 35 (100%) | 7 (100%) |
| Exanthema, n (%) | 39 (92.9%) | 34 (97.1%) | 5 (71.5%) |
| Conjunctivitis, n (%) | 33 (78.6%) | 26 (74.3%) | 7 (100%) |
| Oral involvement, n (%) | 32 (76.2%) | 28 (80%) | 4 (57.1%) |
| Lymphadenopathy, n (%) | 30 (71.4%) | 25 (71.4%) | 5 (71.5%) |
| Edema, n (%) | 22 (52.4%) | 16 (45.7%) | 6 (85.7%) |
| Desquamation, n (%) | 19 (46.3%) | 16 (45.7%) | 3 (43%) |
| Elevated CRP, n (%) | 39 (92.9%) | 32 (91.4%) | 7 (100%) |
| Elevated ESR, n (%) | 33 (78.6%) | 26 (74.3%) | 7 (100%) |
| Hypoalbuminemia, n (%) | 18 (42.9%) | 13 (45.5%) | 5 (71.5%) |
| Anemia, n (%) | 20 (47.6%) | 17 (48.6%) | 3 (42.8%) |
| Thrombocytopenia, n (%) | 1 (2.4%) | 0 (0%) | 1 (14.3%) |
| Leukopenia, n (%) | 1 (2.4%) | 1 (2.85%) | 0 (0%) |
| Leukocytosis, n (%) | 5 (12%) | 4 (11.4%) | 1 (14.3%) |
| Neutrophilia, n (%) | 4 (9.5%) | 0 (0%) | 4 (57%) |
Data are expressed as numbers (n) and percentages (%) of patients.
CRP, C-reactive protein; ESR, erythrocyte sedimentation rate.
With respect to treatment, the utilization of ibuprofen upon arrival at the emergency department, prior to diagnosis, was accompanied by a poor response in terms of the patients’ joints. The administration of a single IVIG dose led to the disappearance of the clinical signs of joint disease in 100% of the cases, and the patients remained asymptomatic 2 months later without sequelae. We also wish to point out that none of the patients with joint involvement had any type of associated cardiac complication.
Incomplete Kawasaki DiseaseThere were 16 patients (38%) with incomplete KD in the present study. We again found a predominance of boys (n=9 [56%]) and Caucasians (n=10 [63%]). All of the children had fever, and other widespread manifestations were exanthema (75%), conjunctivitis (56%) and lymphadenopathy (50%). Oral mucosal involvement (44%) and edemas (25%) were the least common signs. With regard to laboratory tests, we observed an elevation in acute-phase reactants (CRP in 94%, ESR in 75%), again, with a predominance of CRP. We detected no changes in urinary sediment, and few cases of leukocytosis (6%). Another finding was a higher incidence of thrombocytopenia (11%) and of anemia (56%).
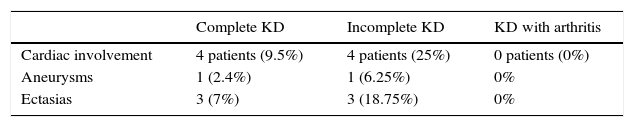
Among the patients with incomplete KD, we found cardiac involvement to be more frequent than in those with complete KD. Of the 8 patients who developed heart disease, 50% were identified as having incomplete KD, with a predominance of ectasia (n=3) versus aneurysm (n=1) (Table 2).
Cardiac Involvement in Complete and Incomplete Kawasaki Disease and in Patients With Arthritis.
| Complete KD | Incomplete KD | KD with arthritis | |
|---|---|---|---|
| Cardiac involvement | 4 patients (9.5%) | 4 patients (25%) | 0 patients (0%) |
| Aneurysms | 1 (2.4%) | 1 (6.25%) | 0% |
| Ectasias | 3 (7%) | 3 (18.75%) | 0% |
KD, Kawasaki disease.
Kawasaki disease is a systemic vasculitis that mostly affects children under 5 years of age. Our study confirms the seasonal variation of KD, with predominance of the winter months in European countries.11 The classical diagnosis is based on the presence of prolonged fever and different clinical signs, which include acute arthritis. The presence of 4 clinical manifestations accompanied by fever in approximately 75% of the children in our study substantiated the diagnosis of complete KD.4 The prevalence of arthritis during the acute phase of KD in our study was 16%, less than that described in previous reports, in which the rate was 31%,12 but greater than that observed by Gong et al. (7.5%).7
This study shows that KD is more common among boys. However, it also reveals that this is reversed in the case of joint involvement, in which girls predominate (n=5; 71%), with a mean age somewhat higher than that reflected in earlier publications.7 There are no previous reports that confirm this finding, but it could be of interest if we consider the known predominance of the female sex in inflammatory joint diseases. The presentation as oligoarticular arthritis with predominance in the lower limbs was the most common articular manifestation in KD, as was observed in our study and others.7 However, Gong et al. reported the polyarticular form with early acute arthritis lasting about 20 days, and less acute oligoarticular involvement lasting around 15 days. The rapid resolution of arthritis in 100% of the patients observed after treatment with IVIG, seems to presuppose a change in the outcome of arthritis in KD, which is currently benign. Moreover, in contrast to information provided in previously mentioned reports, no child required the administration of corticosteroids.7 We have found no studies proposing conjunctivitis as a clinical finding associated with arthritis in KD, either complete or incomplete.
In our study population, we noted the absence of parameters considered to be factors that predicted resistance to IVIG or coronary risk in KD (leukocytosis >30,000/mm,3 thrombocytopenia <300,000/L and CRP>100mm/h, as well as age <6 months), with the exception of hypoalbuminemia, that was observed in a high proportion of children with joint involvement.13 The therapeutic response in the study (81% without sequelae) is comparable to that reported in the literature.4,13
There is no doubt that cardiac involvement continues to determine the prognosis of this disease, especially in incomplete KD.14 Our study showed that, although the percentage of patients with incomplete KS was low, cardiac involvement developed mainly in those children. The study did not demonstrate the presence of cardiac involvement in any of the patients with joint disease, perhaps due to a rapid diagnosis and, consequently, to the early initiation of treatment with IVIG. This datum cannot be confirmed by the present findings as this is solely a descriptive study. A limitation to this report is the small number of patients with arthritis, which impeded us from establishing a hypothesis with a sufficient scientific basis to indicate that an improvement in the arthritis was associated with an improvement in terms of cardiac involvement.
Different mechanisms of action have been proposed for IVIG in KD.9,10 They include a modulation of endothelial cells, inhibition of endothelial cell adhesion molecules and antibodies, reduction of cytokines, macrophage and monocyte modulation, reduction in nitric oxide produced by neutrophils and T and B cell regulation. Although the mechanism of action has yet to be completely clarified, the response to early treatment with ASA plus IVIG to reduce coronary complications has been demonstrated by a number of authors.3,4,13 There is also an evident dose-response effect, that does not vary among the different commercial formulations of IVIG.10 This anti-inflammatory action of IVIG would be supported by studies that focus on the localized inflammatory activity in KD (detection of inflammatory changes in the bronchial epithelium or in synovial fluid in response to certain antigens).12,15
As occurs in cardiac involvement, we could think that IVIG could prevent or at least decrease the development of arthritis in KD and, thus, require a less intensive follow-up on the part of rheumatologists.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of InterestThe authors declare they have no conflicts of interest.
Please cite this article as: Álvarez EP, Rey F, Peña SC, Rubio A, Calvo C, Collado P. ¿La enfermedad de Kawasaki ha perdido su afectación articular? Reumatol Clin. 2017;13:145–149.