Infections are a major cause of morbidity and mortality in patients with systemic autoimmune diseases. The aim of the present study is to describe the frequency of infections in a historical cohort of the SAD polyclinic of the Maciel Hospital, according to the type of disease and treatment received.
Material and methodsAn analytical, retrospective and observational study was conducted in 339 patients with SAD attended at the outpatient clinic in the period from January 1, 2012 to February 28, 2019. Infectious complications were analysed according to treatment and disease.
Results339 cases, median age 56, mostly female. Most cases presented SLE (30.1%) and RA (23.6%), followed by antiphospholipid syndrome (20.4%) and Sjögren's syndrome (12.1%). Hydroxychloroquine (66%), followed by corticosteroids (55.5%) were the most frequently used treatments. Thirteen point three percent received biological therapies: 46.9% of the cases presented some infectious complication, 95% were non-opportunistic. Respiratory infections were the most frequent (48.6%) followed by urinary infections (31.7%) and skin and soft tissue infections (17.6%). On comparing the infected and non-infected groups, significant differences were found in the following variables: methotrexate, mycophenolate, corticoids, biological therapies, combination of drugs, active disease, RA and cases with overlap. The use of hydroxychloroquine and sulfasalazine was associated with a lower risk of infection in patients with RA.
ConclusionsInfections are a frequent complication in patients with RA, due to the immune disturbances of the disease itself and prescribed treatments, mainly corticoids and biologicals. The importance of screening and infection prophylaxis before starting treatment is stressed.
Las infecciones son una importante causa de morbimortalidad en los pacientes con enfermedades autoinmunes sistémicas. El objetivo del presente estudio es describir la frecuencia de infecciones en una cohorte histórica de la policlínica de EAS del Hospital Maciel, según tipo de enfermedad y tratamiento recibido.
Material y métodosSe realizó un estudio analítico, retrospectivo y observacional de 339 pacientes con EAS asistidos en la consulta ambulatoria en el período comprendido entre el 1 de enero de 2012 y el 28 de febrero del 2019. Se analizaron las complicaciones infecciosas, según tratamiento y enfermedad.
ResultadosSe encontraron 339 casos, mediana de edad de 56, mayoría sexo femenino. La mayoría de los casos presentaron LES (30,1%) y AR (23,6%), seguidos de síndrome antifosfolipídico (20,4%) y síndrome de Sjögren (12,1%). La hidroxicloroquina (66%), seguida de los corticoides (55,5%) fueron los tratamientos más frecuentemente utilizados. El 13,3% recibieron terapias biológicas. 46,9% de los casos presentaron alguna complicación infecciosa, 95% fueron no oportunistas. Las infecciones respiratorias fueron las más frecuentes (48,6%), seguidas de las urinarias (31,7%) y de piel y partes blandas (17,6%). Al comparar los grupos de infectados y no infectados se hallaron diferencias significativas en las siguientes variables: metotrexate, micofenolato, corticoides, terapias biológicas, combinación de fármacos, enfermedad activa, AR y casos con solapamiento. El uso de hidroxicloroquina y sulfasalazina se asoció con menor riesgo de infecciones en pacientes con AR.
ConclusionesLas infecciones son una complicación frecuente en los pacientes con EAS, por las alteraciones inmunitarias de la propia enfermedad y por los tratamientos indicados, fundamentalmente corticoides y biológicos. Se destaca la importancia del cribado y profilaxis de infecciones antes del inicio del tratamiento.
Infections are a major cause of morbidity and mortality in systemic autoimmune diseases (SAD) due to immune changes from the disease itself and from immunosuppressant treatments.1,2
Most studies on infections in SAD have been carried out for systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA). There is a high prevalence of infections in SLE, with this being one of the major causes of death during the first five years of the disease. In RA there is twice as high a risk as in the general population.1–7
The infections differ in severity. Mild forms are most common with the most affected sites being respiratory, urinary, skin and soft tissues.8–10 Aetiological agents are varied and may be viral, bacterial and other, either opportunistic or non-opportunistic.2,4,6,7,10–17
Within the opportunistic infections, latent tuberculosis may reactivate in patients receiving corticoid and/or immunosuppressant treatment.18–21
Risk factors associated with the highest infection risk in patients with SLE are: disease activity, presence of lupus nephritis, disease duration, leucopenia, anti-DNA antibodies, dose and time of treatment with corticoids and use of immunosuppresants.1–3,11–14,16 Treatment with hydroxychloroquine (HCQ) has been shown in several studies to be a protective factor against infections.16,22–24
In patients with RA, the risk factors are advanced age, extra-articular disease, disease activity, chronic obstructive pulmonary disease, interstitial pulmonary disease, chronic renal disease, lymphopenia, the use of corticosteroids and disease modifying anti-rheumatic drugs (DMARDS).6,7
Biological therapies have changed the evolution of SAD, although they are associated with a three times as high a risk of infections.6,25–28
SAD present a diagnostic and therapeutic challenge to the doctor, essentially for a patient with fever where it should be clarified whether this corresponds to infection or disease activity. Complete clinical and para-clinical assessment is required to reach a diagnosis and adopt decisions, based on each individual case.24,29
There are no infection review studies in patients with SAD in Uruguay, which motivated the interest of the authors in this subject.
The aim of this study was to describe the frequency of infections in a historical cohort of patients with SAD, according to disease, activity of disease and treatment.
Material and methodsAn analytical, retrospective and observational study of a historical cohort. All patients with a confirmed diagnosis of SAD (33) who attended the SAD outpatient practice of the Hospital Maciel, during the period between 1st January 2012 and 28th February 2019 were included.
Patients who attended the outpatient unit less than twice were excluded.
Written medical files obtained from the medical archive and the electronic registers of the hospital were obtained.
The following variables were recorded: age, sex, origin, high blood pressure (HBP), diabetes mellitus (DM), dyslipidaemia, obesity, tobacco habit, cardiovascular disease (CVD), chronic renal disease (CRD) chronic obstructive pulmonary disease (COPD); diagnostic classification of SAD (in accordance with current classifying criteria); treatment in the three previous months: hydroxychloroquine, synthetic or biologic immunosuppressants; average corticoid dose (prednisone or equivalent) during infection and on diagnosis; infectious complications (organ/compromised system), causal microorganism, opportunistic infections, hospitalisation and intrahospital infections.
Opportunistic infections are considered serious when they require antibiotic therapy by intravenous route or hospitalization, and those which are life-threatening.
Opportunistic infections were considered to be those produced by specific pathogens or presentations which led to the suggestion of immune alteration in the context of treatment with biologics.8–10
Statistical analysis: qualitative variables were represented in tables, using absolute frequencies and percentage relative frequencies and in simple bar graphs (a single variable) or stratified (two variables simultaneously) and sectorial diagrams. Quantitative variables were represented with the use of median and interquartile range minimum and maximum measurement (with normality having been previously studied through the Kolmogorov–Smirnov or Shapiro–Wilk test). To study the association between qualitative variables the chi squared test was used and to estimate medians between two separate groups the Mann–Whitney U test was used. The binary multivariate logistic regression model was used to detect independent predictors of infectious complications, calculating the odds ratio and 95% confidence interval for the different covariables of interest. A ROC curve was performed to determine the safe corticoid dose and a propensity test to assess the same as a factor of confusion. P values under .05 were considered significant. The software used was Microsoft Excel and IBM SPSS version 22.0.
Research was conducted respecting the legal framework in Uruguay and data were treated confidentially. The results were used for training and academic purposes.
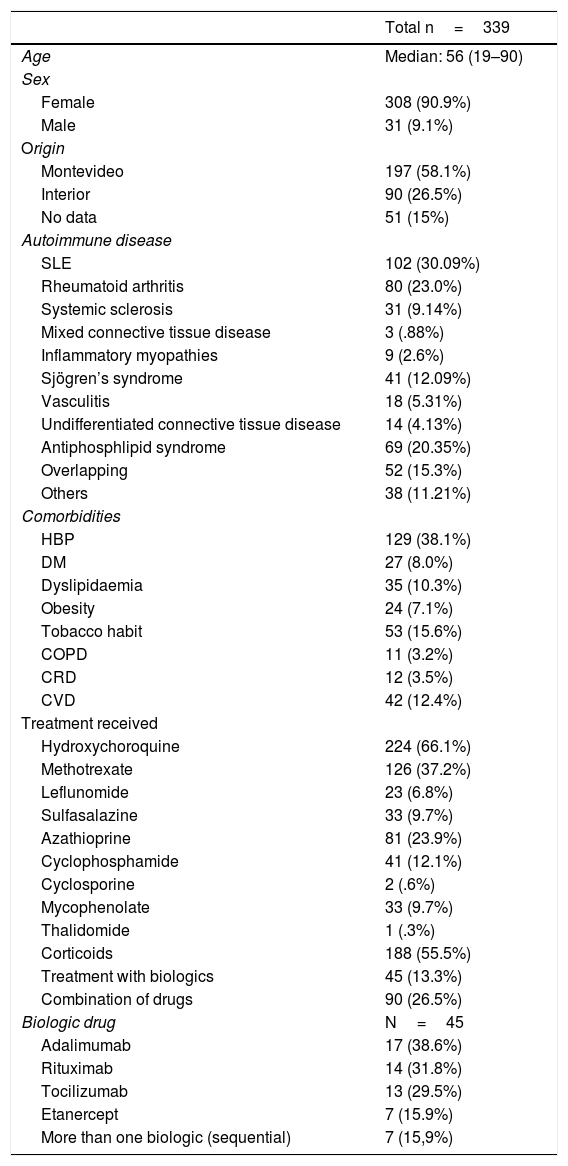
ResultsThe total of patients included in the study were 339, with a median age of 56 years and age range between 19 and 90; 308 cases (90.9%) were female and 31 (9.1%) male.
In the population studied risk factors identified were cardiovascular, DM in 27 (8.0%), HBP in 129 (38.1%), dyslipidaemia in 35 (10.3%), obesity in 24 (7.1%). Other chronic comorbidities were COPD in 11 (3.2%) and CRD in 12 (3.5%), tobacco habit in 53 (15.6%) and CVD in 42 (12.4%).
With regard to the SAD diagnosis, 102 cases corresponded to SLE (30.1%), 80 to RA (23.6%), 69 to antiphospholipid syndrome (APS) (20.4%) and 41 to Sjögren`s syndrome (12.1%). Fifty two cases of overlapping of SAD were found. The most frequent overlapping were SLE with RA and SLE with Sjögren’s syndrome, with 12 cases each, followed by seven cases of RA overlapped with Sjögren’s syndrome. The remaining 21 cases were varied combinations of SAD.
With regard to treatments received, hydroxychloroquine (HCQ) was the drug most frequently prescribed, in 224 cases (66.1%), followed by corticoids in 188 (55.5%), with a mean dose of 15mg/day. The prescribed synthetic immunosuppressants in descending order were: methotrexate, azathioprin, mycophenolate and leflunomide.
Biologics were used in 45 cases (13.3%), which were: adalimumab 17 (38.6%), rituximab 14 (31.8%), tocilizumab 13 (29.5%) and etanercept 7 (15.9%). Seven patients (15.9%) received more than one biologic sequentially.
In 90 patients (26.5%) drugs were combined (Table 1); 143 cases (46.9%) presented with infectious complications, 137 non opportunistic infections of different aetiologies (96.4%). Primary cases of APS were excluded in this analysis.
Population description.
| Total n=339 | |
|---|---|
| Age | Median: 56 (19–90) |
| Sex | |
| Female | 308 (90.9%) |
| Male | 31 (9.1%) |
| Origin | |
| Montevideo | 197 (58.1%) |
| Interior | 90 (26.5%) |
| No data | 51 (15%) |
| Autoimmune disease | |
| SLE | 102 (30.09%) |
| Rheumatoid arthritis | 80 (23.0%) |
| Systemic sclerosis | 31 (9.14%) |
| Mixed connective tissue disease | 3 (.88%) |
| Inflammatory myopathies | 9 (2.6%) |
| Sjögren’s syndrome | 41 (12.09%) |
| Vasculitis | 18 (5.31%) |
| Undifferentiated connective tissue disease | 14 (4.13%) |
| Antiphosphlipid syndrome | 69 (20.35%) |
| Overlapping | 52 (15.3%) |
| Others | 38 (11.21%) |
| Comorbidities | |
| HBP | 129 (38.1%) |
| DM | 27 (8.0%) |
| Dyslipidaemia | 35 (10.3%) |
| Obesity | 24 (7.1%) |
| Tobacco habit | 53 (15.6%) |
| COPD | 11 (3.2%) |
| CRD | 12 (3.5%) |
| CVD | 42 (12.4%) |
| Treatment received | |
| Hydroxychoroquine | 224 (66.1%) |
| Methotrexate | 126 (37.2%) |
| Leflunomide | 23 (6.8%) |
| Sulfasalazine | 33 (9.7%) |
| Azathioprine | 81 (23.9%) |
| Cyclophosphamide | 41 (12.1%) |
| Cyclosporine | 2 (.6%) |
| Mycophenolate | 33 (9.7%) |
| Thalidomide | 1 (.3%) |
| Corticoids | 188 (55.5%) |
| Treatment with biologics | 45 (13.3%) |
| Combination of drugs | 90 (26.5%) |
| Biologic drug | N=45 |
| Adalimumab | 17 (38.6%) |
| Rituximab | 14 (31.8%) |
| Tocilizumab | 13 (29.5%) |
| Etanercept | 7 (15.9%) |
| More than one biologic (sequential) | 7 (15,9%) |
Regarding the number of episodes during the follow-up period, 95 patients (66.9%) presented with a single infection and 42 (33.1%) with two or more.
Out of the total cases, 53 (37.3%) presented with active disease (P .009) and 50 (35.2%) received a combination of drugs at the time of infection (P .036).
Within the non-opportunistic infections, respiratory infections were the most common, with 69 cases (48.6%): 45 (31.7%) acute community-acquired pneumonia, 26 (18.3%) acute tracheobronchitis and 5 (3 5%) otitis media. These were followed in frequency by urinary tract infections in 45 (31.7%) patients, skin and soft tissue in 25 (17.6%) and one case of acute bacterial meningitis.
The microorganism causing infection was identified in 49 cases (32.4%). The most common were Escherichia coli with 21 isolations (14.8%) and Streptococcus pneumoniae with 6 (4.2%). Seven cases (4.9%) corresponded to intrahospital infections.
Thirteen cases (9.2%) of opportunistic infections were observed with the following distribution: four cases of pulmonary tuberculosis, one of them subsequently associated with pneumonia, to Pneumocystis jirovecii, eight cases of herpes zoster and one patient with brain toxoplasmosis.
Regarding severity, 58 cases (40.8%) were severe infections; 38 cases (26.8%) required hospitalisation once, 12 (8.5%) twice and six patients (4.2%) were admitted to the Intensive Care Unit.
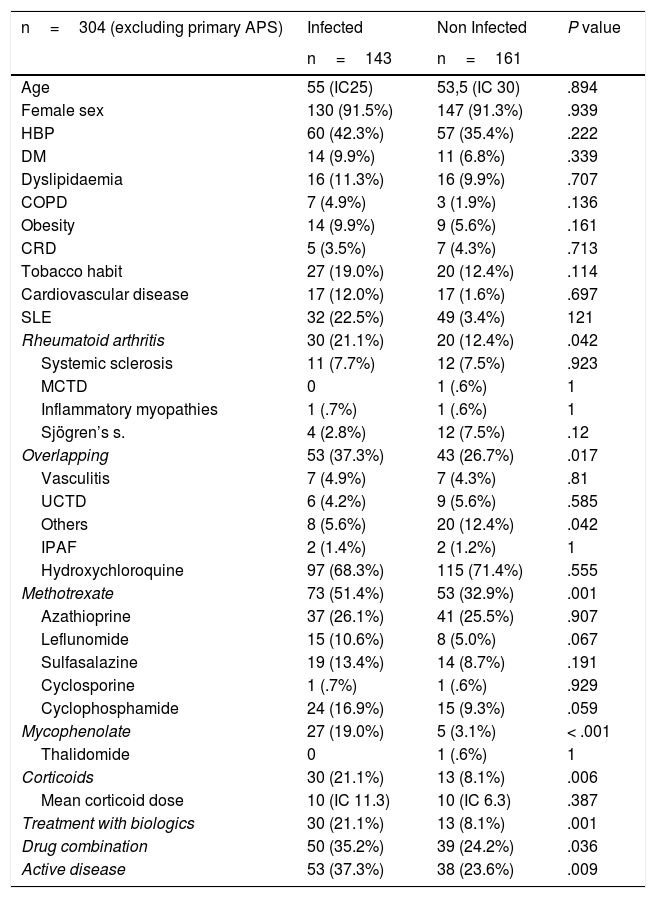
On comparing patient groups with or without infectious complications there was a significant difference in methotrexate, micophenolate, corticoid, biologic therapies, drug combinations, active disease, RA and overlapping variables. Regarding comorbidities, there were no statistically significant differences between both subgroups. A propensity study was performed to eliminate the bias attributable to treatment with corticoids and the use of biologics. A significant increase in risk remained which was two and a half times greater in patients with biologics compared with patients without biologics, OR 2.48 (CI 1.165–4.977) (Table 2).
Comparison between infected and non infected patients.
| n=304 (excluding primary APS) | Infected | Non Infected | P value |
|---|---|---|---|
| n=143 | n=161 | ||
| Age | 55 (IC25) | 53,5 (IC 30) | .894 |
| Female sex | 130 (91.5%) | 147 (91.3%) | .939 |
| HBP | 60 (42.3%) | 57 (35.4%) | .222 |
| DM | 14 (9.9%) | 11 (6.8%) | .339 |
| Dyslipidaemia | 16 (11.3%) | 16 (9.9%) | .707 |
| COPD | 7 (4.9%) | 3 (1.9%) | .136 |
| Obesity | 14 (9.9%) | 9 (5.6%) | .161 |
| CRD | 5 (3.5%) | 7 (4.3%) | .713 |
| Tobacco habit | 27 (19.0%) | 20 (12.4%) | .114 |
| Cardiovascular disease | 17 (12.0%) | 17 (1.6%) | .697 |
| SLE | 32 (22.5%) | 49 (3.4%) | 121 |
| Rheumatoid arthritis | 30 (21.1%) | 20 (12.4%) | .042 |
| Systemic sclerosis | 11 (7.7%) | 12 (7.5%) | .923 |
| MCTD | 0 | 1 (.6%) | 1 |
| Inflammatory myopathies | 1 (.7%) | 1 (.6%) | 1 |
| Sjögren’s s. | 4 (2.8%) | 12 (7.5%) | .12 |
| Overlapping | 53 (37.3%) | 43 (26.7%) | .017 |
| Vasculitis | 7 (4.9%) | 7 (4.3%) | .81 |
| UCTD | 6 (4.2%) | 9 (5.6%) | .585 |
| Others | 8 (5.6%) | 20 (12.4%) | .042 |
| IPAF | 2 (1.4%) | 2 (1.2%) | 1 |
| Hydroxychloroquine | 97 (68.3%) | 115 (71.4%) | .555 |
| Methotrexate | 73 (51.4%) | 53 (32.9%) | .001 |
| Azathioprine | 37 (26.1%) | 41 (25.5%) | .907 |
| Leflunomide | 15 (10.6%) | 8 (5.0%) | .067 |
| Sulfasalazine | 19 (13.4%) | 14 (8.7%) | .191 |
| Cyclosporine | 1 (.7%) | 1 (.6%) | .929 |
| Cyclophosphamide | 24 (16.9%) | 15 (9.3%) | .059 |
| Mycophenolate | 27 (19.0%) | 5 (3.1%) | < .001 |
| Thalidomide | 0 | 1 (.6%) | 1 |
| Corticoids | 30 (21.1%) | 13 (8.1%) | .006 |
| Mean corticoid dose | 10 (IC 11.3) | 10 (IC 6.3) | .387 |
| Treatment with biologics | 30 (21.1%) | 13 (8.1%) | .001 |
| Drug combination | 50 (35.2%) | 39 (24.2%) | .036 |
| Active disease | 53 (37.3%) | 38 (23.6%) | .009 |
IPAF: interstitial pneumonia with autoimmune findings; MCTD: Mixed connective tissue disease; UCTD: undifferentiated connective tissue disease.
An ROC curve was made to estimate the “safe” corticoid dose, which was estimated to be 6.25mg/day, with a sensitivity of .81 and specificity of .33. It was shown that this curve had an area of .618 with no significant difference of .5 (P value=.09).
Through analysis of the frequency of infectious complications, according to type of SAD through univariate binary logistic regression, it was observed that there was a greater risk of infections in cases of RA and overlapping, with an increase of over double the risk with an OR of 2.55 (CI 1.157–5.622) and 2.095 (CI 1.058–4.15), respectively.
Comparison of the frequency of infections in the subgroup of patients with SLE, revealed significant differences with methorexate, with a risk five times greater, OR 4.91 (CI 1.19–20.18). No increased risk was found with significant difference for the other treatments. In the subgroup of RA it was observed that the use of hydroxycholoroquine or sulfasalazine reduced the risk of infections by over 50%, with an OR of .145 (CI .039–.544) and .136 (CI .031–.598), respectively.
DiscussionInfections are a common complication in patients with SAD. In this study we observed it in 46.9% of cases, similar to that reported in international series.1,6,7,12
Infections were more common in cases with RA and in forms of overlapping than in SLE patients (with statistically significant differences), the situation was different from that reported in other studies. One possible explanation could be the greater access to biologics in patients with RA than with SLE, through the National Resources Fund in Uruguay, and that patients with biologics have a greater frequency of infections.
Forty per cent of infections were severe, these being considered of opportunistic cause or requiring hospitalisation, the use of intravenous antibiotic therapy, or those leading to death. Non severe infections were classified as those which were not included in the definition as severe.8–10
In this cohort, most cases were non-opportunistic infections (95%). A third of the patients presented with more than one episode of infection.
The most common infections were respiratory, urinary, skin and soft tissue, similar to those reported in other series.2,4,6,7,10–17 Almost 5% corresponded to intrahospital infections.
Regarding that published relating to microbiology, bacteria are the most frequently isolated microorganisms, followed by virus and fungal infections. The frequency of non-specific bacterial infections is similar to that occurring in the general population, whilst some of them, such as infections caused by Salmonella more often occur in patients with SLE who have lower levels of complementary, asplenia or immunosuppressant therapy.2,4,6,7,10–17
In this series the microbiologic diagnosis was achieved in a third of cases. Bacterial infections were the most common, with Escherichia coli and Streptococcus pneumoniae as the most prevalent microorganisms, coinciding with the microbiological profile of the most common infectious focal points in this and other series.2,4,6,10,11,13,14,16,17
The 13 cases of opportunistic infections were observed in patients with RA and with SLE.
Four cases of pulmonary tuberculosis were found. Tuberculosis was described as most common in subjects with SLE or RA rather than in the general population. With regards to clinical presentation, pulmonary tuberculosis is the most common form (like in this series), but extrapulmonary forms were described with greater frequency and with more serious symptoms than in the general population.2,18–21
Eight cases of herpes zoster were recorded in patients in treatment with synthetic immunosuppressants. A higher frequency of herpes zoster was shown and several studies reported that there was no link with disease activity.2,22,23
The most common mycotic infections were caused by Candida albicans and by Pneumocystis jirovecii (PCP). In this series, a single case of PCP was reported in patients with corticoid treatment associated with several synthetic immunosuppressant drugs.
The risk of opportunistic infections has to be present in this population to carry out screening, prophylaxis and vaccination when applicable.30
Disease activity is a separate risk factor from the development of infectious complications in people with SAD. In the cohort analysed, patients with an active disease presented with a greater frequency of infections, in keeping with that reported in international literature.1–7
The risk of infection varies according to the treatments administered, as reported in several European and North American RA databases.
When the frequency of infections was analysed, depending on the treatments received, the drugs which were associated independently with a higher risk of infection were biologics, mycrophenolate, methotrexate and corticoids alone or combined. The combined use of immunosupressants was significantly associated with a higher risk of infections.
In this series, treatment with biologics had a two and a half times greater risk of infections compared with cases with no biologic treatment, which is similar to that reported in international series.6,9,10,25,27 However, it is of note that the use of treatments with biologics remains a separate risk factor to the use of corticoids.
The German register RABBIT shows that the risk of infections with biologics is greater than treatments with non biologics, and that it increases directly proportionally with the increase in the corticoid dose in both groups.25
In the Spanish BIOBADASER biologics register, 35% of adverse events were infections, with a mortality of almost 32%.27
In a meta-analysis of 70 studies in RA, which involved over 32,000 patients, an overall increase of the risk of opportunistic infections was demonstrated, with 1.7 infections per 1,000 patients treated with biologic therapies.9 The British data register (BSRBR) refers to an infection risk which is multiplied by 4, particularly in the first three to six months with the use of biologic therapies, with tuberculosis being the most common opportunistic infection.6 In another meta-analysis, the infection rate was compared for several inflammatory diseases between anti-TNFs, demonstrating a slightly lower risk with etanercept, with a lower frequency of herpes zoster, tuberculosis and other opportunistic diseases.10
In this cohort analysed, treatment with corticoids was associated with a statistically significant risk of infections. This finding coincided with the different North American registers (CORRONA) and European ones (BSRBR and RABBIT), which shows an increased risk of between 1.5 and 2 times, even when low doses were used (5mg/day prednisone) and five times higher with the use of prednisone above 15-20mg/day.6 Furthermore, each increase of the dose of corticoids multiplied the risk of suffering from a serious infection, with the mean dose being 7.5mg/day.12,16 Although in this study, the “safe” dose of corticoids was estimated to be 6.25mg/day, the difference was not statistically significant, and this fact may be attributed to the loss of data of the dose received and to the sample size.
In this series we found there were significant differences in the frequency of infections regarding the synthetic immunosuppressant drug received.
One outstanding finding is the increased risk associated with methotrexate, in keeping with that reported in the literature. In the CORRONA register a greater risk of infection was demonstrated in patients treated with methotrexate than with other synthetic disease-modifying drugs. However, a meta-analysis published in 2017, which compares the risk of infections with biologics and with biologics combined with methotrexate did not show any significant differences between the two groups.26 One possible explanation to this controversy is that methotrexate is the drug most commonly combined with biologic therapy in patients with RA, as well as being combined with other synthetic immunosuppressants.
Regarding risk of infections associated with the use of mycophenolate, its effect should not be considered in an isolated fashion, since it is regularly used in patients with severe SLE, where there is greater disease activity, compromise of major organs and it is also combined with other drugs such as corticoids.
Hydroxychloroquine has antimicrobial properties, and is also an immunomodulator. Several studies describe the use of HCQ in SAD as a protective factor against the development of infections.16,22,23 In this cohort we saw that hydroxychloroquine use, like that of sulfasalazine, led to a drop of over 50% risk of infections in RA patients.
There are several limitations to this study. Firstly, those derived from its retrospective design with an information bias and the existence of low frequency categories (which hinder statistical analysis, especially in opportunistic infections). Like in the majority of series, the under-recording of non severe infections is due to the fact that these consultations are resolved in other care centres which do not specialize in SAD (information bias). Other limitations are the absence of microbiologic data in all cases, difficulties in recording the accumulated dose of corticoids and administration times, together with immunisation data. Within its strengths, the authors point out the number of patients included, the extent of autoimmune pathologies and essentially, that this is the first national study on infections in patients with SAD.
All of the above is a trigger for the development of new studies of a prospective nature and instrumentation of measures for reducing the frequency of infections in this population, such as the use of screening protocols and infection prophylaxis in patients with SAD prior to beginning immunosuppressant treatments.
ConclusionsInfections are a common complication in patients with SAD. They are linked to the disease itself and to the treatments administered, essentially corticoids and biologics. The correct identification and treatment of them, together with rational use of the drugs may reduce their impact on this population.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Consani Fernández SA, Díaz Cuña CL, Fernández Rey L, Rostán Sellanes S, Maciel Oleggini G, Facal Castro JA. Infecciones en enfermedades autoinmunes sistémicas. Reumatol Clin. 2021;17:582–587.