To describe the results obtained in clinical practice with the use of biological therapy (BT) in patients diagnosed with Takayasu arteritis (TA) and giant cell arteritis (GCA).
MethodsRetrospective single center study of TA/GCA patients who received BT (infliximab [IFX], etanercept [ETN] and tocilizumab [TCZ]). In TA, active disease was defined according to a previous National Institutes of Health study. In GCA, active disease was defined with a modified criteria and clinical manifestations secondary to temporal artery involvement or polymyalgia rheumatica symptoms. Clinical data and outcomes are reported using descriptive statistics.
ResultsFive patients with TA and 5 with GCA were included. The main reason for starting BT was lack of response to prior therapy and/or ≥2 relapses during GC tapering. Five patients started IFX, four TCZ and 1 ETN. Remission was observed before 6 months in all cases. Only one patient had a relapse during long-term follow-up and the overall GC daily dose was reduced by 70%. Two AEs were considered attributable to IFX and one to TCZ.
ConclusionA favorable and sustained response to BT was observed in our patients with TA and GCA. Thus, BT might be considered as an alternative in patients with large vessel arteritis refractory to conventional treatment or with GC related comorbidities.
Describir los resultados obtenidos en la práctica clínica diaria con el uso de la terapia biológica (TB) en pacientes con diagnóstico de arteritis de Takayasu (AT) y arteritis de células gigantes (ACG).
MétodosEstudio retrospectivo monocéntrico de pacientes con AT/ACG que recibieron TB (infliximab, etanercept y tocilizumab). En AT, la enfermedad activa se definió de acuerdo a un estudio previo del National Institutes of Health. En ACG, la enfermedad activa se definió con dichos criterios modificados y manifestaciones clínicas secundarias a afectación de la arteria temporal o síntomas de polimialgia reumática. Los datos y los desenlaces clínicos se muestran mediante estadística descriptiva.
ResultadosSe incluyeron 5 pacientes con AT y 5 con ACG. La razón principal para el inicio de la TB fue la falta de respuesta al tratamiento previo y/o ≥2 recaídas durante la terapia con corticoides. Cinco pacientes comenzaron infliximab, 4 tocilizumab y uno etanercept. La remisión se observó antes de los 6 meses en todos los casos. Solo un paciente tuvo una recaída durante el seguimiento a largo plazo. La dosis diaria de corticoides se redujo globalmente en un 70%. Dos acontecimientos adversos se consideraron atribuibles a infliximab y uno a tocilizumab.
ConclusiónSe observó una respuesta favorable y sostenida a la TB en nuestros pacientes con AT y ACG. Por lo tanto, la TB puede ser considerada una alternativa en pacientes refractarios al tratamiento convencional o con comorbilidades asociadas a los corticoides.
Giant cell arteritis (GCA) and Takayasu arteritis (TA) are chronic inflammatory diseases that mainly affect large elastic arteries. Although GCA is a disease of older people and TA is a disease of younger people, their shared predilection for causing vasculitis of large arteries and their nearly identical histopathologic changes suggest a possible link in pathophysiology and eventually in treatment response.
Response to glucocorticoids (GCs) is generally favorable in GCA and TA but relapses may occur while gradually tapering GCs. Physicians tend to start immunosuppressive (IS) agents together with GCs but there is no evidence showing which is the IS agent of choice.
Serum tumor necrosis factor-α (TNF-α) levels and IL-6 are increased in TA and GCA patients as well.1,2 Therefore, blocking TNF-α or IL-6 with biologic therapy (BT) has emerged as an attractive therapeutic alternative that would move the field forward and decrease the cumulative GC toxicity.
Here, we assessed the clinical, serological, and imaging outcomes of 10 patients with refractory TA and GCA who were administered BT.
Patients and methodsInclusion of patientsPatients with GCA or TA (ACR, 1990) under BT were retrospectively identified from our medical records. All cases were refractory to conventional therapies including GC, or had developed significant adverse effects from these agents. Our hospital ethics committee approved the off-label use of infliximab (IFX), etanercept (ETN) or tocilizumab (TCZ) for these patients, and informed consent was obtained from the patients or their relatives after thorough discussion on the risk/benefit ratio of BT.
Clinical dataThe following patient information was recorded: sex, age, disease duration, prior and concomitant immunomodulatory treatment, reason for starting BT, acute phase reactants [erythrocyte sedimentation rate (mm/h) and C-reactive protein (mg/L)], type and dose of BT used, clinical response at 3, 6, 12, 24 months and last follow-up, follow-up time, imaging when available, revascularization procedures and relevant AEs secondary to BT.
Disease activity assessmentIn TA, disease activity was assessed according to previously published criteria.3 In GCA, active disease was defined as worsening or emergence of ≥2 of the following: (1) fever or other characteristic of systemic involvement (in the absence of another cause); (2) increased ESR and/or C-reactive protein (CRP); (3) clinical manifestation secondary to large vessel involvement and (4) symptoms consistent with polymyalgia rheumatica (PMR). In case of no active disease, the patient was considered in remission. Sustained remission was defined in those who met these conditions for at least 6 months while receiving less than 10mg/day of prednisone.
Imaging proceduresIn TA, magnetic resonance angiography (MRA) was performed in all patients either initially for confirming the initial diagnosis or at a later time for planning surgical procedures. MRA was also used even in the absence of new symptoms or findings, to monitor the activity and distribution of lesions. Each study included the entire aorta and all primary branch vessels. Active or relapsing disease was considered to be present in the setting of new vascular lesions, as visualized by MRA, at sites that were previously unaffected.
Statistical analysisThe data are reported using descriptive statistics. Numerical data are expressed as mean and standard deviation (SD) for continuous variables and percentages for qualitative variables. The ESR and CRP monitoring represent the mean (±SD) of the sum of the individual values of different visits after starting BT.
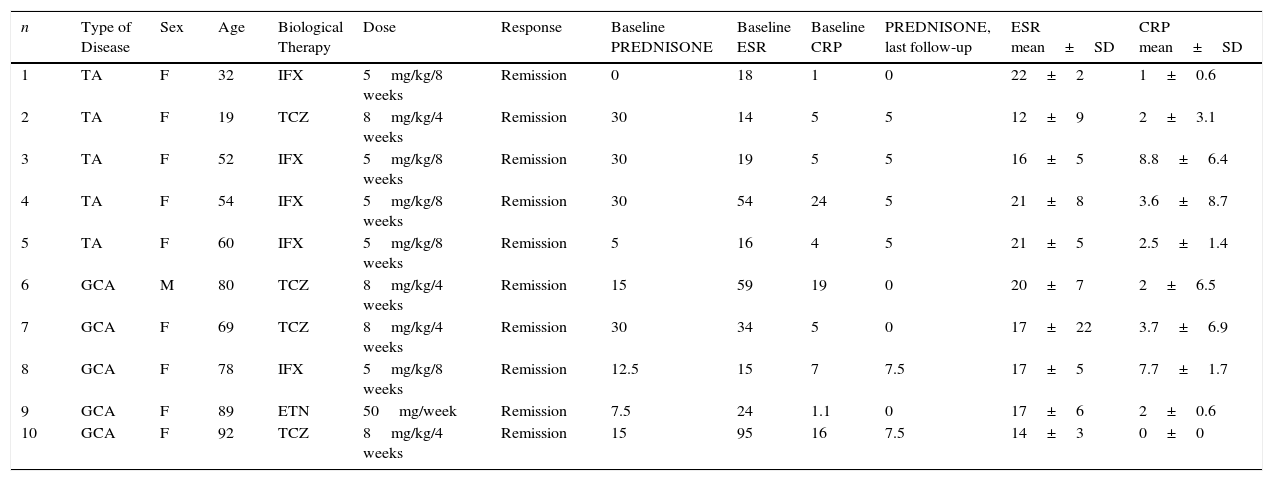
ResultsTen patients, 5 with TA and 5 with GCA under BT were included. Demographics and baseline ESR, CRP and GC dose are described in Table 1.
Baseline clinical characteristics [prednisone dose, acute phase reactants and response obtained after biological therapy (BT)] in Takayasu arteritis and giant cell arteritis patients.
| n | Type of Disease | Sex | Age | Biological Therapy | Dose | Response | Baseline PREDNISONE | Baseline ESR | Baseline CRP | PREDNISONE, last follow-up | ESR mean±SD | CRP mean±SD |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | TA | F | 32 | IFX | 5mg/kg/8 weeks | Remission | 0 | 18 | 1 | 0 | 22±2 | 1±0.6 |
| 2 | TA | F | 19 | TCZ | 8mg/kg/4 weeks | Remission | 30 | 14 | 5 | 5 | 12±9 | 2±3.1 |
| 3 | TA | F | 52 | IFX | 5mg/kg/8 weeks | Remission | 30 | 19 | 5 | 5 | 16±5 | 8.8±6.4 |
| 4 | TA | F | 54 | IFX | 5mg/kg/8 weeks | Remission | 30 | 54 | 24 | 5 | 21±8 | 3.6±8.7 |
| 5 | TA | F | 60 | IFX | 5mg/kg/8 weeks | Remission | 5 | 16 | 4 | 5 | 21±5 | 2.5±1.4 |
| 6 | GCA | M | 80 | TCZ | 8mg/kg/4 weeks | Remission | 15 | 59 | 19 | 0 | 20±7 | 2±6.5 |
| 7 | GCA | F | 69 | TCZ | 8mg/kg/4 weeks | Remission | 30 | 34 | 5 | 0 | 17±22 | 3.7±6.9 |
| 8 | GCA | F | 78 | IFX | 5mg/kg/8 weeks | Remission | 12.5 | 15 | 7 | 7.5 | 17±5 | 7.7±1.7 |
| 9 | GCA | F | 89 | ETN | 50mg/week | Remission | 7.5 | 24 | 1.1 | 0 | 17±6 | 2±0.6 |
| 10 | GCA | F | 92 | TCZ | 8mg/kg/4 weeks | Remission | 15 | 95 | 16 | 7.5 | 14±3 | 0±0 |
Abbreviations: n, number of patient; TA, Takayasu arteritis; GCA, giant cell arteritis; F, female; M, male; IFX, infliximab TCZ, tocilizumab; ETN, etanercept; ESR, erythrocyte sedimentation rate (mm/1hour); CRP, C-reactive protein (mg/L). Acute phase reactants are expressed as mean of overall monitoring±standard deviation (SD).
In TA, the most common lesion sites were supra-aortic arteries (in all cases). Abdominal aorta and its branches were affected in three patients but only 2 patients showed thoracic aorta involvement. Stenotic lesions were the most frequent vascular finding and only one patient presented with aneurismal lesions. Three patients required revascularization procedures prior to BT.
The main reason for starting BT in TA was the appearance of relapses upon GC tapering in all patients and lack of response to prior therapy; methotrexate (n=5), cyclophosphamide (n=3) and mycophenolate mofetil (n=3) were the most common IS agents added at some point to GC. One patient had received adalimumab prior to this study but remission was not achieved and BT was switched to TCZ. Four patients received IFX and 1 patient TCZ. Only one patient received IFX as monotherapy (patient 5) and the remaining four received methotrexate (n=3) 20.8±7.2mg/week (2 subcutaneous and 2 oral administration) or mycophenolate mofetil (n=1) 1000mg/day. Remission was observed before 6 months in all cases. Sustained remission was achieved in all cases during follow-up (mean follow-up 59.6±27.2 months) and GC daily dose was reduced by 79% (Table 1). In cases of sustained remission, careful down-titration was carried out without evidence of relapses. None of the patients developed a new arterial lesion while receiving BT. Furthermore, revascularization procedures were not required during follow-up. All patients tolerated BT without infusion reactions. Other AEs attributable to BT were recurrent infections (one patient with IFX) and mild neutropenia (one patient with TCZ). At last follow-up, 3 patients continued on BT, 1 patient was lost to follow up (moved to another country) and 1 patient discontinued IFX due to recurrent infections.
In GCA the main reason for starting BT was lack of response to prior therapy (MTX in 5 patients, AZA in 2 patients, MMF in 2 patients) and/or ≥2 relapses during GC tapering. Three patients started TCZ, one patient IFX and one patient ETN. Three patients received BT as monotherapy (patients 6–8) and the remaining two received MTX 15mg/week and leflunomide 20mg/day. The remission was observed before 3 (n=4) and 6 months (n=1). Only one patient had a relapse during follow-up (mean follow-up 37.8±33.8 months). This was the patient under ETN therapy who suffered a recurrence at 24 months of BT and six months after GC withdrawal. She presented with PMR, high CRP and ESR without any ischemic complication and resolved with GC reintroduction. Overall, the daily dose of GC in GCA was reduced by 60%. No changes in concomitant IS agents was required. One AE was attributable to IFX (anaphylaxis) and 1 AE attributable to TCZ (mild neutropenia). At last follow-up, 2 patients continued with BT, 1 patient discontinued ETN for sustained remission, 1 patient discontinued BT secondary to AE attributable to IFX and 1 patient died for reasons not attributable to BT.
DiscussionThere are no reported randomized controlled studies on the use of BT in TA. Hoffman et al. first published an open-label trial of anti-TNF therapy in TA (n=15) in 2004.4 Overall, the addition of anti-TNF therapy resulted in improvement in 14 patients and sustained remission in 10 cases. Anti-TNF treatment also allowed for the reduction or elimination of GC therapy in almost all of the patients. In 2008, Molloy et al. published their experience in 25 patients.5 Following anti-TNF therapy, remission was achieved and prednisone was discontinued in 15 patients (60%) and successfully tapered below 10mg/day in additional 7 patients (28%). Schmidt et al. reported in 2012 a single center experience with the use of TNF inhibitors in 20 patients with TA.6 Disease remission was achieved in 18 (90%) but 33% of patients experienced disease relapse while receiving BT. In France, Mekinian et al. (n=15) confirmed also previous reports on the efficacy of TNF inhibitors in refractory TA.7
TCZ could be another option for refractory TA or patients failing to TNF-inhibitors. Abisror et al. retrospectively studied 5 patients from 3 French university hospitals and 39 cases from the literature.8 Clinical and biological activities significantly decreased within 3 months similarly to steroid dose and steroid-dependence rate. Remission was achieved in 33 cases (75%). Moreover, 9 out of 14 patients (65%) who had previously received TNF inhibitors achieved remission. Recently, Cañas et al. reported 8 refractory TA patients treated with TCZ who achieved remission in all cases and 85% GC reduction.9 Last, Loricera et al. published a study on 16 patients with inflammatory aortitis refractory to corticosteroids or to other biologic immunosuppressive drugs (14 cases were large vessels vasculitis), TCZ appeared to be effective and relatively safe.10 Most recently, the same study group evaluated the efficacy of TCZ in patients with refractory TA (n=8). Clinical and laboratory parameters improved in most cases.11 Both TCZ and TNF inhibitors seem to be effective in refractory TA but comparative studies to determine the better strategy for these biologics are lacking.
Despite the reported experience in TA with BT and the lack of clinical trials, GCA first published randomized trial was terminated prematurely due to lack of benefit with IFX.12 However, the protocol of steroid tapering used in this study could have negatively influenced the final results. Two subsequent controlled trials failed to demonstrate a steroid sparing effect of etanercept and adalimumab.13,14 However, the response in 2 of our patients suggests a potential role for these therapies in selected GC refractory cases. Regarding TCZ, our findings are in agreement with a recently published systematic review and meta-analysis of GCA patients treated with TCZ.15 All the 19 patients treated with TCZ plus prednisone achieved disease remission and a steroid sparing effect with complete discontinuation of GC in nine patients. Notably, 3 of our patients with GCA were successfully treated with TCZ monotherapy. Finally, TCZ therapy showed rapid and maintained improvement in patients with refractory GCA and/or with unacceptable AEs related to GCs in a multicenter open-label study of 22 patients.16
In spite of the limitations of a retrospective study and the small number of patients included, our data provide positive evidence on the potential benefit of TNF inhibitors and TCZ in patients with large vessels vasculitis refractory to conventional treatment. TNF inhibitors or TCZ have a role as steroid-sparing agents in refractory LVV patients, with acceptable safety profile. TCZ might be the first choice biologic therapy in refractory GCA patients. In the absence of randomized controlled trials addressing this specific clinical situation, this type of evidence can help in our daily clinical practice.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
FundingThis research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
Conflict of interestRGV has received funding for research or teaching from MSD, Roche, BMS, Abbvie, and UCB; has participated on advisory boards for Actelion, BMS, UCB, Pfizer, Roche, Hospira, Janssen and Sandoz, has delivered presentations sponsored by Roche, BMS, Pfizer, UCB, Sandoz.