The double and simultaneous molecular interaction between antigen-presentig cells (APC) and T lymphocytes is essential for the optimal activation of the immunological response and requires the participation of two membrane receptor groups. Abatacept is a fusion protein that selectively modulates one of these two ways, by binding to CD80 and CD86 receptors on APC. In this way, the drug inhibits T cell activation, selectively blocking the specific interaction of CD80/CD86 receptors to CD28 and, therefore, inhibiting T cell proliferation and B cell immunological response. This pharmacological action results in the normalization of inflammatory mediators in rheumatoid arthritis patients and in a safe and efficacious clinical response. Abatacept in combination with methotrexate prevents the progression of joint damage and improves physical function in rheumatoid arthritis patients.
La interacción molecular doble y simultánea entre las células presentadoras de antígeno (CPA) y los linfocitos T es imprescindible para la activación óptima de la respuesta inmunitaria y requiere de la participación de dos grupos de receptores de membrana. El abatacept es una proteína de fusión que modula selectivamente una de estas dos vías, uniéndose a los receptores CD80 y CD86 de las CPA. De esta forma el fármaco inhibe la activación de las células T, bloqueando selectivamente la unión específica de los receptores CD80/CD86 al CD28 y como consecuencia inhibiendo la proliferación de las células T y la respuesta inmunitaria de las células B. Esta acción farmacológica se traduce en la normalización de los niveles de los mediadores inflamatorios en los enfermos con artritis reumatoide y en una respuesta clínica segura y eficaz. El abatacept en combinación con metotrexato evita la progresión de la lesión articular y mejora la función física en enfermos con artritis reumatoide.
Rheumatoid arthritis (RA) is an autoimmune disease, polygenic in nature, characterized by polyarthritis with systemic manifestations and increased and severe morbidity.1,2 RA affects 0.5%–1% of the population, causing a decrease in quality of life, significant physical disability and a substantial economic cost.3–6 The clinical expression of disease is varied, ranging from mild self-limiting forms to a very aggressive, rapid evolution that culminates with the destruction of the affected joint and the resulting disability.7
Genetic studies have confirmed the existence of a genetic substrate, partly related to certain genes encoding proteins involved in T cell responses.1 These findings reinforce the importance of the role attributed to T cells in the initiation and perpetuation of the abnormal immune response in this disease.8
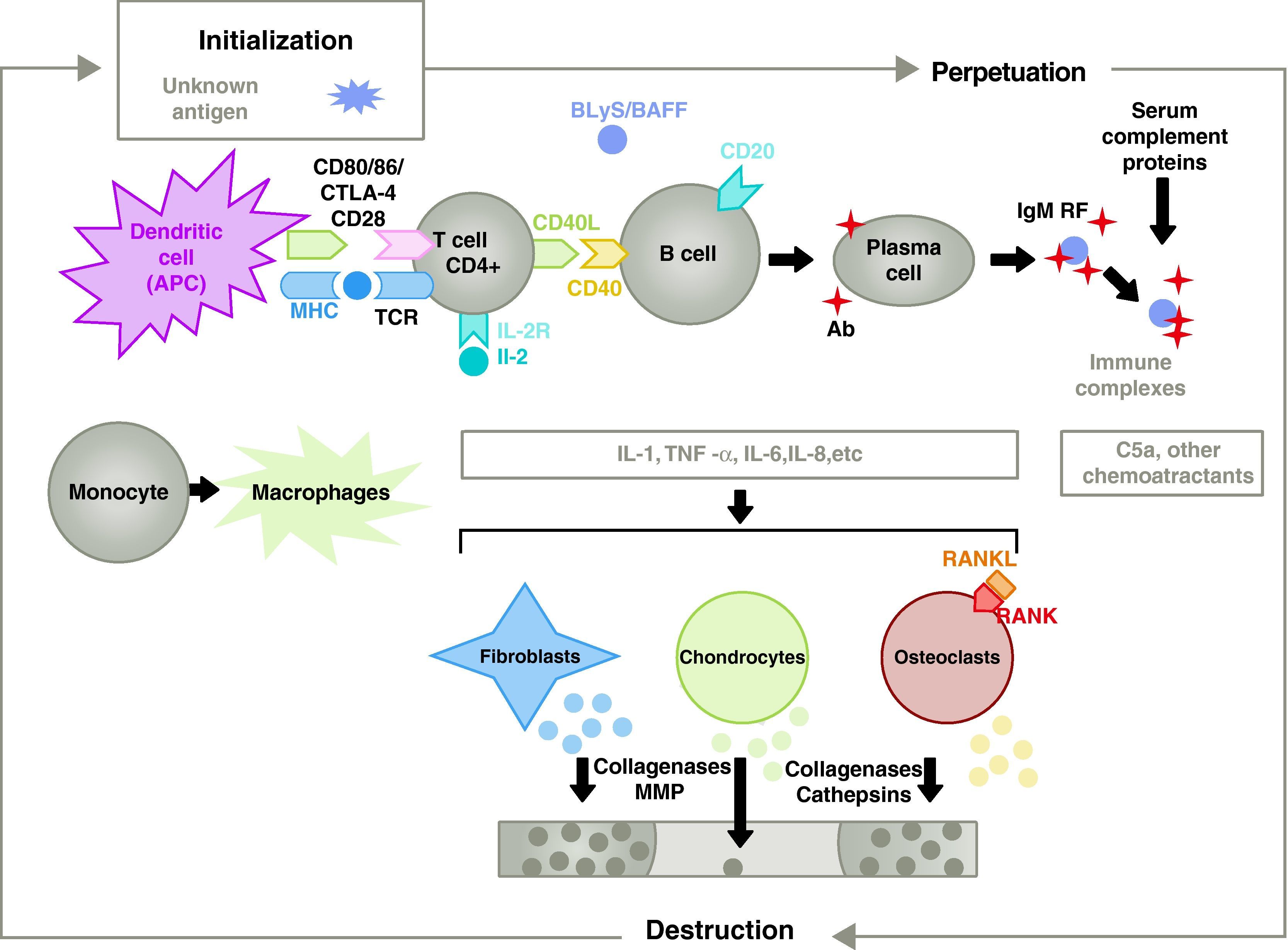
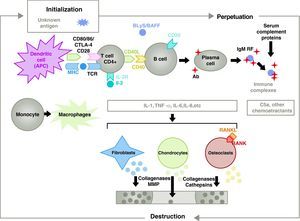
The pathogenesis of RA is complex and involves different cell populations related to the innate and adaptive immune response. Resident cells in the synovium, such as fibroblastic synoviocytes B or macrophages of the intima, and inflammatory cells from the blood as T lymphocytes, B lymphocytes and monocytes9 are involved in the pathogenesis. They all contribute to the aggressive transformation of the synoviocyte B phenotype and the development of an intense inflammatory infiltrate with the end result of cartilage and subchondral bone destruction10,11 (Fig. 1).
Pathophysiology of rheumatoid arthritis. General pathophysiological organization of rheumatoid arthritis. AC, antibody; BAFF, B cell activating factor; BLyS, B lymphocyte stimulator; CD, cluster of differentiation; CPA, antigen-presenting cell; CPH, MHC; CTLA4, lymphocyte-associated antigen 4 T cytotoxic C5a fraction complement 5a, FR, rheumatoid factor; Ig, immunoglobulin; IL, interleukin; MMP, matrix metalloproteinases; RANK, receptor activator of nuclear factor B kappa; RANKL, receptor activator ligand for the nuclear factor B kappa; RCT, T cell receptor; TNF, tumor necrosis factor.
Current treatment of RA is based on the administration of Disease modifying antirheumatic drugs (DMARDs) used alone or in combination.12 These drugs slow joint destruction, i.e., they are able to modify the natural course of the disease.4,13 However, the percentage of patients with a satisfactory clinical response is low and often requires the addition of a biologic drug in a high percentage of patients.9,13–15
In recent years new molecules and therapeutic targets whose blockade could reduce or eliminate the chronic inflammatory response have been identified. One of these new molecules is abatacept. Abatacept is a fully humanized protein construct, consisting of the extracellular domain of human cytotoxic T lymphocyte-associated antigen 4 (CTL4) and a genetically engineered fragment of the Fc region of human immunoglobulin G1 (IgG1), which inhibits the costimulation T cells acting on the true nucleus of the immune response and, therefore, at the onset of the disease.
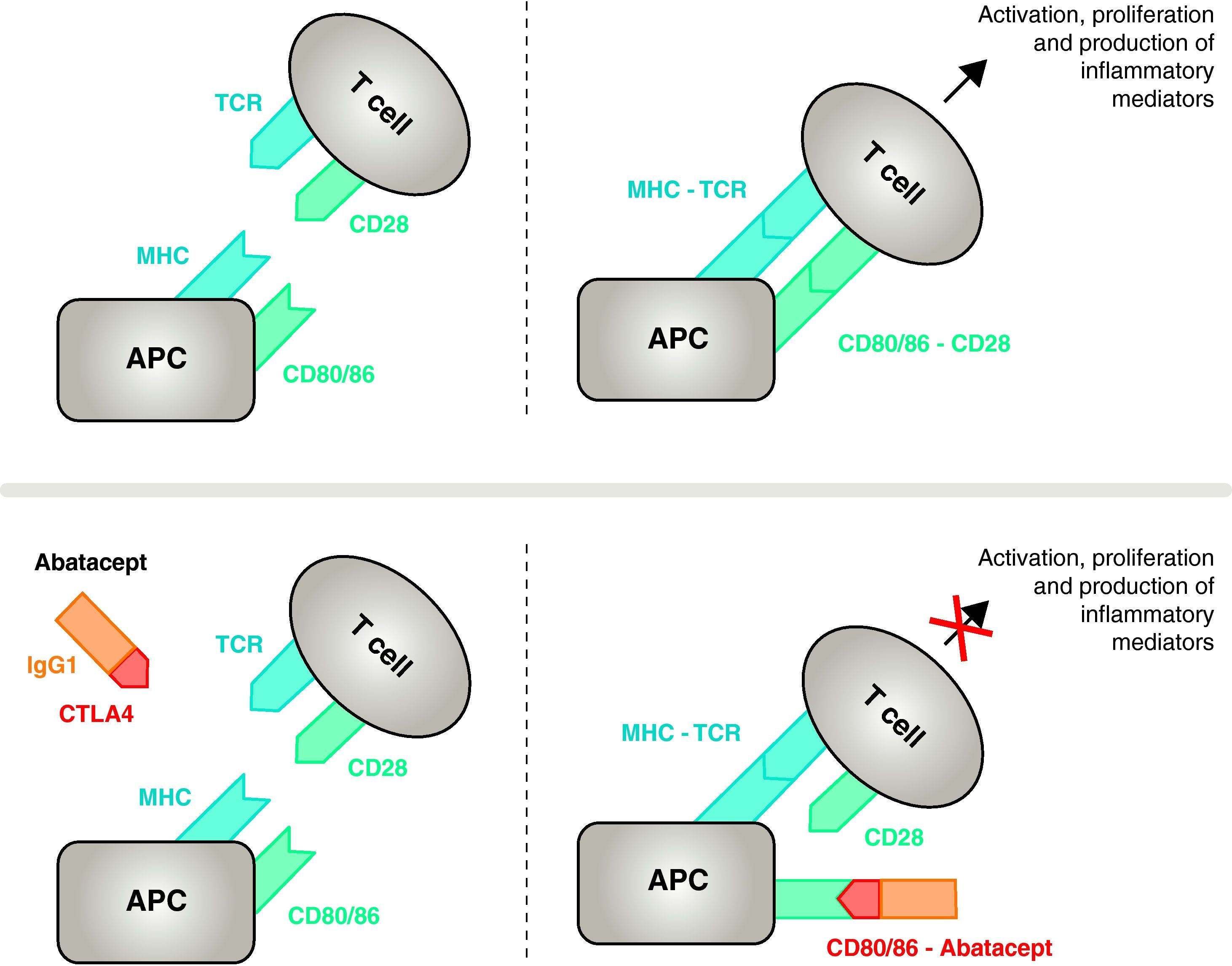
T Cell ActivationEffective immune activation of T cells requires the participation of two groups of membrane receptors on antigen presenting cells (APC)14 (Figs. 1 and 2). The first is the vehicle used by APCs to provide the previously processed specific antigen to the T cell. Despite the enormous effort devoted to this research, we are still unable to identify the arthritogenic antigens triggering RA.8 Presentation by APCs of an antigen against which a specific immune response is mounted is organized through a trimolecular complex comprising: molecules of major histocompatibility complex (MHC) present in the APC, the antigen against which the immune response develops and a membrane receptor on the T cell (TCR) specific for that antigen15 (signal or signaling pathway of the immune response 1).
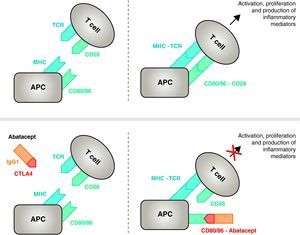
Mechanism of action of abatacept. The abatacept fragment comprising the extracellular domain of CTLA4 binds to CD80/CD86 receptors, preventing or displacing its interaction with the CD28 receptor. In this way, it selectively blocks the specific binding of CD80/CD86 to CD28 receptor, which is, pathophysiologically, a block of the second signal for immune activation and, therefore, activation of T cells CPA, antigen-presenting cell; MHC, major histocompatibility complex; TCR, T cell receptor.
To inhibit full activation, T cells require a second set of intercellular communication receptors between APCs and T cells that occurs through costimulatory pathways and constitutes the so-called 2 signal immune response.14 Although several costimulatory pathways exist, one is essential, the binding of receptors CD80 (B7-1)/CD86 (B7-2) on the membrane of the CPA with the CD28 receptor on T cells.10,16 Simultaneous activation of both triggers intense intracellular signaling in T cells, essential for full activation, proliferation, survival and cytokine production 8. At 24–48h of the activation of T lymphocytes, the same intracellular signaling initiates a regulatory mechanism that aims to deactivate the response itself. This induces the expression of CTLA411 on the lymphocyte cell membrane with the task of competing with CD28 because of its greater binding affinity to CD80/CD86.17,18
The activation of both T cell subsets, CD4+ and CD8+ depends on the CD28 costimulatory receptor. CD4+ T cells are helper T cells. They recognize the peptides presented by MHC class II molecules present on the APC. These antigens originate from the exogenous pathway that processes pathogens such as bacteria. Many autoimmune diseases are associated with a pathological response of CD4+ T cells. For their part, CD8+ T cells are cytotoxic lymphocytes (CTL). CD8+ T cells recognize antigens, mainly viral and tumoral presented by class I molecules MHC. Upon activation, CD8+ cells mediate target cell destruction through the production of perforin, granzymes and interferon (IFN)-g. Both subtypes of T cells are activated by costimulation with CD2815, although the activation of CD8+ T cells is less dependent on this costimulation pathway. In fact, while all CD4+ cells express CD28 on their membrane, this only occurs in about 50% of CD8+.19 In addition, CD4+ cells have been shown to exhibit a greater response to CD2820 binding. Moreover, the CD28 promoter is not an absolute requirement for activation of CTL.21 All of which would provide a double therapeutic benefit in clinical practice. On the one hand, abatacept acts preferentially on the target cell in the pathogenesis of the disease. Furthermore, the reduced action on the activity of CD8+ lymphocytes would ensure a better safety profile in terms of viral and tumoral complications.
Activation of CD4+ T cells is the starting point for a proinflammatory cascade with production of large amounts of cytokines and cell proliferation that, if perpetuated and maintained, as in RA, leads to very active chronic inflammation, capable of destroying the tissues in which it is triggered, mostly the joints in the case of the RA8 (Fig. 1). The synovium begins to proliferate due to infiltrating cells from the blood, including T lymphocytes themselves and their subtypes, as well as B lymphocytes The monocytes differentiate into macrophages and osteoclasts and also activate articular chondrocytes. In this environment there are large amounts of proinflammatory cytokines such as interleukin (IL)-1, IL-6 and tumor necrosis factor (TNF) and many others. B cells also produce autoantibodies such as rheumatoid factor or anti citrullinated peptide antibodies. All of these lead to the destruction not only of the synovial membrane but also the underlying bone and cartilage.22
Biotechnology in the Treatment of Rheumatoid ArthritisBecause of the aforementioned research, biotechnology manufacturing of various molecules aimed at blocking specific targets has been developed and commercialized. The first generation was characterized by the appearance of TNF neutralizing drugs: etanercept, infliximab and adalimumab and anakinra, which inhibit the action of IL-1. Subsequently, new molecules have emerged, such as abatacept, to modulate the costimulation of the immune response, certolizumab and golimumab to block TNF, rituximab against the CD20 receptor of B lymphocytes, and tocilizumab which blocks IL-6.7,23–26
Despite huge leap in terms of therapeutic efficacy due to the introduction of these drugs, a substantial percentage of patients, estimated between 25% and 40%, do not respond to currently marketed drugs or biologicals or are affected by the occurrence of adverse events.27–32 The need to improve this situation remains an encouragement in the pursuit and development of new molecules aimed at regulating different therapeutic targets that could improve the therapeutic efficacy, such as the case of abatacept, which selectively modulates the activation of T cells.33
Abatacept is a protein construct that is produced through recombinant DNA technology in hamster ovary cells.34,35 This molecule was designed to interfere with the regulation of costimulatory pathways in T cells, which play an important role in the pathogenesis of various autoimmune diseases, infections, rejection of transplanted organs and tumor immunity.36
Abatacept is used in combination with methotrexate in RA patients who have had an inadequate response or intolerance to other DMARDs, including methotrexate (MTX) or an inhibitor of TNF-alpha. In polyarticular juvenile idiopathic arthritis it is indicated in patients 6 years or older who have had an inadequate response to other DMARDs including at least one TNF neutralizing drug.35
Mechanism of Action of AbataceptAbatacept is a selective modulator of the CD80/86-CD28 costimulatory signal, and as previously discussed it is essential for activation of T cells Abatacept inhibits T cell activation, selectively blocking specific binding of the CD80/CD86 receptor in APC to CD28 on T cell (Fig. 2).22,37 The pharmacological strategy looks to inhibit the accelerated immune/inflammatory response, characteristic of the disease, and restore normal homeostasis in the immune system. In fact, competition between endogenous CD28 and CTLA4 for binding to CD80/86 is the physiological mechanism used to regulate and, where appropriate, conclude a normal immune response. Abatacept, by blocking the binding of CD80/86 to CD28 inhibits the transmission of a second signal of the immune response, which indirectly produces a negative signal on T cell activation. In addition, abatacept probably has a greater effect in preventing the formation of a costimulatory signal in T cells, inactivating those already active, that are not bound to T cell CTLA4
Supporting Drug for Use1. Why is abatacept included in the group of immunomodulatory drugs? Basically, because it produces cell depletion, especially of T cells due to the pharmacological action exerted by not blocking selectively a particular cytokine, avoiding the radical suppression of essential pathways for proper immune response functioning.8
2. How does it prevent the binding of the Fc region of the molecule to its receptor? The Fc region of abatacept is genetically modified, so that does not bind to CD16 and CD32 receptors, and does so very weakly to the CD64 receptor. This design circumvents cellular responses mediated by the Fc receptor such as antibody-dependent cellular cytotoxicity (ADCC) and complement dependent cytotoxicity (CDC).18 Both are associated with cell lysis, with potential adverse effects that may be seen in prolongued38 treatments. Therefore, the modified fragment of IgG1 appears to be active, thereby preventing adverse events resulting from the ADCC.39
3. Anti-inflammatory effect of abatacept. Abatacept significantly reduces many of the inflammatory mediators in patients with RA, restoring them to normal, a fact demonstrated in several clinical trials used when researching the drug.
In a Phase II-b, 1-year, placebo-controlled study in patients with RA and inadequate response to MTX, samples were taken and serum levels of selected markers were measured in the days before the infusion in order to study the effect of abatacept on mediators and proinflammatory cytokines. A group of patients received MTX and abatacept 10mg/kg, according to regular schedule. The control group, meanwhile, was treated with MTX and placebo. One year after treatment, markers in the abatacept group of 10mg/kg had normalized, while remained elevated in the placebo group (TNF: 7.4 vs 10.3pg/ml; FR: 159 vs 225U/l, sIL-2R: vs 1228.3. 1697.1pg/ml IL-6: 7.3 vs 19.9pg/ml).40
4. Immunogenicity. According to data on the drug, only 187 of 3877 (4.8%) patients with RA treated for up to 8 years with abatacept developed antibodies to the drug during the treatment.41 Antibodies to abatacept were evaluated in patients after stopping the drug (>42 days after the last dose), and in 103 of 1888 (5.5%) were seropositive. By contrast, in another study of 2000 patients abatacept, antibodies were measured and it was concluded that abatacept has low immunogenicity.42,43
5. Abatacept and tuberculosis. TNF participates in the inflammatory response and immunopathology of tuberculosis (TB). In vitro studies show that TNF increases the phagocytic activity and mycobactericidal macrophage, whereas in vivo were involved in the initial formation and the subsequent maintenance of granulomas, something that controls the growth of mycobacteria and limits its spread. In a chronic model of reactivation of latent TB in mice, we studied the evolution of the infection in mice treated with abatacept compared to another group treated with a murine monoclonal anti-TNF.42 4 months after infecting C57BL/6 mice with Mycobacterium tuberculosis and, once confirmed they had a latent TB infection, the mice were treated for 16 weeks with one of two experimental interventions. After this time, all the mice treated with anti-TNF died from disseminated TB with a mean survival of 44 days. On the contrary, none of the mice treated with abatacept died.
While the concentration of serum IFN-g did not change in the abatacept group, it was elevated in mice with anti-TNF. This increase was attributed to the increased infiltration of CD4+ and CD8+ caused by the widespread dispersal of the bacterial colonies.
So, while mice treated with anti-TNF therapy showed 100% mortality, abatacept did not alter the ability of mice to organize an inflammatory response capable of controlling the spread of tuberculosis. However, there still is not enough clinical data to confirm these findings in humans.
6. Antiresorptive effect of abatacept on bone remodeling. Osteoclast activity is increased in RA, both in the joint, causing bone erosions, as well as systemically, reaching levels associated with generalized osteoporosis.44,45
In fact, an increase of the ligand receptor activator of nuclear factor NF-kB (RANKL) has been shown in the synovial membrane.45,46 Abatacept dose-dependently inhibits murine osteoclast formation and the osteoclastogenic activity assessed in vitro. This was studied in murine osteoclasts cultured on dentin plates, which measured the number of resorption pits after 6 days of adding different doses of abatacept.47
The drug significantly decreased the area of bone resorption. This data suggest that abatacept is a molecule that binds directly the osteoclast precursor cells, inhibiting their differentiation. This mechanism could explain the anti-erosive effect of the drug in patients with RA. In fact, patients treated with abatacept showed a decreasing trend in RANK and its ligand RANKL levels in the synovium, all associated with increased osteoprotegerin.48 Although the exact mechanism underlying this observation is unclear, these findings correlate well with radiological improvement observed in patients treated with abatacept.
7. Effects of abatacept in other immune cells. Although the APC is the target cell which binds abatacept and macrophages also express CD80/86 receptors on their surface, there are few studies investigating the action of the drug on the activity of these cells. Indeed, a recent in vitro study has shown that macrophages exhibited marked CD80/86 receptor expression and treatment with abatacept reduced cytokine49 production substantially. These results suggest that the mechanism of drug action could be extended to the regulation of macrophage lineage, key cells in the pathogenesis of the disease.
Abatacept also suppresses follicular migration of antigen-specific T cells and, consequently, the collaboration between T cells and follicular B cells in the lymph node. This finding has been observed in situ in lymph nodes of BALB/c50 mice. After transfusing such mice with antigen specific prestimulated T cells, a subsequent immunization of mice showed T cell proliferation and migration to the B lymphocyte area. In mice treated with abatacept, T cell proliferation and migration was blocked, limiting its presence in most cases in the lymph node paracortex. Thus, prolonged treatment with abatacept reduces proliferation, mobility and distribution of intraganglionar autoantigen memory lymphocytes, which could lead to the decrease of autoantibodies.
Conclusions on the Mechanism of Action of AbataceptAbatacept is a fully humanized protein construct, consisting of the extracellular domain of human cytotoxic T lymphocyte-associated antigen 4 (CTL4) and a genetically modified fragment of the Fc region of IgG1, designed to interfere with the regulation of costimulation of T lymphocytes The drug inhibits the activation of T cells by selectively blocking the specific binding of CD80/CD86 to the CD28 receptor and, therefore, inhibiting T cell proliferation and immune responses of B lymphocytes This pharmacological action results in decreased levels of inflammatory mediators in patients with RA and in a safe and effective clinical response.
Conflict of InterestDr. Gabriel Herrero-Beaumont has received research grants from Bristol-Myers-Squibb. Dr. Santos Castañeda has received education and research grants form Abbott, MSD and Pfizer.
Herrero-Beaumont G, et al. Mecanismo de acción de abatacept: concordancia con su perfil clínico. Reumatol Clin. 2012; 8(2): 78–83.