Cat scratch disease (CSD) is an infectious disorder caused by Bartonella henselae and characterized by fever and granulomatous lymphadenopathy. Immunosuppression is a risk factor for the development of atypical forms of the disease. We report the case of a 52-year-old woman who presented with fever and bilateral inguinal lymph node enlargement. She did not have apparent contact with animals. The patient was receiving etanercept therapy for rheumatoid arthritis. Lymph node biopsy demonstrated granulomatous lymphadenitis. She was successfully managed by discontinuing etanercept and by treatment with minocycline. She developed clinical remission and typical seroconversion. Infection with Bartonella should be considered in the differential diagnosis in rheumatoid arthritis patients with lymphadenopathy of unknown origin.
La enfermedad por arañazo de gato (EAG) es una enfermedad infecciosa causada por la proteobacteria Bartonella henselae, caracterizada por fiebre y linfadenopatía granulomatosa. La inmunosupresión es un factor de riesgo para el desarrollo de formas atípicas de la enfermedad. Presentamos el caso de una mujer de 52 años de edad que presentó fiebre y adenomegalias inguinales bilaterales. No tenía contacto aparente con animales. La paciente estaba recibiendo tratamiento con etanercept por artritis reumatoidea. La biopsia del ganglio linfático informó de una adenopatía granulomatosa. Evolucionó favorablemente con la interrupción de etanercept y el tratamiento con minociclina. Presentó remisión clínica y la seroconversión típica. La infección por Bartonella debería ser considerada como un diagnóstico diferencial en pacientes con artritis reumatoidea, con linfadenopatía de origen desconocido.
The proteobacterium Bartonella henselae (BH) is a small aerobic Gram-negative bacillus, that is highly adaptable to several mammals as reservoirs. In humans it produces cat-scratch disease (CSD), characterized by self-limiting fever and granulomatous lymphadenopathy. In 5%–15% of cases, it can lead to severe forms with ocular, glandular, neurological, pulmonary and/or osteoarticular involvement or thrombocytopenic purpura.1,2
The utilization of biological agents in individuals with rheumatic diseases, frequently in contact with household pets, increases concern about the development of opportunistic diseases, especially with atypical presentation.3–9
Case ReportThe patient was a 52-year-old woman with a 29-year history of rheumatoid arthritis (RA). She had received treatment with methotrexate, leflunomide and meprednisone (MP). At the age of 45 years, she was diagnosed with “diffuse sclerosing glomerulopathy and amyloidosis”. Thus, treatment was begun with etanercept at a dose of 50mg/week, and her clinical signs improved.
Thirty days before visiting her physician, she developed a febrile syndrome and self-limiting diarrhea (one week). She had no apparent contact with animals. At admission, she was subfebrile, weak, adynamic, anorexic and had myalgia and arthralgia. Physical examination revealed bilateral painful inguinal lymphadenopathy, with no other relevant signs. An echocardiogram and respiratory function tests were normal. Chest and abdominal computed tomography demonstrated only inguinal lymph node enlargement.
Laboratory tests revealed hematocrit at 33%, hemoglobin: at10.8g/dL, white blood cells at 8500/mm3 (neutrophils, 72%, lymphocytes, 19%, monocytes, 6%, eosinophils, 2% and basophils, 1%). Erythrocyte sedimentation rate: 110mm/h; C-reactive protein: 10.2mg/dL; glucose; 93mg/dL; urea; 62mg/dL; creatinine; 2.4mg/dL; ferritin: 232ng/mL (normal range: 12–150ng/mL). The hepatic profile was normal, β2 microglobulin was 12.6mg/dL (normal range: 0.8–2.2mg/dL); immunoglobulin (Ig) A: 668mg/dL; IgG: 1680mg/dL and IgM: 174mg/dL.
Tumor markers (cancer antigen [CA] 15-3, CA 19-9, CA-125 and carcinoembryonic antigen [CEA]) were negative. Virus serology (human immunodeficiency virus and hepatitis B and C viruses and syphilis (the Venereal Disease Research Laboratory test) were negative. There was no evidence of acute infection by cytomegalovirus, Epstein–Barr or toxoplasmosis. Blood and urine cultures were also negative.
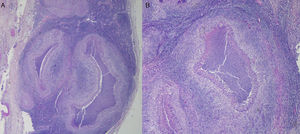
The biopsy of a lymph node revealed a histological picture of “granulomatous lymphadenitis”, which suggested a diagnosis of CSD (Fig. 1). Periodic acid Schiff (PAS), Grocott and Ziehl–Neelsen stains were negative. Immunohistochemistry (cluster of differentiation [CD] 3, CD20) showed residual lymphocytes. Flow cytometry revealed no evidence of a lymphoproliferative process. Tests and cultures for common microorganisms, mycobacteria and fungi were negative. We did not detect Chlamydia trachomatis, atypical mycobacteria or Mycobacterium tuberculosis in the methods based on polymerase chain reaction.
Serological tests for BH showed that the patient was positive for IgM with a titer of 1:32 (cutoff value: 1:16) and for IgG with a titer of 1:160 (cutoff value: 1:10). With the diagnosis of CSD, treatment was begun with minocycline at a dose of 100mg/12h/12 weeks, which achieved the remission of the symptoms and of lymphadenopathy. Once treatment had been completed, IgM was negative and the IgG titer had increased to 1:320. Treatment with etanercept was reinitiated to control RA, without unforeseen eventualities.
DiscussionCat-scratch disease especially affects children and young adults, only rarely involving elderly individuals, and is associated with contact with animals (which did not occur in our patient). We report, to the best of our knowledge, the first case of CSD in a patient with RA being treated with etanercept.
The diagnosis of CSD is based on a combination of clinical, epidemiological (contact), bacteriological, serological and histological findings. There is no diagnostic criteria considered as a reference pattern.10,11
In the immunocompetent host, it usually presents as subacute lymphadenopathy accompanied by fever and mild involvement of general health, although there can be atypical manifestations such as fever of unknown origin, hepatosplenic granulomas, neuroretinitis, seizures, osteomyelitis, etc.1,2,10,11
In immunocompromised patients, there are reports of bacteremia, endocarditis, bacillary angiomatosis and peliosis hepatis.1–11 Our patient presented with a prolonged febrile syndrome and inguinal lymphadenopathy.
The primary lesion developed near the site of inoculation, 3–10 days after the scratch or bite. The immune response includes an increase in the response of T helper cells mediated by interferon-γ and macrophage recruitment and stimulation, with the formation of granulomas. During infection, the infected macrophages produce high levels of tumor necrosis factor-α (TNF-α). Thus the use of TNF-α inhibitors interferes with the inflammatory response, favoring the dissemination and bacteremia of BH.2,11
The lack of exposure to cats can be explained by the fact that other animals (arthropods) can transmit the bacteria. It is known that many patients with CSD deny having had any contact with cats.12,13
A review of the literature on CSD in patients who received biological agents (Table 1) demonstrated that lymphadenopathy is reported in all the articles, but only in three of eight patients was the lesion or entry site identified. Fever, present in the patients taking etanercept, did not occur in every case.3–9
Characteristics of Our Patient and Comparison With Cases Retrieved From the Literature Review.
| Case report | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sex | Age (y) | Disease | Drug | Source | Lymphadenopathy | Fever | Diagnosis | Treatment |
| W | 52 | RA | Etanercept | ? | Inguinal | Yes | IgM+, IgG+, Path | Minocycline |
| Literature review | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sex | Age (y) | Disease | Drug | Source | Lymphadenopathy | Fever | Diagnosis | Treatment | Ref. |
| M | 51 | PsA | Etanercept | L | Mesenteric. Splenic abscesses. Panniculitis | Yes | IgG+ | Clarithromycin | 3 |
| M | 50 | AS | Etanercept | L | Cervical, submaxillary | Yes | IgG+, Path: PCR+ | Doxycycline Roxithromycin | 4 |
| W | 74 | Crohn's | Infliximab | C | Mesenteric, pericelíac, mediastinal Panniculitis | Yes | IgG+ | Tigecycline | 5 |
| W | 23 | AS | Infliximab | L | Inguinal. Subcutaneous nodes in arm | No | Path: W–S+ | Moxifloxacin Amikacin | 6 |
| M | 36 | RA | Tocilizumab | C | Axillary and mediastinal Subcutaneous nodes in arm | No | IgG+, Path: PCR+ | Doxycycline Gentamicin | 7 |
| M | 29 | sero−RA | Tocilizumab | C | Epitrochlear and axillary | No | IgG+, Path: W–S+ | Azithromycin | 8 |
| M | 36 | Ps | Adalimumab | C | Cervical | No | AP: PCR+ | Doxycycline | 9 |
AS, ankylosing spondylitis; C, contact; L, lesion; M, man; Path, pathology; PCR, polymerase chain reaction; Ps, psoriasis; PsA, psoriatic arthritis; RA, rheumatoid arthritis; Ref., reference; sero-RA, seronegative rheumatoid arthritis; W, woman; W–S, Warthin–Starry stain;?, unknown.
The microbiological diagnosis of BH is mostly based on the detection of antibodies by indirect immunofluorescence (IIF) or enzyme-linked immunosorbent assay (ELISA). The IgG titers in the general population vary with the risk of exposure. A titer of IgG ≥1:64 is considered to be indicative of the diagnosis (sensitivity and specificity >90%). The duration of IgM is limited (3 months).14
Isolation in culture is very difficult (it requires 2–6 weeks) and, in general, is negative. This must be taken into account, especially in cases of fever of unknown origin, neuroretinitis, encephalitis, peliosis or bacillary angiomatosis and in endocarditis with negative cultures. Molecular tests in blood or tissues can be valuable.1,10–14
Histopathology, which is helpful for the differential diagnosis, serves as a guide but is not pathognomonic, and reveals necrotizing granulomas, microabscesses and follicular hyperplasia. Warthin–Starry stain only demonstrates the presence of bacilli in lymph nodes that contain suppurative granulomas. Thus, although negative, it does not rule out the diagnosis.15 The differential diagnosis of granulomatous lymphadenitis includes an extensive spectrum of infectious causes (bacterial agents—among them BH, chlamydias, fungi, mycobacteria, parasites, spirochetes and rickettsias) and noninfectious causes (lymphatic or secondary neoplasms, vasculitis, Crohn's disease, celiac disease, sarcoidosis, Kikuchi–Fujimoto disease, berylliosis, etc.).16
Treatment should primarily take into consideration the clinical manifestations and the immunological status of the patient. The recommendations are based on case reports and expert opinion, since there are few clinical trials.2,11,17,18
In immunocompetent individuals, the disease is generally self-limiting and may not require antibiotics. In a clinical trial with azithromycin, the use of the drug had a slight effect on the reduction of lymph node volume in a shorter period of time.
In the case of intolerance, other possibilities are clarithromycin, rifampicin, trimethoprim-sulfamethoxazole and ciprofloxacin. In immunocompromised patients and in atypical presentations, antibiotic therapy is always suggested. The recommended combinations in hepatosplenic or disseminated disease include rifampicin plus azithromycin or gentamicin and, in neurological disease or neuroretinitis, the use of doxycycline plus rifampicin is proposed.2,11,17,18
In patients taking biological agents the drugs most widely employed were tetracyclines and macrolides. The course in every case was favorable, with variable periods of remission, that were not immediate in the majority, but there were no relapses. Biological therapies could be reinitiated. as in our case, once the disease was under control.3–9
In short, we present the case of a woman with RA who was being treated with etanercept, with a clinical, serological and pathological diagnosis of CSD. Her response was favorable to the therapeutic approach. The increase in the use of biological agents and the number of families with pets should call attention to this condition, even in the absence of an evident lesion or contact with animals.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of InterestThe authors declare they have no conflicts of interest.
Please cite this article as: Orden AO, Nardi NN, Vilaseca AB, Colombini AC, Barrios NG, Vijnovich Barón A. Enfermedad por arañazo de gato en paciente con artritis reumatoidea tratado con etanercept. Reumatol Clin. 2018;14:303–306.