To analyze the Health related Quality of Life (HRQoL) and physical function in rheumatoid arthritis (RA) patients and compare it with the general population. We also intended to analyze about disease activity influence in HRQoL and functional capacity, as well as determine potential determinants for these outcomes.
Material and methodsA cross-sectional study was conducted in RA patients from a university hospital of Portugal. We obtained Short Form 36, EuroQoL 5D, health assessment questionnaire, visual analog scale for pain and patient's assessment of disease activity. Comparisons between SF-36 and EQ-5D values with our population reference values were conducted using the Mann–Whitney test. Data were compared in different levels of disease activity, using Kruskal Wallis test and Fisher's exact test. A multiple regression analysis was conducted to identify the potential determinants of outcomes.
ResultsRA sample showed significantly lower values than the portuguese general population on physical summary measure of SF-36 (median=32 vs. 50, p<0.001) and EQ-5D (median=0.620 vs. 0.758 respectively; p<0.001). Lower disease activity levels had better PROs and this was true even when compared patients achieving remission with those in low disease activity. The HAQ (r2=67%), VAS-P (r2=62%) and VAS-DA (r2=58%) were the variables that strongly related to SF-36. Considering HAQ, the strongest relation was found with VAS-P, VAS-DA and age (r2=60%, 61% and 33%, respectively). Multiple regression analysis identified HAQ, VAS-P and educational status as determinants of the HRQoL; age, female gender, employment, VAS-P and VAS-DA as determinants of physical function.
ConclusionImpairment of HRQoL in RA patients is enormous. We found significant differences between different levels of disease activity, showing higher HRQoL and functional capacity at lower disease activity levels.
Analizar la evaluación del Health related Quality of Life (HRQoL) y la función física en pacientes con artritis reumatoide (AR) y compararla con la población general. También se pretende analizar la influencia de la actividad de la enfermedad en el HRQoL y la capacidad funcional, así como definir los determinantes potenciales de estos resultados.
Material y métodosSe realizó un estudio transversal en pacientes con AR de un hospital universitario de Portugal. Se obtuvieron los cuestionarios Short Form 36, EuroQol 5D, Health Assessment Questionnaire, la escala analógica visual de dolor y la evaluación de la actividad de la enfermedad. Las comparaciones entre los valores de SF-36 y EQ-5D con los valores de referencia de nuestra población se conducierón utilizando el test de Mann-Whitney. Los datos se compararon en diferentes niveles de actividad de la enfermedad utilizando el test de Kruskal Wallis y la prueba exacta de Fisher. Se realizó un análisis de regresión múltiple para identificar los determinantes potenciales de los resultados.
ResultadosLa muestra de AR mostró valores significativamente más bajos que la población general portuguesa en la medición física de SF-36 (mediana=32 vs. 50, p<0,001) y EQ-5D (mediana=0,620 vs. 0,758, respectivamente, p<0,001). Los pacientes con niveles de actividad de la enfermedad más bajos tuvieron mejores PRO y esto fue cierto incluso cuando se compararon los pacientes que alcanzaron la remisión con aquellos en baja actividad de la enfermedad. El HAQ (r2=67%), VAS-P (r2=62%) y VAS-DA (r2=58%) fueron las variables fuertemente relacionadas con SF-36. Con respecto a el HAQ, se encontró una relación más fuerte con VAS-P, VAS-DA y la edad (r2=60%, el 61 y el 33%, respectivamente). El análisis de regresión múltiple identificó el HAQ, el VAS-P y el nivel de estudios como determinantes del HRQoL y la edad, el género femenino, el empleo, VAS-P y VAS-DA como determinantes de la función física.
ConclusiónEl deterioro del HRQoL en los pacientes con AR es enorme. Se encontraron diferencias significativas entre los diferentes niveles de actividad de la enfermedad, mostrando mayor HRQoL y capacidad funcional en niveles más bajos de actividad de la enfermedad.
The prevalence of RA has been estimated at about 0.5–1.0% worldwide1 and 0.7% in Portugal2 and the majority of patients are in a working age.3
RA is associated with pain, fatigue, functional disability and deterioration of emotional state and if not treated early, can lead to irreversible structural and functional damage. For these reasons RA represents health-related quality of life (HRQoL) and economic burden to patients and society.4
Currently, there are many treatment options although none of them completely treat RA. Thus, the aim of treatment is not only to achieve remission but minimize the disease consequences on patients by increasing the HRQoL and physical function.5–7 HRQL is an additional metric to assess patients’ subjective perspective on their experience of pain and its adverse impact on their lives.8,9
With the advent of the biologic therapy, a growing attention was dedicated by physicians to the precise evaluation of outcomes of therapy.10 Disease activity is the strongest predictor of disability and that clinical and laboratory markers are important in their assessment and treatment response. However, patient's perspective of RA disease worsening or flare represents an experience extending beyond standard clinical outcome measures.11,12 Some authors consider that outcomes of a clinical intervention obtained by the patient i.e., patient-reported outcomes (PRO) are seemed to be of more importance than any other outcomes like clinical-reported. Several data as impact of disease on condition on daily life can be obtained only from patient.
Thus, PROs are not only an important aim of treatment but also an important long-term prognostic factor.13,14 Beyond this, increased emphasis it is given to the fact that these tools also allow a cost-effectiveness analysis of treatments.15 Thus, we assisted a growing interest by an assessment not only based on objective parameters, but also an assessment that includes the patient's subjective well-being.16
Various generic and specific scales are used for evaluating PROs. These scales not only measure the effectiveness of the treatment as well as assess whether this effectiveness is truly significant in the patient's perspective.
With disease modifying antirheumatic drugs (DMARDs), clinical symptoms as well as radiological joint damage are prevented. Achieving clinical remission would ideally be associated with achieving PROs comparable to those in the general population. To date, there have been limited studies about how the changes in the disease activity and classical clinical data relate with changes in the various PROs.17 Furthermore, it is not clear yet whether aiming for remission is worthwhile, especially when compared with low disease activity.
The present study aims (1) to analyze the HRQoL in a sample of patients with RA under biological therapy; (2) to compare HRQoL of patients with RA to that of the portuguese general population; (3) to inquiry about disease activity influence in HRQoL and functional capacity; (4) to determine potential determinants for these outcomes.
MethodsStudy design and patientsWe performed a cohort cross-sectional study. The evaluating period was between October 2013 and July 2014 from a university hospital in the north of Portugal. Consecutive RA patients were recruited by participating study physicians during routine clinic visits. We included adult RA patients, diagnosed according to the 2010 ACR/EULAR classification criteria, under biological therapy. Patients lacking ability to answer the questionnaires were excluded. Additional exclusion criteria were not applied. A total of 154 patients were included. The medical ethics committee approved the study protocol.
Outcomes measuresHRQoL was captured by the Short Form Health Survey (SF-36) and EQ-5D.
SF-36 measures physical and mental health on eight subscales (physical functioning – PF, physical role functioning – PRF, bodily pain – BP, general health perceptions – GHP, vitality – VT, social role functioning – SRF, emotional role functioning – ERF, mental health – MH), which are summed up in two scales, the physical and mental summary measures (PSM and MSM). Each scale comprises four dimensions scored from 0 to 100, with higher scores indicating better HRQoL. We calculated summary scores of the PCS and MCS, as well as single domain scores. These component scores are standardized on the basis of worldwide population norms to a mean of 50 and a standard deviation of 10.18,19
EQ-5D is applicable to a variety of different illnesses and treatments and provides a simple descriptive profile and a single index value for health status. The five dimensions included in the EQ-5D are: mobility; self-care; usual activities; pain/discomfort and anxiety/depression. Each dimension has three levels (none, some or major problems) and together defines 243 health states (3 to the power of 5 gives the 243 possible combination), to which has been added “unconscious” and “dead” for a total of 245 health states. EQ-5D scores range between −0.594 and 1 (full health).20
Limitations in activities of daily life were assessed by portuguese version of the Health Assessment Questionnaire (HAQ) which comprises eight categories. A score of 0 (no difficulty), 1 (some difficulty), 2 (much difficulty or need of assistance) or 3 (unable to perform) is given to each question; the highest score in each category represents the score for that category. The sum of scores is then divided by the number of categories, yielding a total score ranging from 0 (best) to 3 (worst).21
Visual Analog Scales (VASs) were used, on which patients had to indicate on a scale from 0 (none) to 100 (worst) mm their rating of disease activity (VAS-DA) and pain (VAS-P).
Data collectionData collection was performed in the rheumatology day-care hospital unit and through the longitudinal national database “reuma.pt”. The collected variables were C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), numbers of swollen and tender joints by using a 28-joint count (SJC28, TJC28); VAS-P and VAS-DA, and HAQ. Composite indices, such as the simplified disease activity indice (SDAI) and the Disease Activity Score using 28-joint counts (DAS28-4V) were calculated by informatic system. Age, gender, level of education, marital and employment status as well as additional disease characteristics – disease duration, rheumatoid factor (RF) and anti-cyclic citrullinated peptide antibody (anti-ACCP) status, extra-articular manifestations (rheumatoid nodules, ocular, rheumatoid vasculites and pleuropulmonary) and DMARDs use – were also recorded. The application of questionnaires, available in the computer system, was held through face-to-face interviews.
Statistical analysesIn the description of socio-demographic and clinical characteristics as well as in the description of PROs (eight domains of SF-36, PSM, MSM, EQ-5D, HAQ, VAS) were used means and standard deviations for continuous variables with symmetrical distribution and median and range (minimum and maximum) for the variables continuous with asymmetric distribution. Categorical variables were expressed as proportions. To compare HRQoL of our RA patients to that of the portuguese general population, we used SF-36 published data by Ferreira et al.22 Measures of central tendency were compared using the Mann–Whitney test. To investigate whether outcomes are different at different levels of disease activity we divided sample according to level of disease activity by SDAI: remission (REM) SDAI≤3.3, low disease activity (LDA) 3.3<SDAI≤11, moderate disease activity (MDA) 11<SDAI≤26 and high disease activity (HDA) SDAI>26. The comparison of outcomes was performed using Kruskal Wallis test and Fisher's exact test, for medians and categorical variables, respectively. To assess the relationship between clinical data and the PROs, Spearman correlations (ρ) were performed. To identify explanatory variables of HRQoL and functional capacity we conducted a multiple linear regression (stepwise selection) and included variables statistically significant in univariate analysis and those considered clinically relevant. We assessed correlations between the continuous variables and HRQoL with Spearman correlation to check for collinearity. Linear regression model assumptions were examined and satisfied. Residual plots were examined for goodness of fit. Analyses were performed using the program SPSS Version 21. The p-values below 0.05 were considered significant.
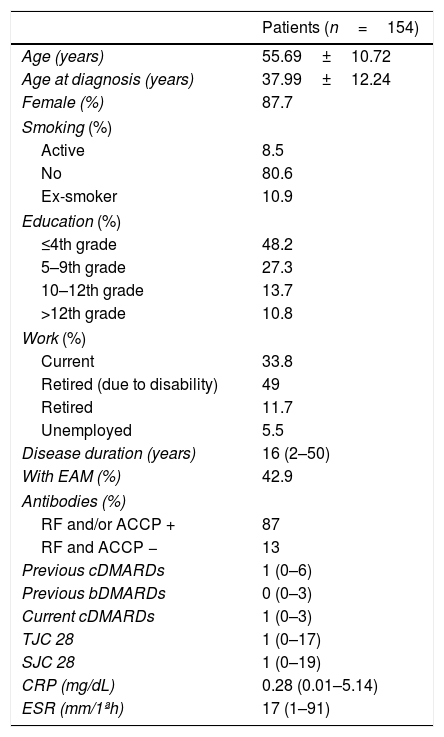
ResultsSocio-demographic, clinical and laboratory characteristics are documented in Table 1. At the moment of this study we found that 8.4% (n=13) were in REM, 53.2% (n=82) had LDA, 33.8% (n=52) had MDA and 4.5% (n=7) had HDA. Most patients (n=107) were under the first biological agent and the median time under the drug was 6 years (range 0–10). Eight patients (5.2%) were under the current drug for 10 years and 27 (17.5%) for 2 years or less.
Demographic and clinical patient data.
| Patients (n=154) | |
|---|---|
| Age (years) | 55.69±10.72 |
| Age at diagnosis (years) | 37.99±12.24 |
| Female (%) | 87.7 |
| Smoking (%) | |
| Active | 8.5 |
| No | 80.6 |
| Ex-smoker | 10.9 |
| Education (%) | |
| ≤4th grade | 48.2 |
| 5–9th grade | 27.3 |
| 10–12th grade | 13.7 |
| >12th grade | 10.8 |
| Work (%) | |
| Current | 33.8 |
| Retired (due to disability) | 49 |
| Retired | 11.7 |
| Unemployed | 5.5 |
| Disease duration (years) | 16 (2–50) |
| With EAM (%) | 42.9 |
| Antibodies (%) | |
| RF and/or ACCP + | 87 |
| RF and ACCP − | 13 |
| Previous cDMARDs | 1 (0–6) |
| Previous bDMARDs | 0 (0–3) |
| Current cDMARDs | 1 (0–3) |
| TJC 28 | 1 (0–17) |
| SJC 28 | 1 (0–19) |
| CRP (mg/dL) | 0.28 (0.01–5.14) |
| ESR (mm/1ªh) | 17 (1–91) |
EAM, extra-articular manifestations; RF, rheumatoid factor; ACCP, anti-cyclic citrullinated peptide antibody; cDMARDs, classic disease-modifying anti-rheumatic drugs; bDMARDs, biological disease-modifying anti-rheumatic drugs; SJC28, swollen joints by using a 28-joint count; TJC28, tender joints by using a 28-joint count; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate.
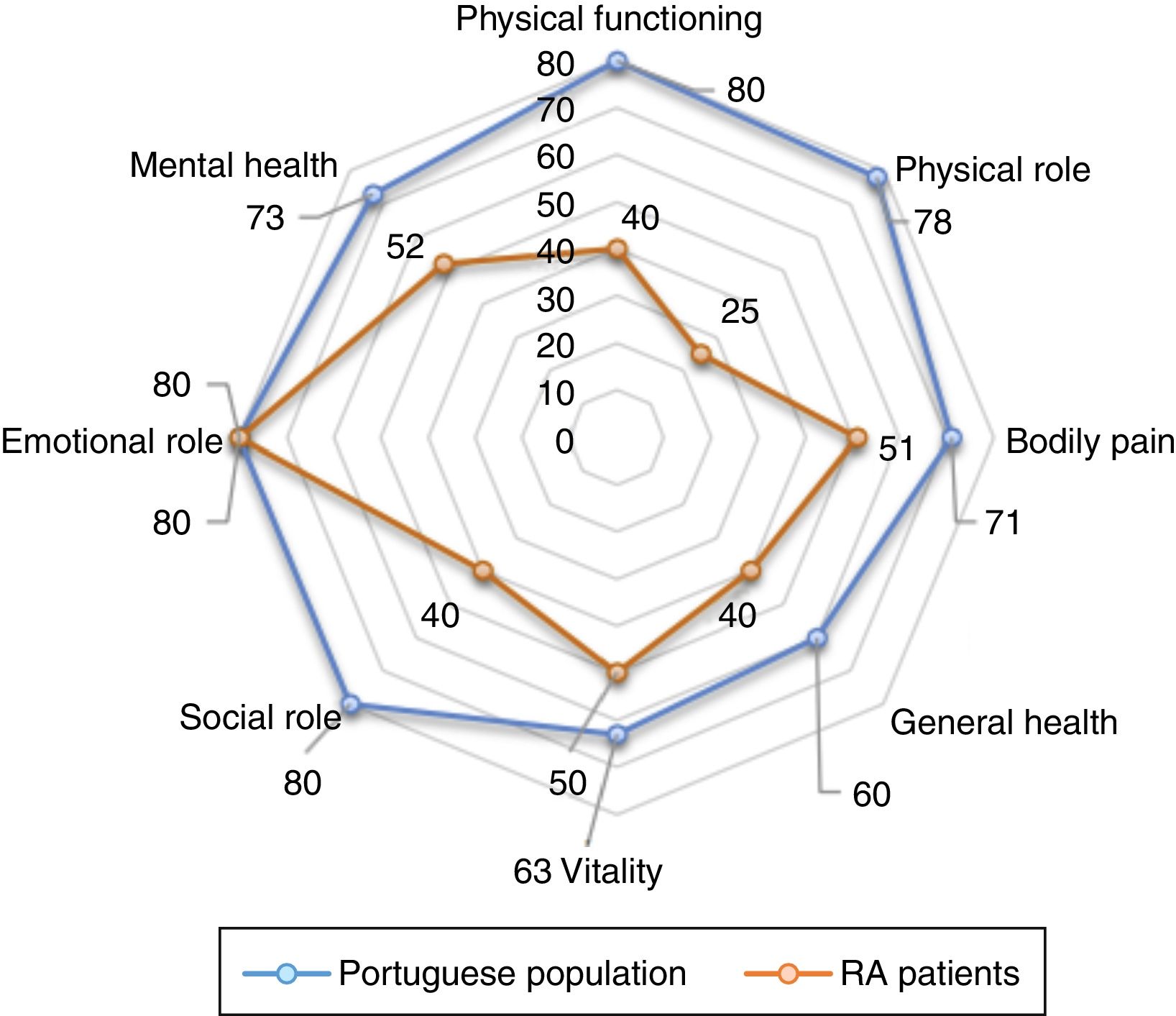
After analysing all domains of the SF-36 we observed that the domain with lowest score was the “Physical Performance” (median 25) followed by the domain “General Health” and “Physical Functioning” (median 40 for both). Mental domains showed higher values; among them, domain “Social Functioning” showed the lowest score (median 40; range 3–53). The domain “Role-Emotional” had a median of 100 (range 0–100) (Table 2).
Patient-reported outcomes (PRO).
| Sample (n=154) | REM (n=13) | LDA (n=82) | MDA (n=52) | HDA (n=7) | p Value between REM and LDA* | p Value between all groups** | |
|---|---|---|---|---|---|---|---|
| VAS-P | 50 (0–100) | 6 (0–30) | 32.5 (0–95) | 60 (0–100) | 70 (50–100) | <0.001 | <0.001 |
| VAS-GH | 50 (0–100) | 0 (0–20) | 40 (0–80) | 60 (20–100) | 70 (50–100) | <0.001 | <0.001 |
| HAQ | 1.25 (0.00–2.88) | 0.50 (0.00–1.13) | 1.13 (0.00–2.75) | 1.63 (0.38–2.88) | 2.00 (1.13–2.75) | <0.001 | <0.001 |
| EQ-5D | 0.620 (−0.349–1.000) | 0.848 (0.708–1.000) | 0.673 (−0.349–1.000) | 0.516 (−0.349–0.812) | 0.516 (0.053–0.760) | <0.001 | <0.001 |
| SF-36: PH | |||||||
| PF | 40 (0–100) | 75 (45–95) | 40 (0–100) | 30 (5–70) | 30 (5–60) | <0.001 | <0.001 |
| PRF | 25 (0–100) | 100 (0–100) | 50 (0–100) | 25 (0–100) | 0 (0–75) | 0.002 | <0.001 |
| BP | 51 (0–100) | 84 (10–100) | 52 (0–100) | 41 (0–100) | 12 (10–62) | 0.011 | <0.001 |
| GHP | 40 (0–72) | 45 (35–72) | 40 (10–72) | 35 (15–65) | 35 (0–50) | 0.020 | 0.009 |
| SF-36: MH | |||||||
| VT | 50 (20–70) | 50 (40–55) | 50 (20–70) | 50 (30–60) | 45 (45–55) | 0.655 | 0.815 |
| SRF | 40 (3–53) | 51 (28–52) | 40 (13–53) | 40 (3–53) | 51(15–53) | 0.118 | 0.153 |
| ERF | 100 (0–100) | 100 (0–100) | 100 (0–100) | 67 (0–100) | 100 (0–100) | 0.023 | 0.054 |
| MH | 52 (0–100) | 84 (28–100) | 56 (0–100) | 44 (4–100) | 44 (32–72) | 0.004 | 0.001 |
| PSM | 32 (14–66) | 54 (18–62) | 37 (15–66) | 24 (14–53) | 20 (14–45) | <0.001 | <0.001 |
| MSM | 52 (23–69) | 51 (33–64) | 52 (23–66) | 51 (36–69) | 53 (43–65) | 0.820 | 0.839 |
VAS-P, visual analog scales of pain; VAS-GH, visual analog scale for general health; HAQ, Health Assessment Questionnaire; EQ-5D, EuroQol five dimensions questionnaire; SF-36, Short Form Health Survey; PH, physical health; PRF, physical role functioning; BP, bodily pain; GHP, general health perceptions; VT, vitality, SRF, social role functioning; ERF, emotional role functioning; MH, mental health; PSM, physical summary measures; MSM, mental summary measures.
Values presented as medians (range). P-value between different groups of disease activity by *Mann-Whitney and ** Kruskal-Wallis test.
Likewise, we found that in SF-36 summary measure, PSM was lower than MSM (median 32 vs. 52). With the application of EQ-5D also observed a low score (median 0.620, range −0.349 to 1.000). In our sample, 5% of patients (n=6) had scores on the EQ-5D less than zero (states worse than dead). Median score was 1.250 (range 0.000–2.875) for HAQ (Table 2).
When comparing primary outcomes between genders we found significant statistical differences only for the HAQ in which woman presented worse results (median 1.38 vs. 0.88; p=0.024). There were no significant differences in PROs when evaluated according to disease duration (≤10 or >10 years), RF and ACCP status, presence of extra-articular manifestations or current biological therapy.
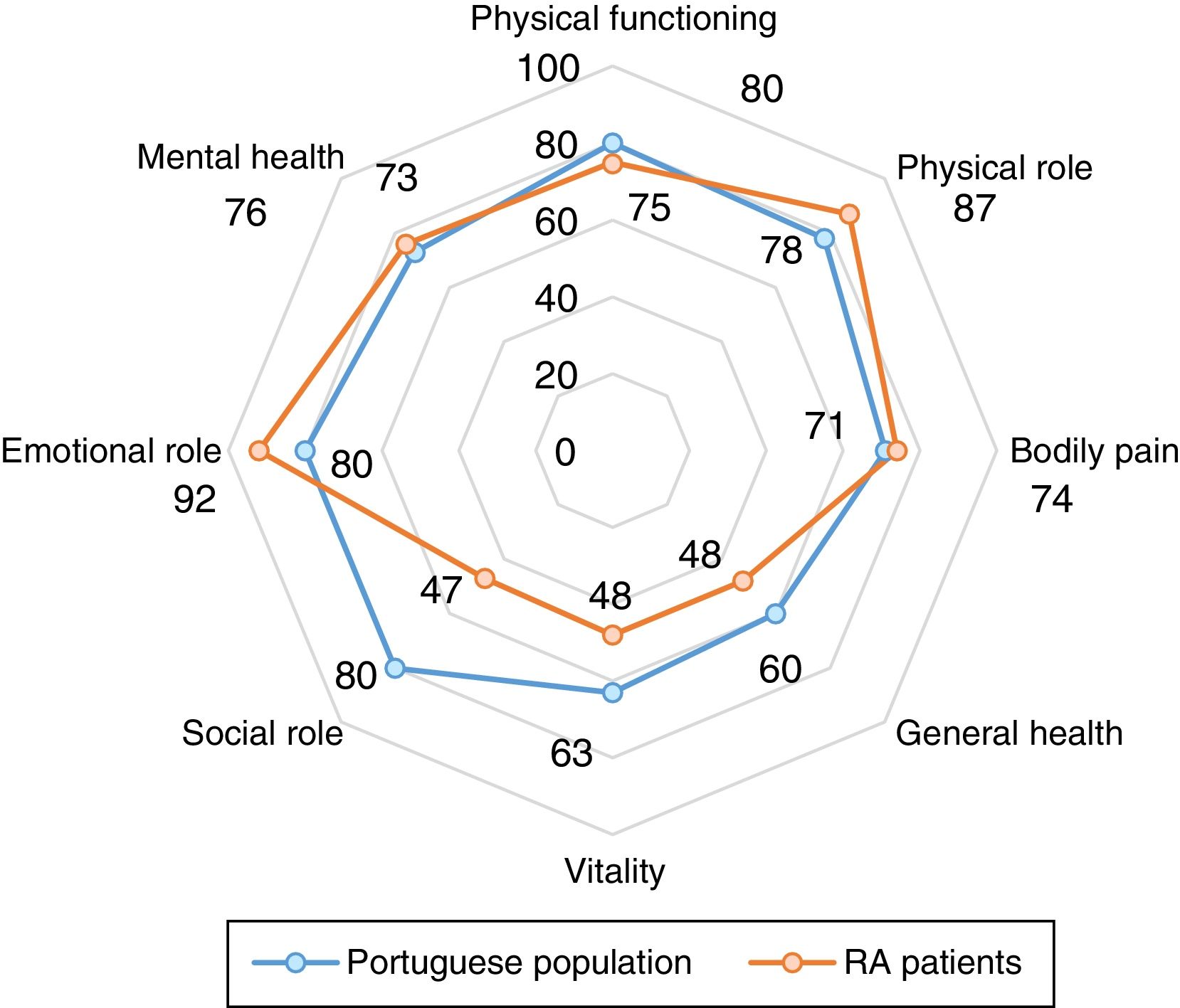
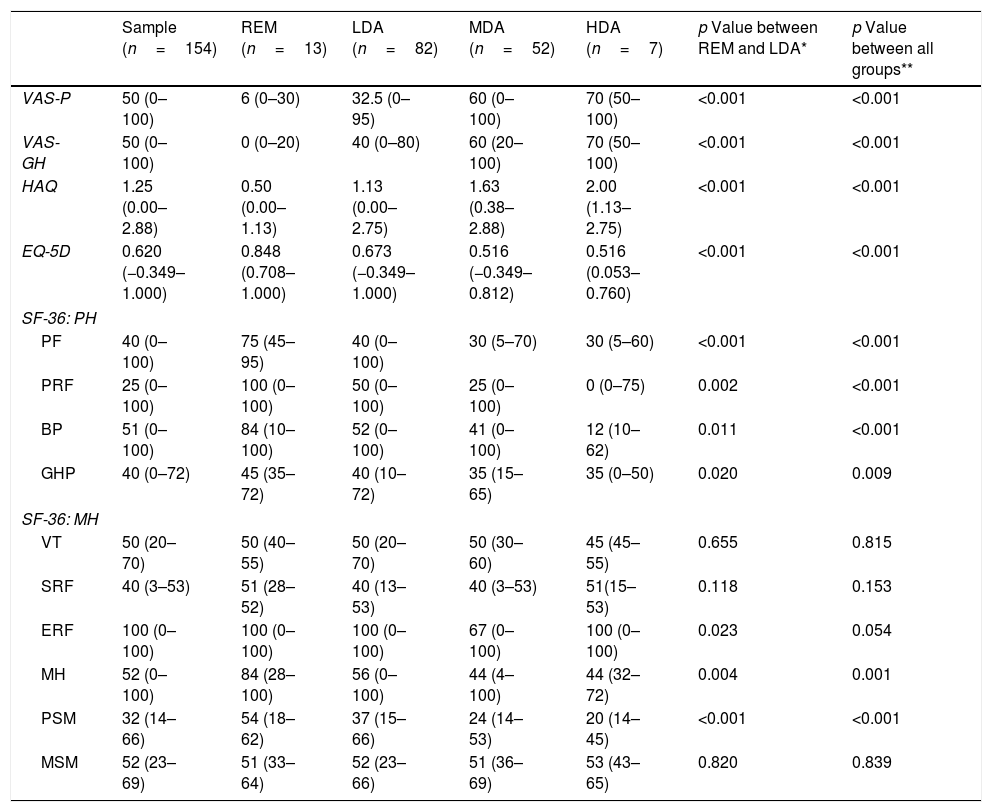
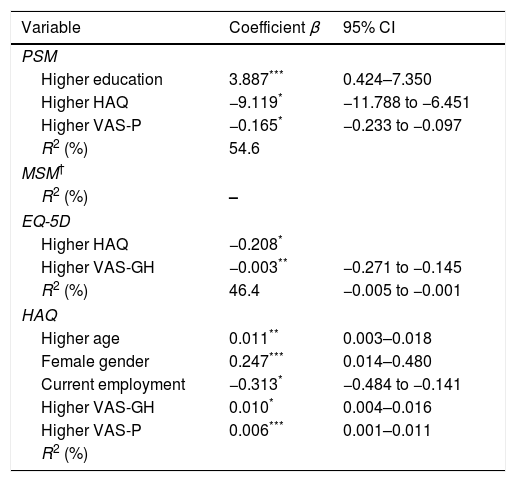
Comparison of HRQoL of RA patients with general Portuguese populationHRQoL of our RA patients is lower than the general Portuguese population. Our patients had significantly lower scores in all domains of the SF-36, except for “Emotional role functioning” (Fig. 1). Regarding summary measures there is a significant difference in MSF (median 32 vs. 50, p<0.001) but not in the MSM (median 50 vs. 52, p=0.503). The same was observed through EQ-5D in which our patients had lower median score than general Portuguese population (0.620 vs. 0.758, p<0.001). Comparing the HRQoL of general Portuguese population with our patients in clinical remission, we found similars scores for the several individual domains and the summary measures of SF-36 (Fig. 2).
Regarding EQ-5D values, we also found identical values between REM group and the general population (0.848 vs. 0.758).
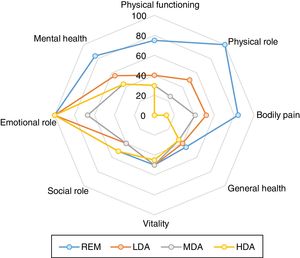
HRQoL and functional disability correlate significantly with disease-activity levelsThe performed analysis by disease activity groups showed significant differences in all applied instruments (Table 2).
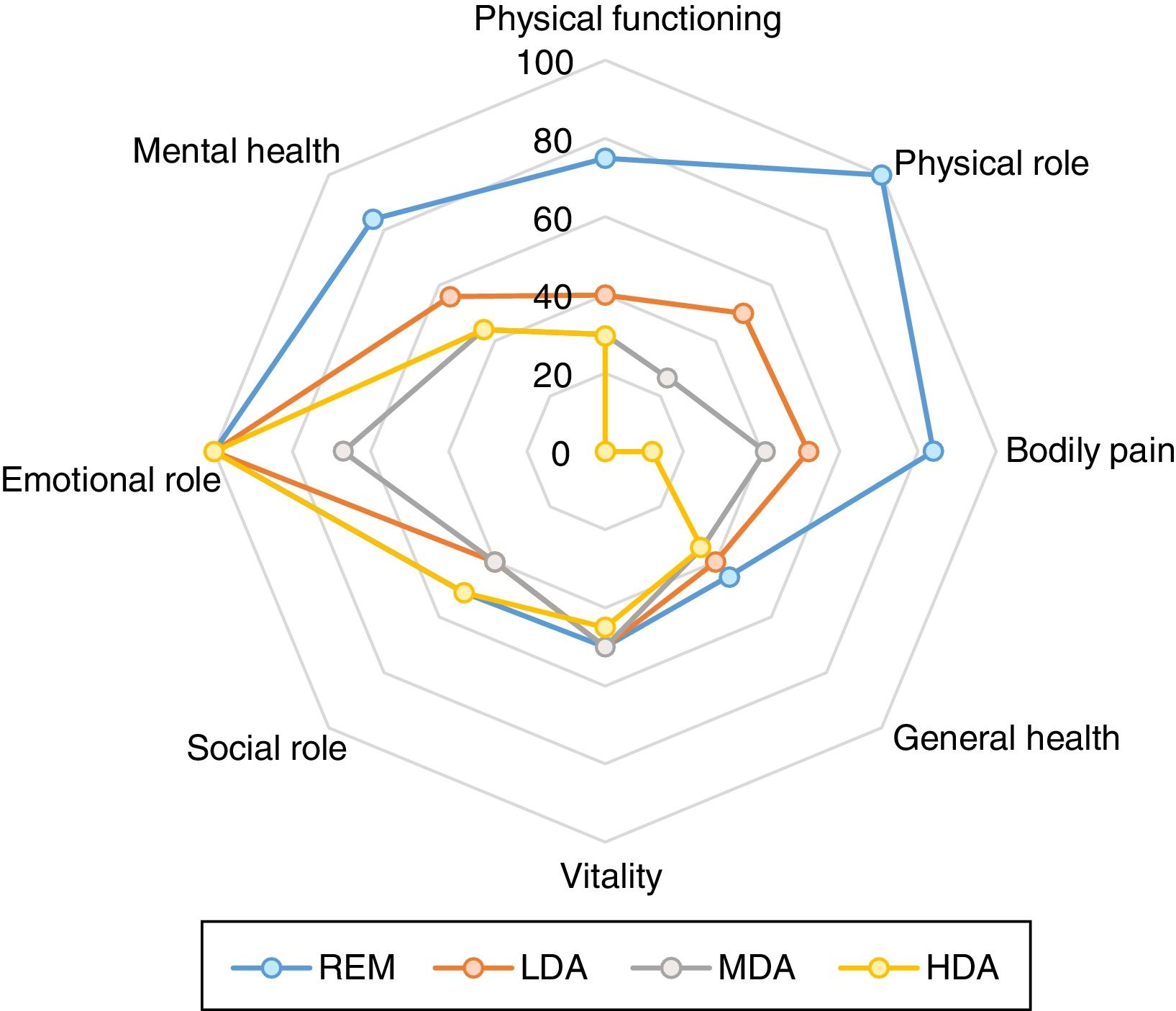
When we access the functional capacity using the domains of physical health of the SF-36 (PF, PRF, BP and GHP) we also found statistically significant difference, showing worst scores with highest disease activity (p<0.001 for all analyzes). For the SF-36 mental health domains we only observed progressive decrease in the score with increase of level of activity disease on the “mental health” domain (p=0.001) (Table 2 and Fig. 3). The same was true for the SF-36 summary measures, i.e., decreasing scores of PSM with increasing levels of disease activity (p<0.001) and overlapping MSM values (Table 2). When we assessed potential differences in HRQoL by other measures, such as EQ-5D, we found very similar results (p<0.001) (Table 2).
Comparison of SF-36 among levels of disease activity determined by SDAI (remission (REM SDAI≤3.3; low disease activity (LDA) 3.3<SDAI≤11; moderate disease activity (MDA) 11<SDAI≤26 and high disease activity (HDA) SDAI>26)). Significant differences of different domains of SF-36 was found (p≤0.001 for all analyses by Kruskal Wallis test).
Comparing functional disability by HAQ at the four levels of disease activity, we observed significant differences between groups, showing a HAQ increase with increasing disease activity (p<0.001) (Table 2).
In order to compare the benefit of remission with state of low disease activity with respect to the PRO we directly compared REM group with LDA (Table 2). In this analysis we observed a statistically significant difference in all PRO (SF-36, EQ-5D, HAQ and VAS) except in “Vitality” and “Social role functioning” domains and mental summary measure of the SF-36.
HRQoL and functional disability have a weak correlation with clinical and laboratory dataWe evaluated the relationship between the various clinical and laboratory parameters with the PRO through the Spearman correlation (ρ).
The analyzed clinical and laboratory variables were: current age and age at disease onset, disease duration, education level, CRP, ESR, tender and swollen 28-joint count, DAS28 change, VAS-P and VAS-GH. We also studied the relationship between the PSM of SF-36 and initial and actual HAQ.
For PSM we concluded that variables with strongest relationship were those that correspond to other PRO, namely current and initial HAQ (ρ=−0.67 and −0.48, respectively), VAS-P (ρ=−0.62) and VAS-GH (ρ=−0.58) (p<0.001 for all analyses). The relationship with tender and swollen 28-joint count, although significant, was much lower (ρ=−0.35 and −0.20, respectively). There was no correlation with ESR (ρ=0.02, p=0.84) nor with CRP (ρ=0.07, p=0.365). For MSM no significant correlations were identified.
The HAQ also showed weak correlation with ESR, CRP and 28-joint count. The strongest correlations were observed with PSM of SF-36 (ρ=0.674), VAS-GH (ρ=0.609), VAS-P (ρ=0.601) and initial HAQ (ρ=0.595) (p<0.001 for all analyzes).
Again, we verified that variables with strongest correlation with EQ-5D were other PRO (VAS-GH ρ=−0.573 and VAS-P ρ=−0.560, p<0.001 for both) and not the clinical or laboratory classics data.
There was not a significant association between the SF-36, EQ-5D and HAQ with the variation of DAS28-4V.
Determinants of HRQoLThe potential explanatory variables considered for summary measures of the SF-36, EQ-5D and HAQ were age, gender, level of education, employment status, disease duration, VAS-GH, VAS-P, tender and swollen 28-joint count, ESR, CRP and DAS28-4V change. For SF-36 and EQ-5D we also included HAQ questionnaire. The regression coefficients are documented in Table 3. HAQ, VAS-P and level of education were retained as explanatory variables for PSM. The explained variance (R2) was 55% (p<0.001) and HAQ had the stronger effect on PSM (β=−9.119, p<0.001). For MSM, the model has not retained any significant explanatory variable. Therefore, we found no demographic or clinical variable to explain mental health.
Results of multiple linear regression of summary measures of SF-36, EQ-5D and HAQ against significant variables.
| Variable | Coefficient β | 95% CI |
|---|---|---|
| PSM | ||
| Higher education | 3.887*** | 0.424–7.350 |
| Higher HAQ | −9.119* | −11.788 to −6.451 |
| Higher VAS-P | −0.165* | −0.233 to −0.097 |
| R2 (%) | 54.6 | |
| MSM† | ||
| R2 (%) | – | |
| EQ-5D | ||
| Higher HAQ | −0.208* | |
| Higher VAS-GH | −0.003** | −0.271 to −0.145 |
| R2 (%) | 46.4 | −0.005 to −0.001 |
| HAQ | ||
| Higher age | 0.011** | 0.003–0.018 |
| Female gender | 0.247*** | 0.014–0.480 |
| Current employment | −0.313* | −0.484 to −0.141 |
| Higher VAS-GH | 0.010* | 0.004–0.016 |
| Higher VAS-P | 0.006*** | 0.001–0.011 |
| R2 (%) | ||
PSM, physical summary measures; HAQ, Health Assessment Questionnaire; VAS-P, visual analog scales of pain; MSM, mental summary measures; EQ-5D, EuroQol five dimensions questionnaire; VAS-GH, visual analog scale for general health.
HAQ and VAS-GH were retained as explanatory variables for EQ-5D and the model's explanation of the variance was significant (46.4%). As observed for PSM, HAQ was the variable with stronger effect on the EQ-5D (β=−0.208, p<0.001).
Studying HAQ determinants, we found that higher age, female gender, current employment, VAS-P and VAS-GH explained 50% of the variation. Presence of current employment was the variable with the greatest impact on functional capacity (β=−0.313).
Clinical and laboratory classic data (joint count, acute-phase reactants and DAS28-4V change) showed no explanatory effect on HRQoL and functional capacity of these patients.
DiscussionThe present study provides information on the effect of RA on HRQoL and functional capacity, in a real world setting.
Our results corroborate previous studies showing that patients with RA had not only significant physical function and mental health impairment but also poor HRQoL when compared to general Portuguese population.23 Differences are particularly notable on physical domains of SF-36 however, RA patients also present levels of mental health subscales significantly lower than general population.
In our study we verified values of EQ-5D corresponding to states of “full health”. Several factors may explain this finding. One of the disadvantages of the EQ5D is that may suffer from ceiling effect. The questionnaire is not sensitive enough to discriminate between different levels of health because the dimensions are probably not sufficiently disaggregated, leading individuals to respond at the highest level. These finding may indicate that individuals with significant morbidity are misclassified as in full health on the EQ-5D descriptive system.
When we evaluated disease activity influence in HRQoL and functional capacity, we found significant negative correlation (HRQoL and functional impairment with higher disease activity levels). A interesting finding in our study was to note a stronger correlation between SF-36 and measures of disability and pain (HAQ and VAS-P) than with measures directly related to disease process (ESR, CRP, joint count). Another finding was the benefit found in these outcomes when REM status was compared directly with LDA. Thus, indicators of quality of life, functional capacity, pain and appreciation of global health patient-reported complementing the clinical evaluation.
With regard to the potential determinants of these outcomes, we verified that older age and female gender was associated with higher HAQ score. This data confirms previous results.24 Physical function declines with age and it is known that women have lower threshold for pain and that men have more physical strength than women. Current employment was also related to better HAQ scores.
With regard to HQoL, we identified higher education level and functional capacity as potential determinants.
Unlike other results,18 the duration of disease showed no significant linear relationship with none of the outcomes.
Several limitations of our study should be addressed. We studied a non-representative sample of the Portuguese population. Although the sample was from a reference center of Portugal, this is a convenience sample and the sample size is potentially small. This is a cross-sectional analyses; therefore, prevents to establish directionality of interpretation, i.e., unknown if group of patients in remission and low disease activity showed better SF-36, EQ-5D and HAQ when presented higher levels of disease activity. Other limitation is the fact that the joint damage was not assessed. This becomes relevant for two reasons. We observed that most patients studied have disease for over 10 years and we know that in late disease the functional capacity is most associated with joint damage.18 The other reason is when we compare our sample with general population and observed that although all domains that compose the PSM were significantly lower, the difference is more pronounced for “physical functioning” and “physical role functioning” domains than for the “bodily pain” domain. Many patients with inactive disease but with important damage have functional limitations but do not refer pain complaints. Finally, the Portuguese population used to compare the HRQoL presents some characteristics that may affect the validity of the results. About 40% of the people interviewed report some type of pathology (namely musculoskeletal). However, this data has not been clinically confirmed based solely on patient-reported. Data such as gender and age were similar. Most were female and were between 30 and 69 years of age. In this way, we believe that there was no great influence on the results.
Strengths of the present analyses were the inclusion of a sample of patients with a broad range of clinical characteristics, rather than the more homogeneous populations of the trials; the focus on the more-stringent SDAI remission criteria, which allowed the recognition of the differences between REM and all other states, including LDA; it addressed quality of life not only through the state of health (SF-36) but also by assessing the value of health (EQ-5D) reported by the patient. The fact that it included the utility measure will enable future cost-effectiveness studies. Finally, analysis of summary measures of SF-36 as well as each of the domains provides a clearer picture of the dimensions affected by the disease.
We consider our results useful to better understand the impact of disease and establish a basis for future prospective studies with larger samples to assess the efficacy of therapeutic interventions. The focus on seriously ill patients in need of costly therapy may also be useful for cost-utility analysis studies of biotech drugs.
ConclusionRA has considerable impact on quality of life and functional capacity being the impairment of HRQoL enormous when compared to general population. We verified lower HRQoL and functional capacity with higher disease activity levels and we saw that reaching remission seems to be a desirable state also from patient perspective. Since the clinical and laboratory data are weakly correlated with HRQoL and functional capacity, we believe that access to PROs is very important in order to complete the disease assessment.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
FundingNot applicable.
Authors contributionsAll authors make substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data. All critically revise it for important critical content and give final approval of the version of the article accepted for publication.
Conflicts of interestNone of the authors has any conflicts of interest to declare.
The authors would like to thank all of the people who participated in the collection of the data.