To identify synovitis and tenosynovitis active by using the Ultrasound 7 (US 7) scoring system in patients with rheumatoid arthritis (RA) in clinical remission induced by synthetic disease-modifying antirheumatic drugs (DMARDs).
MethodsThis is a multicentric, cross-sectional, observational study including 94 RA patients >18 years old who were in remission as defined by the 28-joints disease activity score (DAS28) <2.6 induced by synthetic DMARD during at least 6 months. Patients with a previous or current history of biologic DMARD treatment were not included in the study. Demographic and clinical data were collected by the local rheumatologist; the US evaluation was performed by a calibrated rheumatologist, who intended to detect grayscale synovitis and power Doppler (PD) using the 7-joint scale. Intra and inter-reader exercises of images between 2 ultrasonographers were realized.
ResultsPatients’ mean age was 49.1±13.7 years; 83% were women. The mean disease duration was 8±7 years and remission lasted for 27.5±31.8 months. The mean DAS28 score was 1.9±0.66. Grayscale synovitis was present in 94% of cases; it was mild in 87.5% and moderate in 12.5%. Only 12.8% of the patients had PD. The metatarsophalangeal, metacarpophalangeal, and carpal joints of the dominant hand were the joints more frequently affected by synovitis. Tenosynovitis by grayscale was observed in 9 patients (9.6%). The intra and inter-reading kappa value were 0.77, p<0.003 (CI 95%, 0.34–0.81) and 0.81, p<0.0001 (CI 95%, 0.27–0.83) respectively.
ConclusionsLow percentage of synovitis and tenosynovitis active were founded according to PD US by 7 score in RA patients under synthetic DMARDs during long remission. This score has benefit because evaluate tenosynovitis, another element of subclinical disease activity.
Identificar la sinovitis y tenosinovitis activa mediante el uso de ultrasonido en un índice de 7 articulaciones (US7) en pacientes con artritis reumatoide (AR) en remisión clínica inducida por fármacos antirreumáticos modificadores de la enfermedad sintética (DMARD).
MétodosSe trata de un estudio observacional multicéntrico, transversal, que incluyó a 94 pacientes con AR mayores de 18 años que estaban en «remisión de acuerdo a DAS-28<2,6 inducida por DMARD sintético durante al menos 6 meses». Los pacientes con antecedentes previos o actuales de tratamiento biológico no fueron incluidos en el estudio. Los datos demográficos y clínicos fueron recogidos por el reumatólogo tratante; la evaluación de US fue realizada por un reumatólogo experimentado, para detectar sinovitis en escala de grises y power Doppler (PD) utilizando la escala de 7 articulaciones. Se realizaron ejercicios intra e inter-lector de imágenes entre 2 ultrasonografistas.
ResultadosLa edad media de los pacientes fue de 49,1±13,7 años; el 83% eran mujeres. La duración media de la enfermedad fue de 8±7 años y la remisión duró 27,5±31,8 meses. La media de DAS-28 fue de 1,9±0,66. La sinovitis en escala de grises estuvo presente en el 94% de los casos; fue leve en el 87,5% y moderada en el 12,5%. Solo el 12,8% de los pacientes tenían PD. Las articulaciones metatarsofalángicas, metacarpofalángicas y carpales de la mano dominante fueron las articulaciones más frecuentemente afectadas por la sinovitis. La tenosinovitis en escala de grises se observó en 9 pacientes (9,6%). El valor de kappa intra e inter-lector fue 0,77, p<0,003 (CI 95%: 0,34-0,81) y 0,81, p<0,0001 (CI 95%: 0,27-0,83), respectivamente.
ConclusionesSe observó un bajo porcentaje de sinovitis y tenosinovitis activa de acuerdo a PD por US7 en pacientes con AR tratados con DMARD sintéticos. Esta escala tiene ventaja porque evalúa tenosinovitis, otro elemento de la actividad inflamatoria subclínica.
The aim of treatment in rheumatoid arthritis (RA) is to suppress the disease activity and induce remission as quickly and for as long as possible, to prevent joint damage and loss of function.1 The assessment of disease activity relies on composite indexes that include clinical findings. However, several reports have shown that a high percentage of patients categorized on remission by clinical methods may develop radiographic progression (irreversible structural damage) throughout follow up because of subclinical synovitis that is not detectable with simple X-ray.2 Magnetic Resonance Imaging (MRI) is perhaps the best tool to assess subclinical synovitis and early loss of the articular cartilage and erosions development, but its high cost and sometimes the need for enhancing medium to evaluate synovitis among others limit its accessibility.3
In this context, US is a highly useful technique low cost, multiplanar, and non-invasive technique that does no produce any radiation, does not require contrast medium, and can be repeated as many times as necessary. Grayscale (GS) and power Doppler (PD) may show subclinical synovitis in patients with RA in remission achieved by the use of synthetic or biologic disease-modifying antirheumatic drugs (DMARDs).4 GS synovitis suggest the development or worsening of bone erosions5 and the presence of PD signal increases at least >1 the probability of flare up in clinical remission.6
US is superior to clinical examination in detecting joint inflammation in undifferentiated arthritis, and in established RA.7 Joint exploration by US has been used to reclassify patients who clinically have fewer inflamed joints by showing synovitis in those which are apparently normal.7 Also, it has validity comparable to MRI with contrast medium and arthroscopy to detect synovitis, and is superior to simple X-ray to detect erosions.8 There are different scales to evaluate joint inflammation. Today, it has been shown that the US 7 scale is equally sensitive as the 78-joint scale to evaluate changes in grayscale and PD.9
The importance of proper monitoring of the disease is that timely modifications of treatment can be made, which may really change the course of the disease, by preventing structural damage and permanent disability.10 It is therefore that the objective of this investigation was to evaluate the presence of inflammatory activity, by means of the US 7 scale, in patients with RA treated with synthetic DMARDs who were clinically in remission according to the DAS28 score.
Material and methodsWe conducted a prospective, cross-sectional, observational phase IV study between March, 2013, and June, 2014 in three centers in México City. Ninety-four patients over 18 years of age with RA11 receiving treatment with synthetic DMARDs (methotrexate, hydroxychloroquine, chloroquine, leflunomide, sulfasalazine) (either as monotherapy or combined therapy), without corticosteroid and treatment without modifications in clinical remission by DAS28 score (<2.6) at least for 6 months before they were included in the study. Only subjects whose treatment remained stable for 6 months before entering the study were included. We excluded patients who had received glucocorticoid infiltration during 6 months prior to inclusion in the study. All patients agreed to participate in the study and signed an informed consent form. Patients currently or past treated with anti-TNFs such as infliximab, adalimumab, certolizumab pegol and golimumab, or with circulating receptor fusion proteins like etanercept, were not included in the study. The National Bioethics Committee for clinical research approved the protocol.
The sample included 100 patients of whom 94 patients completed the two visits planned for the study. At visit 1 (days −10 to 0), informed consent was presented to patients and signed by those who accepted their participation; then, inclusion and exclusion criteria were checked; and data on clinical evaluation and therapeutic history was collected. One experimented rheumatologist conducted clinical evaluation. At visit 2 (days 1–7), patients were sent to only one of the centers to underwent the US 7 scale (grayscale and PD) joint evaluation9 performed by a rheumatologist, who was blinded to clinical findings. The average time for each evaluation was 15min.
US studies were carried out with a Esaote MyLab 25® US equipment (Biomedica, Genoa, Italy) equipped with a 10–18MHz linear transducer; PD was adjusted according to the following parameters: frequency, 8.0, PRF, 0.500, wall filter, 3, gain between 50 and 70. PD gain was set to a level below its appearance from cortical bone.12 The US 7 scale included the wrist, 2nd, and 3rd metacarpophalangeal (MCP) joints, 2nd and 3rd proximal interphalangeal (PIP) joints, and 2nd and 5th metatarsophalangeal (MTP) joints on the clinically dominant side,9 according to EULAR US guidelines.13
OMERACT definitions were used to describe synovitis, tenosynovitis, and erosions. Synovitis is seen as effusion defined as abnormal, hypoechoic or anechoic, compressible, displaceable intraarticular material, without PD signal, and synovial hypertrophy as abnormal hypoechoic, poorly compressible intra-articular tissue that may exhibit PD signal.14 Tenosynovitis: abnormal, hypoechoic or anechoic (relative to tendon fibers) tendon sheath widening which can be related both to the presence of abnormal tenosynovial fluid and/or hypertrophy15 thickened, hypoechoic or anechoic tissue with or without fluid in the tendon sheath. Erosion: an intraarticular discontinuity of the bone surface visible in 2 perpendicular planes.15 Also, the presence of paratenonitis, was evaluated, and defined as a hypoechoic halo around the tendon with or without the presence of PD.9 Tenosynovitis or paratenonitis, and erosions were reported as present or absent (1 or 0).
Grayscale synovitis was evaluated semi-quantitatively: 0 absent, 1 mild, 2 moderate, and 3 severe. Grade 1 describes a hypoechoic or anechoic area which does not extend beyond the joint capsule interline; in grade 2, the joint capsule is raised parallel to the articular area; and grade 3 shows notable widening of the joint capsule.9 The presence of PD was graduated as follows: grade 0 without intra-articular signal; grade 1, up to 3 signals in the joint area; grade 2, more than 3 signals in less than 50% of the intraarticular area; grade 3, more than 50% of the intraarticular area.16 US evaluation was performed only by one ultrasonographer. Intra and inter-reading exercise were done by 2 ultrasonographers.
Statistical analysisMedian, standard deviation (SD), and percentages were used to describe patients’ clinical characteristics and US findings. Intra and Inter-reader reliability were calculated by the Cohen's kappa coefficient. Kappa values were assigned the following designations: below 0.20 as poor, 0.21–0.40 as fair, 0.41–0.60 as moderate, 0.61–0.80 as good, and 0.81–1 as excellent. The analyses were performed using the SPSS ver. 21.0 statistical computer software (SPSS, Inc., Chicago, IL, USA).
ResultsClinical and demographic data are shown in Table 1. Ninety-four patients were included; six were excluded for failing to meet the criterion of DAS28 below 2.6. As expected, most patients were women. Disease duration was 27.5+31.8 months and DAS28 was 1.9+0.66.
Clinical and demographic data for patients.
| n=94 | Value |
|---|---|
| Mean age, years (±SD) | 49.1±13.7 |
| Sex female/male, n (%) | 83 (88)/11 (12) |
| AR seropositive, n (%) | 76 (80.8%) |
| AR erosive, n (%) | 45 (47.8) |
| Mean disease duration, years (±SD) | 8±7 |
| Mean duration of disease remission by DAS28 in months (±SD) | 27.5±31.8 |
| Mean DAS28 score (±SD) | 1.9±0.66 |
| Mean CPR, mg/dL (±SD) | 1.02±1.76 |
| Mean ESR, mm (±SD) | 18.1±9.8 |
| Mean Pain Visual Analogue Scale (±SD) | 9.7+13.9 |
| Treatment | No. (%) of patients |
|---|---|
| Methotrexate | 87 (92.5) |
| Sulfasalazine | 27 (28.7) |
| Leflunomide | 14 (14.9) |
| Hydroxychloroquine | 14 (14.9) |
| Patients with methotrexate plus another DMARD | 42 (44.6) |
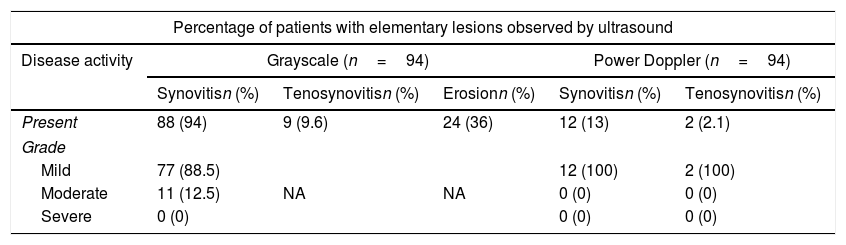
Table 2 shows the prevalence of elementary lesions in all joints evaluated. In grayscale we observed a high prevalence of synovitis in 94% (88 patients), although in almost 90% changes were mild and moderate in only 12.5%; there were no severe lesions found in the study. Only 12 (13%) of the 94 patients had PD signal and in all it was mild. Tenosynovitis was observed in 9 (9.6%) in grayscale and with PD only 2 (2.1%). Paratenonitis was not observed in any case. Erosions were found in 34 (36%) patients.
Prevalence of elementary lesions in all joints evaluated by US 7 score.
| Percentage of patients with elementary lesions observed by ultrasound | |||||
|---|---|---|---|---|---|
| Disease activity | Grayscale (n=94) | Power Doppler (n=94) | |||
| Synovitisn (%) | Tenosynovitisn (%) | Erosionn (%) | Synovitisn (%) | Tenosynovitisn (%) | |
| Present | 88 (94) | 9 (9.6) | 24 (36) | 12 (13) | 2 (2.1) |
| Grade | |||||
| Mild | 77 (88.5) | NA | NA | 12 (100) | 2 (100) |
| Moderate | 11 (12.5) | 0 (0) | 0 (0) | ||
| Severe | 0 (0) | 0 (0) | 0 (0) | ||
NA=not applicable.
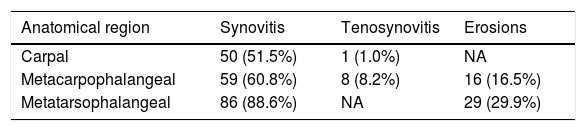
Table 3 shows the frequency of elementary lesions by US grayscale in most affected regions. The prevalence was high in metatarsal joints, followed by MCP and carpal joints (88.6%, 60.8%, and 51.5% respectively). On the other hand, tenosynovitis was more common in hand flexors in MCP joints than in the carpus (8.2% vs. 1%). Erosions were more common in metatarsal heads than in metacarpal joints (29.9% vs. 16.5%).
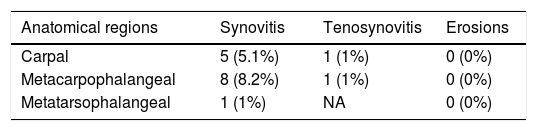
Table 4 presents the distribution of elementary lesions with PD signal. The highest prevalence was in MCP joints, then in wrists, and the lowest in MTP joints (8.2%, 5.1%, and 1.0% respectively). In only 1% of patients, PD was observed in tenosynovitis of finger flexors at the metacarpal level and 1% in carpus. None of the erosions showed PD signal.
The intra and inter-reading kappa value were respectively 0.77, p<0.003 (CI 95%, 0.34–0.81) and 0.81, p<0.0001 (CI 95%, 0.27–0.83).
DiscussionIn this study the prevalence of subclinical synovitis, in grayscale was high and low by PD, similar as shown by a systematic review of the literature, where percentages ranging from 73% to 95% and from 8.5% to 62%.7 Thus, in turn, corroborates the discrepancy between the criterion of clinical remission used and US findings. Some studies have reported that, regardless of the specific criteria of remission used, when evaluated by US, synovitis active is detected in 60–80% of patients.17 The use of US to monitor patients in clinical remission can help to predict those likely to suffer flare-up or structural damage.17,18 Because some studies have demonstrated high percentage of synovial hypertrophy on US in patients considered to be in remission, this state should be defined only in the absence of synovitis by this technique.19
The sites of greatest prevalence of synovitis are the carpal and the MCP joints.14 However, in our study, we observed more synovitis in the metatarsal joints, which correlates with reports indicating that the feet are affected in around 90% of patients during the course of the disease, 91% in women and 85% in men.20 Additionally, this is the region that conditions the highest rates of disability,21 which underscores the importance of including such joints in scanning for synovitis. In relation to PD, multiple studies have reported that a signal was present in around 50% of patients in clinical remission in MCP and carpal joints of the dominant hand. Our findings are some similar, since we detected a lower percentage of activity, perhaps of the longer remission time in our group.16,22
The prevalence of tenosynovitis in long term RA ranges from 5% to 55%.23 We found tenosynovitis in less than 10% of patients, predominantly in the third flexor of the dominant hand. This finding may be related to biomechanical factors due to the high range of movement of that tendon compared with others.23 Tenosynovitis may be very important for monitoring RA, because could conditioned tendon damage and consequently disability.24 Lillegraven et al. have demonstrated that tenosynovitis assessed by US predicts the development of erosive joint damage in a cohort of early RA patients.25 Two studies have shown that tenosynovitis is not present in true remission, suggesting the high value of its assessment by US.26,27 Furthermore, damaged tendon as assessed by US correlates very well with bone structural damage assessed by X-ray.27 Today, however, there is neither established scale nor which tendons should be included in US evaluation for monitoring.24
Bone erosions including in this score pertain to the realm of structural damage. In this study, around 30% had erosions in the MTP joints and 16% in the MCP joints. For patients with established diagnosis of RA, perhaps bone erosions have not great value, and may be more useful in patients with suspected early-stage RA. However, some data suggest that bone erosions may sensitive to change.28
To the best of our knowledge, this is the first study that applied US 7 scale to detect any residual inflammatory activity in clinical remission, in joint and tendon level with vantage of consuming less time than more extended count joint and had been used to evaluate response to treatment.29
LimitationsWe considered another method, such as magnetic resonance imaging (MRI) was needed to compare the findings, but MRI is not a feasible technique to evaluate multiple joints at the same time. Additionally, control group was absence.
ConclusionsThe prevalence of synovitis and tenosynovitis assessed by US 7 score is low in patients with rheumatoid arthritis in clinical remission by DAS28 during long time of disease remission. The US 7 score is useful because add additional value to detect tenosynovitis that appear other instruments to measure real remission state.
FundingThe present work was supported by unrestricted AbbVie grants.
Conflicts of interestThe authors declare have not conflict of interest.
The authors thank Angel Mario Coll Muñoz, Senior Operations Manager Clinics, Juan Carlos Pozos Immunology Division Medical Manager and José Luis Cañadas, Medical Director and Regulatory Affairs, all in AbbVie for their valuable help.