New trends in osteoarthritis research focus on the use of biological therapy; in this context, the use of Plasminogen Activator Inhibitor-1 (PAI-1) is considered a potential therapeutic strategy to prevent extracellular matrix (ECM) degradation in osteoarthritis (OA) management. However, in vitro studies have not demonstrated its effect on the expression of ECM homeostasis-related genes.
MethodsHuman OA cartilage-derived chondrocytes were used to generate scaffold-free spheroids under hypoxia conditions. The spheroids were exposed to PAI-1 for 24h, and cell viability was measured. Then qRT-PCR was used to analyze the expression of ECM components and degradative enzymes, including COL2A1, SOX9, ACAN, COL1A1, MMP3, MMP9, MMP13, ADAMTS4, ADAMTS5, TIMP1, TIMP2, TIMP3, uPA and tPA.
ResultsPAI-1 treatment consistently maintained cell viability and chondrocyte spheroid integrity. At the 50ng/mL concentration, PAI-1 increased the gene expression of COL2A1 and reduced SOX9, ACAN, MMP3, MMP9, TIMP2, and tPA. Moreover, the functional COL2A1/COL1A1 ratio was significantly increased in PAI-1-treated spheroids.
ConclusionOur results suggest that PAI-1 treatment exerts a complex and multifaceted influence on spheroids’ ECM. While it supports matrix integrity by reducing the gene expression of ECM remodeling enzymes, such as MMPs and ADAMTS5, it also induces unfavorable changes in chondrogenesis-related marker genes, such as SOX9 and ACAN. These findings indicate that the cellular response to PAI-1 is not unidirectional, warranting further investigation to understand its precise biological implications.
Las nuevas tendencias en la investigación de la osteoartritis (OA) se centran en el uso de terapias biológicas; en este contexto, el uso del inhibidor del activador del plasminógeno-1 (PAI-1) se considera una estrategia terapéutica potencial para prevenir la degradación de la matriz extracelular (ECM) en el manejo de la OA. Sin embargo, los estudios in vitro no han demostrado su efecto en la expresión de genes relacionados con la homeostasis de la ECM.
MétodosSe utilizaron condrocitos derivados de cartílago humano con OA para generar esferoides sin andamios bajo condiciones de hipoxia. Los esferoides fueron expuestos a PAI-1 durante 24h y se midió la viabilidad celular. Posteriormente, se utilizó qRT-PCR para analizar la expresión de componentes de la ECM y enzimas de degradación, incluidos COL2A1, SOX9, ACAN, COL1A1, MMP3, MMP9, MMP13, ADAMTS4, ADAMTS5, TIMP1, TIMP2, TIMP3, uPA y tPA.
ResultadosEl tratamiento con PAI-1 mantuvo consistentemente la viabilidad celular y la integridad del esferoide de condrocitos. A una concentración de 50ng/ml, el PAI-1 aumentó la expresión génica de COL2A1 y redujo la expresión de SOX9, ACAN, MMP3, MMP9, TIMP2 y tPA. Además, la relación funcional COL2A1/COL1A1 aumentó significativamente en los esferoides tratados con PAI-1.
ConclusiónNuestros resultados sugieren que el tratamiento con PAI-1 ejerce una influencia compleja y multifacética sobre la ECM de los esferoides. Si bien promueve la integridad de la matriz al reducir la expresión génica de enzimas que remodelan la ECM, como las MMPs y ADAMTS5, también induce cambios desfavorables en marcadores relacionados con la condrogénesis como SOX9 y ACAN. Estos hallazgos indican que la respuesta celular al PAI-1 no es unidireccional, lo que justifica una mayor investigación para entender sus implicaciones biológicas precisas.
Osteoarthritis (OA) is characterized by cartilage damage and synovial inflammation that negatively impacts the quality of life.1–3 Different factors play a pivotal role in OA progression, such as aggrecanases responsible for the cleavage of aggrecan, a key component of cartilage extracellular matrix (ECM).4 Cytokines, conversely, are signaling molecules secreted by immune cells and chondrocytes within the joint, contributing to the inflammatory response and ECM breakdown.5 Matrix metalloproteases (MMPs) are a group of enzymes involved in the degradation of various ECM components, including collagens.6 Understanding the intricate interplay between these factors is essential for developing effective strategies to mitigate OA progression.
In this context, the Plasminogen Activator Inhibitor-1 (PAI-1) inhibits plasmin that activates MMPs in OA. It is primarily expressed in subchondral bone and cartilage.1,7 Moreover, the PAI-1 gene and protein expression is significantly reduced in human osteoarthritic chondrocytes, leading to diverse functional consequences for ECM homeostasis.8,9 This study has focused on observing the influence of PAI-1 within a hypoxia-regulated 3D culture system, emphasizing its potential to enhance cartilage-specific gene expression.
Materials and methodsSourcing of human articular chondrocytesWe obtained hyaline cartilage biopsies from 13 knee surgery patients and signed an informed consent letter approved by the Ethics and Research Committee from the Instituto Nacional de Rehabilitación Luis Guillermo Ibarra Ibarra. Twelve patients underwent total knee arthroplasty due to OA graded III–IV; one had meniscal repair and anterior cruciate ligament (ACL) reconstruction. All procedures followed standard protocols and used the same anesthetic approach.
Primary cell cultureCartilage samples were collected in DMEM-F12 medium supplemented with 10% antibiotic–antimycotic. As previously reported, each piece was subjected to mechano-enzymatic disaggregation to obtain chondrocytes.10 Isolated cells were cultured in DMEM-F12 medium supplemented with 1% antibiotics and 10% Fetal Bovine Serum (FBS) under a controlled atmosphere at 37°C, 5% O2, 5% CO2, and saturation humidity until achieving a confluence of 80%.
Chondrocyte spheroidsThe spheroids were generated using a 4% agarose chip that was negatively printed with a customized silicone mold (Research Micro Stamps, Clemson, SC, USA); each mold was 1.8mm in diameter with circular pillar structures of 250μm high and 250μm diameter.10 The spheroids culture was maintained using DMEM-F12 supplemented with 5% FBS, 10ng/mL TGF-β1, 0.5% insulin-transferrin-selenium 1×, 100μM dexamethasone, 200μM ascorbic acid, and 1% antibiotics. This culture was maintained under hypoxic conditions for 14 days. The chondrocytes from each spheroid came from a single patient.
Viability assayThe safety of PAI-1 supplementation at varying concentrations was validated through cell viability assays on chondrocyte spheroids. We employed the calcein/propidium iodide (PI) test, following the manufacturer's instructions of the LIVE/DEAD Viability/Cytotoxicity Kit (Thermo-Fischer Scientific, Waltham, MA, USA). The images were visualized in an EVOS FL Auto inverted fluorescence microscope (Thermo-Fischer Scientific, Waltham, MA, USA). All determinations were performed in triplicate across three independent events. Quantification of relative fluorescence units (RFUs) for the green channel was performed to assess the calcein signal in chondrocyte spheroids. The calcein fluorescence signal and spheroid area were quantified using the ImageJ software (NIH, Bethesda, MD, USA).
Gene expressionThe protective role of PAI-1 against ECM degradation was determined by assessing gene expression by qRT-PCR of specific ECM constituent markers, including type II collagen (COL2A1), SOX9, aggrecans (ACAN), type I collagen (COL1A1); as well as enzymes associated with ECM degradation such as MMP3, MMP9, and MMP13, A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) 4, ADAMTS5, tissue inhibitors of metalloproteinases (TIMP) 1, TIMP2, TIMP3, and the plasminogen activators uPA and tPA. Commercial primers were obtained from Qiagen, and customized primers were obtained from T4Oligo. References and sequences are shown in Supplementary Tables 1 and 2. The qPCR was carried out in RotorGene Q thermocycler under the following conditions: 95°C – 10min, 40 cycles at 95°C – 15s, 60°C – 40s, using RT2 SYBR Green FAST Mastermix (Qiagen, Germany). Expression was normalized with the endogenous gene RPL27 in each sample. The data was analyzed using the 2−ΔCT method.
Statistical analysisThe normality of the data was determined through the Shapiro–Wilk test. The Student's t-test was used to assess the effect of PAI-1 between the groups with normal distribution. The Mann–Whitney U test was performed for data with non-normal distribution. One-way ANOVA was used for comparison analysis of spheroid viability treated with PAI-1. A p-value of <0.05 was considered significant. Data are presented as median with interquartile range (IQR) if not otherwise stated. Statistical analysis was conducted using the STATA software package v.16 (StataCorp Texas, USA), and graphs were generated in GraphPad Prism version 9.0.1.
ResultsAnthropometric features of cartilage donorsThe median age of cartilage donors was 62 years (IQR=61–66). Consistent with extant literature, the gender distribution of our sample supports the pivotal role of sex in the OA pathogenesis, with females comprising 84.62% of the donors. Further, the calculated mean for body mass index was registered at 29.34±3.8kg/m2, revealing a predominance of overweight status among the participants.
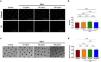
PAI-1 effect on spheroid viabilityNo alteration in cell viability of spheroids treated with different concentrations of PAI-1 was observed after 24h of incubation (Fig. 1a). Our analysis utilized ANOVA and Tukey's post hoc test to evaluate the differences among the RFU of the control (20.1 RFU±1.53) and tested concentrations of PAI-1 corresponding to 10ng/mL, 50ng/mL, and 100ng/mL (20.6 RFU±3.56, 21.6 RFU±2.34, 20.6 RFU±3.27, respectively). The results revealed no statistically significant differences among the groups (p=0.548) (Fig. 1b). Furthermore, it was noted that the spheroid three-dimensional form was maintained (Fig. 1c, d).
Viability and integrity assay of spheroids treated with PAI-1. Spheroids were incubated with varying concentrations of PAI-1 and stained with 5ng/mL of Calcein and 20ng/mL of PI (a). Relative fluorescence units (RFUs) were quantified to assess spheroid viability (b). Spheroid integrity was analyzed, and surface area was measured in mm2 (c, d). The scale bar represents 1000μm. p-Values were obtained from one-way ANOVA and Tukey's post hoc test.
Fig. 2 shows the relative expression units (REU) of uPA, tPA, COL1A1, MMPs, ADAMTs, TIMPs, COL2A1, SOX9, and ACAN in spheroids treated at 50ng/mL PAI-1. Regarding genes responsible for maintaining ECM integrity, an increased expression of COL2A1 was observed in PAI-1-treated spheroids versus control (p=0.016). Although there was an increase in COL1A1 expression in the PAI-1 group, it was not statistically significant (p=0.244). Of particular significance, we observed that the COL2A1/COL1A1 ratio was higher in spheroids supplemented with PAI-1 in comparison to the control group (p=0.005).
Gene expression of chondrogenic markers and enzymes associated with extracellular matrix degradation: SOX9, ACAN, COL2A1, COL1A1, MMPs, ADAMTs, uPA, tPA, and TIMPs. p-Values obtained from the Mann–Whitney U test. The blue circles and triangles represent the outliers for the control and PAI-1 groups, respectively. The gene expression assay was performed using all patients included in the study (n=13).
Nevertheless, PAI-1 exerted a downregulation of ACAN, registering a median of 0.39 REU; meanwhile, the control group had a median of 2.46 REU. We also observed a similar trend for SOX9 expression, with a significant difference (p=0.022) between the PAI-1 and control groups.
The PAI-1 supplementation significantly decreased the expression of MMP3 and MMP9, ECM remodeling enzymes. A significant decline in MMP3 expression was observed in the PAI-1 group versus control (p<0.001). Furthermore, MMP9 expression was down-regulated in spheroids treated with PAI-1 (p=0.001). Although MMP13 tended to decrease in the PAI-1 group, it was not statistically significant. On the other hand, aggrecanases were also modulated by PAI-1 supplementation. A substantial decrease in ADAMTS5 expression was observed compared to the control group, with a p-value of 0.022. Nevertheless, PAI-1 did not induce changes in ADAMTS4 expression. Regarding TIMP1, TIMP2, and TIMP3, PAI-1 treatment tended to decrease their expression. However, we found a statistically significant difference for only TIMP2 comparing control and PAI-1 groups (p=0.022).
The uPA expression showed a slight difference between the control and PAI-1 groups; nevertheless, it was not statistically significant. On the other hand, PAI-1 induced a considerable reduction of tPA gene expression (p=0.016).
DiscussionOA is a prevalent multifactorial disease that affects healthcare costs and quality of life in older adults.11–14 This impact has spurred extensive ongoing research to understand its underlying mechanisms and explore innovative treatment approaches.
Insights from this research have led to a paradigm shift toward 3D culture conditions that allow chondrocytes to form an ECM, vital for facilitating cell–cell interaction and stimulating the expression of major structural components in cartilage. In this sense, it has been reported an enhancement of proteoglycans such as aggrecans in spheroids cultured in hypoxia, as well as SOX9, which is essential for cartilage development.10,15–17 Moreover, the COL2A1/COL1A1 ratio is used to indicate chondrocyte differentiation. In this sense, the increase of the COL2A1/COL1A1 ratio in spheroids supplemented with PAI-1 reflected its effect on modulating chondrocyte differentiation. Our in vitro model accurately replicates the native environment, thus providing a more realistic platform for studying OA pathogenesis and testing potential treatments.
Our data showed a decreased tPA gene expression in the PAI-1 group compared to the control in our 3D culture. This is significant because tPA is critical in activating the plasminogen-plasmin-MMP pathway, leading to cartilage degradation. A decreased tPA gene expression induced by PAI-1 could be implicated in an effective blocking of the tPA pathway's activation, protecting the ECM from degradation. Concurrently, reduced expression of ADAMTS4 and 5, enzymes known to degrade proteoglycans, in the PAI-1 group suggests a slowdown in cartilage degradation, further implying the protective role of PAI-1.
PAI-1 also appeared to inhibit potential inflammatory activity, as evidenced by decreased gene expression of MMP3 and MMP9 in chondrocytes, suggesting a reduced cartilage collagen breakdown by MMPs. Conversely, the PAI-1 group exhibited lower expression of TIMPs than the control group. Elevated TIMP1 levels have been previously associated with advanced stages of OA, underscoring its potential use as a diagnostic marker.18
A prior study by Moritake et al.19 highlighted the pivotal role of PAI-1 in cartilage matrix degradation and impaired chondrogenesis in murine chondrocytes, particularly under IL-1β stimulation, by regulating MMP expression and type II collagen levels. PAI-1 deficiency significantly reduced the MMP-3, -9, and -13 mRNA levels and MMP-13 activity, otherwise enhanced by IL-1β. Conversely, active PAI-1 treatment increased MMP expression, underscoring its crucial role in upregulating MMPs in response to inflammatory stimuli. These findings align with our results, demonstrating that PAI-1 modulates ECM dynamics through its dual effects on ECM-degrading proteinases and collagen synthesis. Furthermore, PAI-1 deficiency reversed the suppression of type II collagen mRNA levels induced by IL-1β, further implicating PAI-1 in cartilage matrix degradation via impaired collagen synthesis. IL-1β, a key proinflammatory cytokine in OA, exacerbates cartilage degradation by upregulating MMP-13, aggrecanases, and plasminogen activators.20,21 This mechanism aligns with the overexpression of matrix-degrading enzymes and proinflammatory cytokines that hallmark OA progression.22 Importantly, our findings revealed that PAI-1 treatment did not compromise chondrocyte viability and integrity, as demonstrated by consistent calcein uptake and negligible propidium iodide signal in 3D spheroids cultured for 14 days, even at varied PAI-1 concentrations. These results confirm the safety of PAI-1 and emphasize its complex role in balancing ECM protection and degradation in OA pathogenesis. While the regulatory role of PAI-1 in cartilage homeostasis is evident, the multifactorial nature of OA suggests the involvement of other cytokines and pathways in disease progression. Understanding these interactions is critical to developing comprehensive therapeutic strategies targeting PAI-1 and related pathways for effective OA management.
Adding PAI-1 to the 3D culture of human osteoarthritic chondrocytes displayed low expression of genes essential for chondrocyte development, such as SOX9 and ACAN. A feedback mechanism might explain this effect, as ECM-related gene expression often undergoes dynamic regulation to maintain the balance between matrix production and degradation. However, our results do not provide direct evidence to confirm this hypothesis. Instead, we demonstrated that PAI-1 exerts variable effects on the cartilage matrix, including protective roles, by inhibiting ECM-degrading enzymes and potentially harmful ones such as reduced SOX9 and ACAN expression. These findings highlight the complexity of PAI-1's role in ECM regulation and emphasize the need for further studies to determine whether it could have therapeutic potential in cartilage homeostasis.
ConclusionOur results suggest that PAI-1 treatment exerts a complex and multifaceted influence on spheroids’ ECM. While it supports matrix integrity by reducing the gene expression of ECM remodeling enzymes, such as MMPs and ADAMTS5, it also induces unfavorable changes in chondrogenesis-related marker genes like SOX9 and ACAN. The increase of the COL2A1/COL1A1 ratio in PAI-1-treated spheroids demonstrates its effect on modulating chondrocyte differentiation. These findings indicate that the cellular response to PAI-1 is not unidirectional, warranting further investigation to unravel its precise biological implications.
Authors’ contributionSubstantial intellectual contributions to study conception and design (ALR, CMA, CSA), acquisition of data (CSA, CMA, OGM, MJHR), or analysis and interpretation of data (CSA, LEMG, GAMN, ALR); drafting the article or revising it critically for important intellectual content (CSA, CMA, ALR, CPV); final approval of the version to be published (ALR, CPV, GAMN); and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved (ALR, CMA, LEMG). All authors have reviewed and approved the final version of the manuscript.
Ethics approvalThis research complied with the Declaration of Helsinki. This study was approved by the Instituto Nacional de Rehabilitación Luis Guillermo Ibarra Ibarra Ethics and Research Committee (39/20).
FundingThis research was funded by Secretaría de Educación, Ciencia, Tecnología e Innovación de la Ciudad de México, grant number SECTEI/229/2021.
Conflicts of interestThe authors declare no conflict of interest.
Data availabilityThe datasets analyzed during the present study are available from the corresponding author upon reasonable request.
We sincerely thank Maria de Jesus Hernandez Rocha for her invaluable contribution to this research. Her assistance and dedication significantly enriched our study, and we greatly appreciate her efforts and expertise.