Vogt–Koyanagi–Harada (VKH) disease is a rare multi-systemic disease, of autoimmune origin that affects the ear, eyes, central nervous system (CNS), and the skin. It shares some autoimmune mechanisms with rheumatoid arthritis (RA) but is extremely rare in association.
ObjectiveTo describe the clinical features of the presentation of VKH disease in a patient with a previous diagnosis of RA in clinical remission.
Case reportWe describe the case of a 47-year-old female patient with RA (RF+ ACCP+) diagnosed in 2014, initially treated with methotrexate, and then with hydroxychloroquine. We highlight the presence of associated metabolic syndrome, and increasingly evident alopecia.
The patient attended the emergency department with intense headache and blurred vision. Given the suspicion of intracranial hypertension, she was admitted to the neurology department who then consulted the rheumatology and ophthalmology departments who, after their initial examination described the following findings:
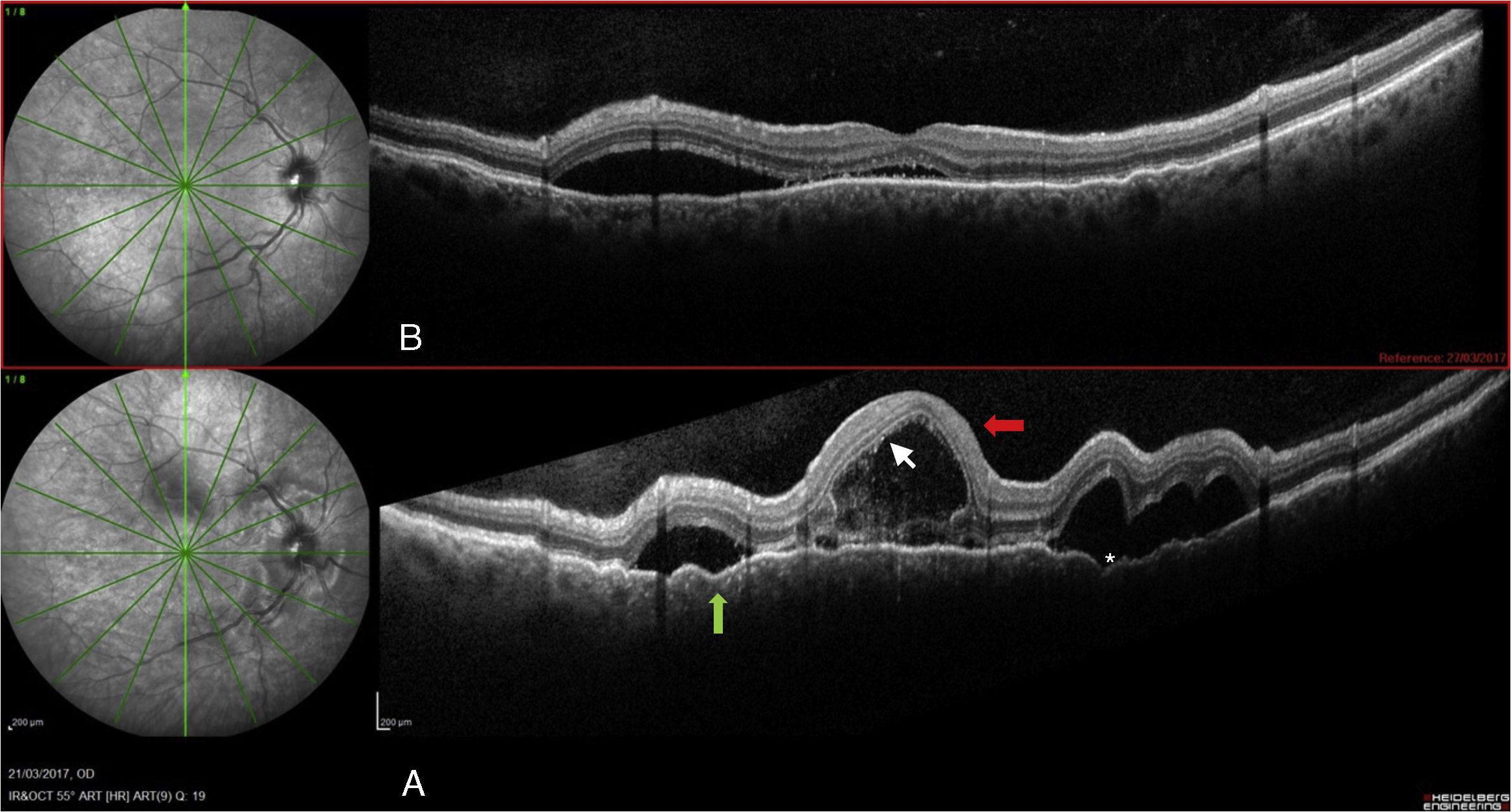
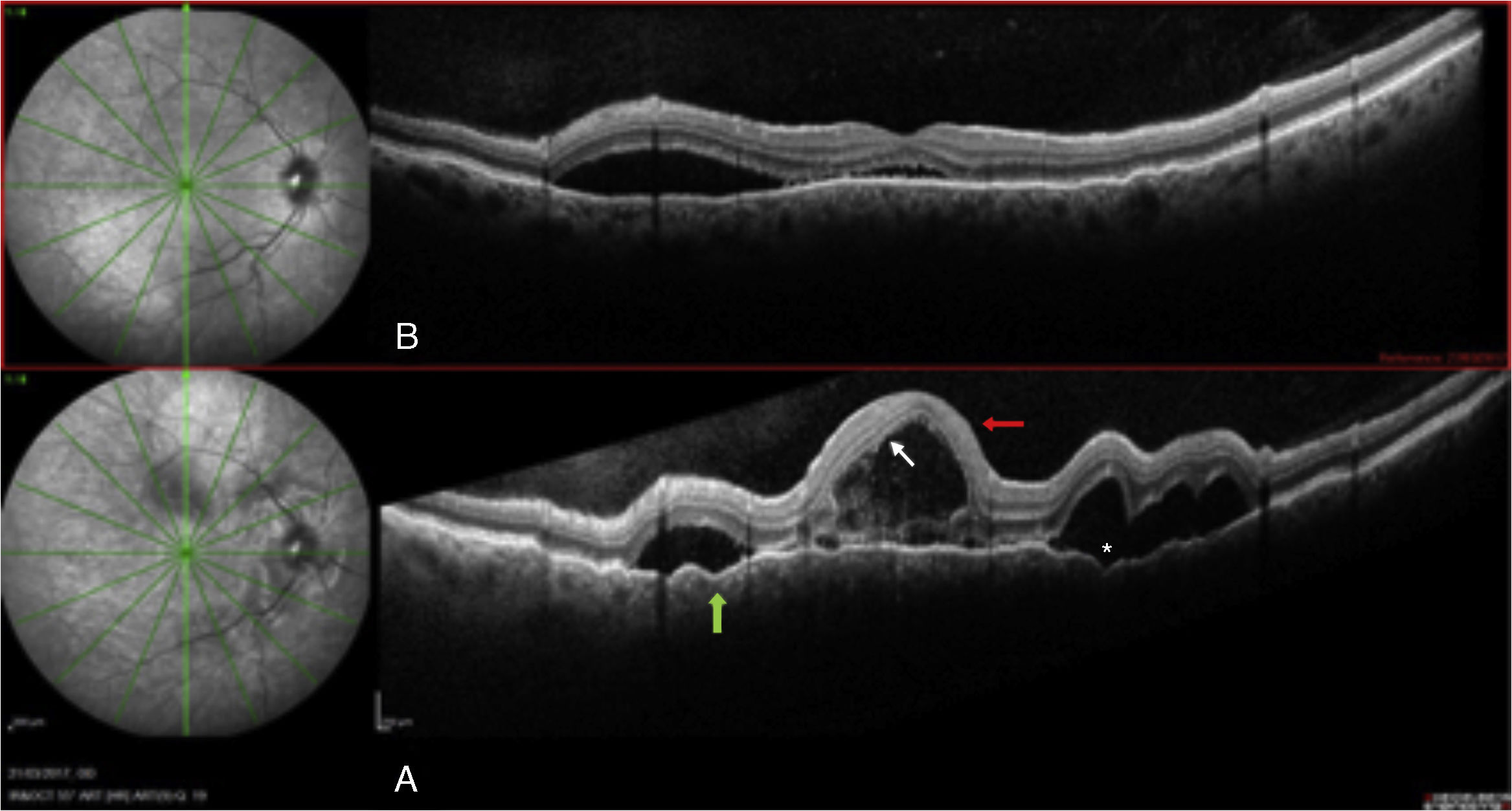
Visual acuity (VA) .16 and .3, respectively, with no inflammatory signs in the anterior segment. OCT-spectralis® (Heidelberg) (SD-OCT)showed: multilobulated serous detachments affecting the macular and peripapillary area, septation of the detachments was observed together with increased choroid thickness (734μ, Figs. 1 and 2). Autofluorescence revealed hypoautofluorescence with a hyperautofluorescent halo surrounding the detachments, and fluorescein angiography showed the typical “starry sky” image with multiple leakage sites, and diffusion in the late stages.
- 1.
No history of perforating trauma or prior surgeries.
- 2.
No evidence of previous eye disease.
- 3.
Bilateral eye involvement (A or B, according to the stage of the disease):
- A.
Early manifestations:
- •
Presence of diffuse choroiditis (focal areas of subretinal fluid or bullous serous retinal detachment).
- •
If the lesions are not that evident, there should be: areas of delayed choroidal filling (FAG), placoid areas of hyperfluorescence, and optic nerve staining.
- •
Diffuse thickening with no posterior scleritis (ultrasound).
- B.
Late manifestations:
- •
History of previous manifestations.
- •
Ocular depigmentation: Sugiura sign or sunset glow fundus.
- •
Others such as numerous chorioretinal scars, recurring or chronic anterior uveitis.
- 4.
Neurological and or auditory findings:
- •
Meningismus.
- •
Tinnitus and/or hearing loss.
- •
Pleocytosis in cerebrospinal fluid.
- 5.
Dermatological findings, after uveitis:
- •
Alopecia, vitiligo, poliosis.
VKH syndrome is defined as:
- •
Complete, when the patient meets criteria 1 to 5.
- •
Incomplete, when the patient meets criteria 1 to 3, plus the 4th or 5th criteria.
- •
Probable, when only ocular criteria 1 to 3 are present.
Therefore, our patient fully met the clinical criteria established by the American Uveitis Society (AUS), published in 2001, in sections 1, 2, 3A, 4: pleocytes in CSF, and 5 alopecia. Finally, a diagnosis of complete acute uveitic stage VKH was made.
CSF showed the presence of pleocytosis.
Microbiological study discounted acute or latent tuberculosis infection.
QuantiFERON®-TB gold: negative.
CSF culture at 45 days: culture negative for mycobacteria, negative for bacteria, and negative for mycosis.
Serology for hepatitis C virus, hepatitis C, HIV and syphilis was negative.
There is currently no consensus as to the therapeutic regimen for patients with VKH disease.
Treatment was started with 3 1g boluses of methylprednisolone and improved VA was achieved: .7 in both eyes.
In relation to the pharmacological management of our patient, it is important to highlight her history of an associated metabolic syndrome manifested by chronic hypertension, type 2 diabetes mellitus treated with oral antidiabetic agents, obesity and dyslipidaemia. After induction treatment with intravenous boluses of corticosteroids, the patient suffered metabolic decompensation complicated by sustained hyperglycaemia and hypertensive crisis, therefore we decided to start early treatment with an oral immunosuppressant as a steroid saver, which would also ensure induction initiated with corticosteroids to serve as bridge therapy until starting biological therapy.
We studied the different treatment options and straight away discounted methotrexate due to a fatty liver and hypertransaminasaemia, and known prior intolerance. We ruled out cyclosporine due to its potential nephrotoxic effect, and the coexistence of kidney failure risk factors (chronic hypertension and diabetes mellitus). A major limitation to treatment in our case was the patient's obesity. Therefore, given our experience with the drug we decided to start mycophenolate mofetil as a steroid saver until we could establish biological therapy as the definitive treatment, in this case adalimumab.
The patient was monitored using SD-OCT (Heidelberg) obtaining sequential images that showed a reduction in the serous detachments, persistent impairment of the external retina, and residual perimacular hyperautofluorescence.
DiscussionDiagnosis of VKH disease is clinical. The characteristic eye involvement is non-infectious granulomatous panuveitis.1 The presence of neurological manifestations makes an urgent differential diagnosis necessary given the possibility of infection of the CSF. Therefore, detailed medical history and a complete examination are compulsory.
RA associated with VKH disease is extremely rare.2 The most common eye involvement in RA is keratoconjunctivitis sicca, which is present in 25% of patients. In contrast, bilateral panuveitis is unusual.3
By means of CT and MRI we ruled out the presence of space-occupying, vascular or inflammatory lesions.
By testing the CSF, we found the presence of pleocytosis, and the microbiological study discounted acute or latent tuberculosis infection.
Given the patient's history of RA, ocular ultrasound ruled out posterior scleritis.
Eventually we made a diagnosis of complete VKH in the acute uveitic phase. Our patient fully met the clinical criteria of the AUS, published in 2001, in sections 1, 2, 3A, 4: pleocytosis in CSF, and 5: alopecia.
Our patient responded favourably to systemic corticosteroids, however, due to her prior metabolic syndrome and to prevent complications associated with prolonged steroid use, administration of immunosuppressant treatment was necessary from the start of the disease. To achieve complete remission, mycophenolate mofetil was prescribed as bridge therapy until starting a TNF inhibitor.4–6
ConclusionDiagnosis of VKH disease is challenging, and differential diagnosis is compulsory. A multidisciplinary approach to autoimmune diseases enables diagnosis and prompt treatment, improves the prognosis of these diseases, and significantly reduces sequelae to the vital organs.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Paz-Solarte JA, Hernández-Garfella M, Rueda-Cid A, Campos-Fernández C, Calvo-Catala J. Rara asociación entre la enfermedad de Vogt-Koyanagi-Harada y la artritis reumatoide. Reporte de un caso clínico de la consulta multidisciplinar de uveítis. Reumatol Clin. 2020;16:59–61.