Cat scratch disease (CSD) is an infection caused by Bartonella henselae after exposure to infected fleas from cats. Commonly occurs in children and young adults with regional lymphadenopathy and presents a self-limited course.1 In patients under biological drugs the disease may be severe and disseminated. A differential diagnosis with more common granulomatous infections, like tuberculosis, should be considered in the field of immunosuppression.2
The authors report the case of a 38 year-old man diagnosed with Ankylosing Spondylitis (AS) in 2000. He presented inflammatory back pain, anterior uveitis, enthesitis, bilateral sacroiliitis and positive HLA-B27. He was first treated with infliximab (2004–2009) and was after changed to adalimumab because of recurrent uveitis. Before infliximab, he had been treated with isoniazid for latent tuberculosis, following a positive tuberculin skin test.
On December 2013, he referred asthenia and new onset of left axillar nodules lasting for two weeks without fever or other complaints. He had nodules around 30mm limited to left axillar region, without skin lesions. The cardiopulmonary, abdominal and neurological examinations were unremarkable. He denied known contacts with tuberculosis patients. He was owner of a cat which frequently scratched him but he did not recall any special lesion. Adalimumab treatment was stopped and diagnostic workup was performed.
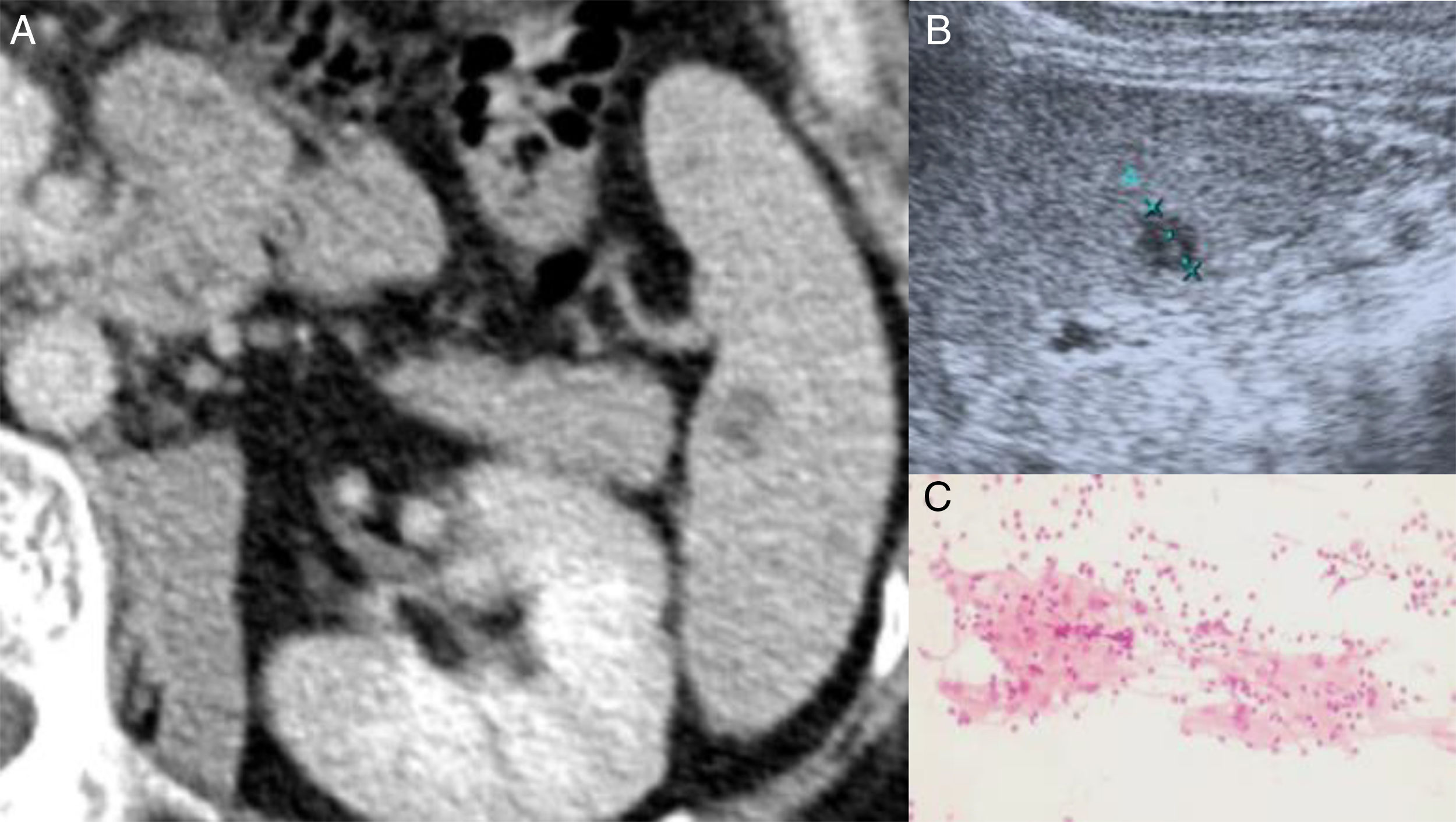
Except for Erythrocyte sedimentation rate (38mm/1st hour) and C Reactive Protein (9.0mg/L), no others relevant analytical findings were found. Quantiferon–Gold in Tube was negative and thoracic X-ray was normal. Axillar ultrasound examination revealed multiple hypoechoic nodules sized between 20–30mm. After fine needle aspiration procedure, the cytology revealed granulomatous inflammation (Fig. 1), being Ziehl Neelsen stain, culture for mycobacteria and PCR for: Mycobacterium (tuberculosis, avium and intracellulare), Borrelia Burgdorferi and Nocardia all negative. The PCR for Bartonella spp. was positive as were blood antibodies for Bartonella henselae (IgG titer 1:256; IgM <1:32). Due to CSD diagnosis, azithromycin was prescribed for 5 days. After one month, he presented complete regression of lymphadenopathies. Abdominal CT revealed splenic hypodense millimetric nodules and abdominal ultrasound showed two splenic nodules (11 and 8mm); there was no previous exam to compare (Fig. 1). Regarding procedure risks, it was decided by a multidisciplinary team to avoid splenic biopsy and ciprofloxacin during 3 weeks was prescribed. Regular ultrasound examinations revealed stability of the splenic lesions. Adalimumab was re-started on September 2014 because of exacerbation of AS activity. After more than three years, he remains asymptomatic and abdominal ultrasound revealed slight reduction of splenic nodules (8 and 5mm).
(A) Contrast-enhanced CT scan of the abdomen reveals a hypodense splenic nodule near the splenic hilum. (B) Abdominal ultrasound of the spleen depicts the same lesion as a hypoechoic nodule without posterior acoustic enhancement. (C) Cytology from lymphadenopathy's fine needle aspiration suggestive of granuloma inflammation.
We herein report, to our knowledge, the second case of CSD on adalimumab treatment. Splenic nodules raised the concern of splenic dissemination of the disease that we cannot certainly attribute to Bartonella infection since biopsy was not undertaken. Tuberculosis, being our first concern, was completely excluded. In a retrospective study of adverse event reports on TNFα blockers users, an increased risk of granulomatous infections, particularly tuberculosis, was documented, but only one case was caused by Bartonella infection.2 Later on, 10 CSD reports were described: seven patients were under TNFα blockers (one case on adalimumab), one case on abatacept and two on tocilizumab.3–11 The majority presented regional lymphadenopathy and had good therapeutic response, restarting biologics 2–3 months after. However, in three published cases, visceral infection presenting with mesenteric panniculitis, splenic nodules and, one of those, with liver granulomatosis, were documented.3,8,11
It should be stressed that new onset lymphadenopathy and granulomatosis in patients under biological therapies should raise the suspicion of CSD, particularly in the field of epidemiologic risk. The possibility of systemic dissemination should be kept in mind due to underlying immunosuppression.
Conflict of InterestThe authors declare they have no conflicts of interest.