Chronic widespread pain represents one of the cornerstones in the definition of fibromyalgia. Pain severity can be measured through different instruments, among which the Graded Chronic Pain Scale represents an outstanding framework to assess pain. Its revised version (GCPS-R) has been recently created to adhere to the new paradigmatic definition of chronic pain. Despite the relevance and clinical impact of the GCPS-R, its validation into Spanish has not been performed yet.
ObjectivesTo develop a cross-cultural Spanish-language adaptation of the GCPS-R in a sample of patients diagnosed with fibromyalgia. Besides, we aimed to carry out an initial psychometric analysis of the questionnaire in this population.
MethodsAn observational, prospective, longitudinal study was conducted among a sample of subjects with fibromyalgia. The translation and cross-cultural adaptation of the GCPS-R was performed, and the new version of the instrument was administered to patients with fibromyalgia. Construct validity was assessed by means of factor analysis, whilst internal consistency, convergent validity, and test–retest reliability were also performed.
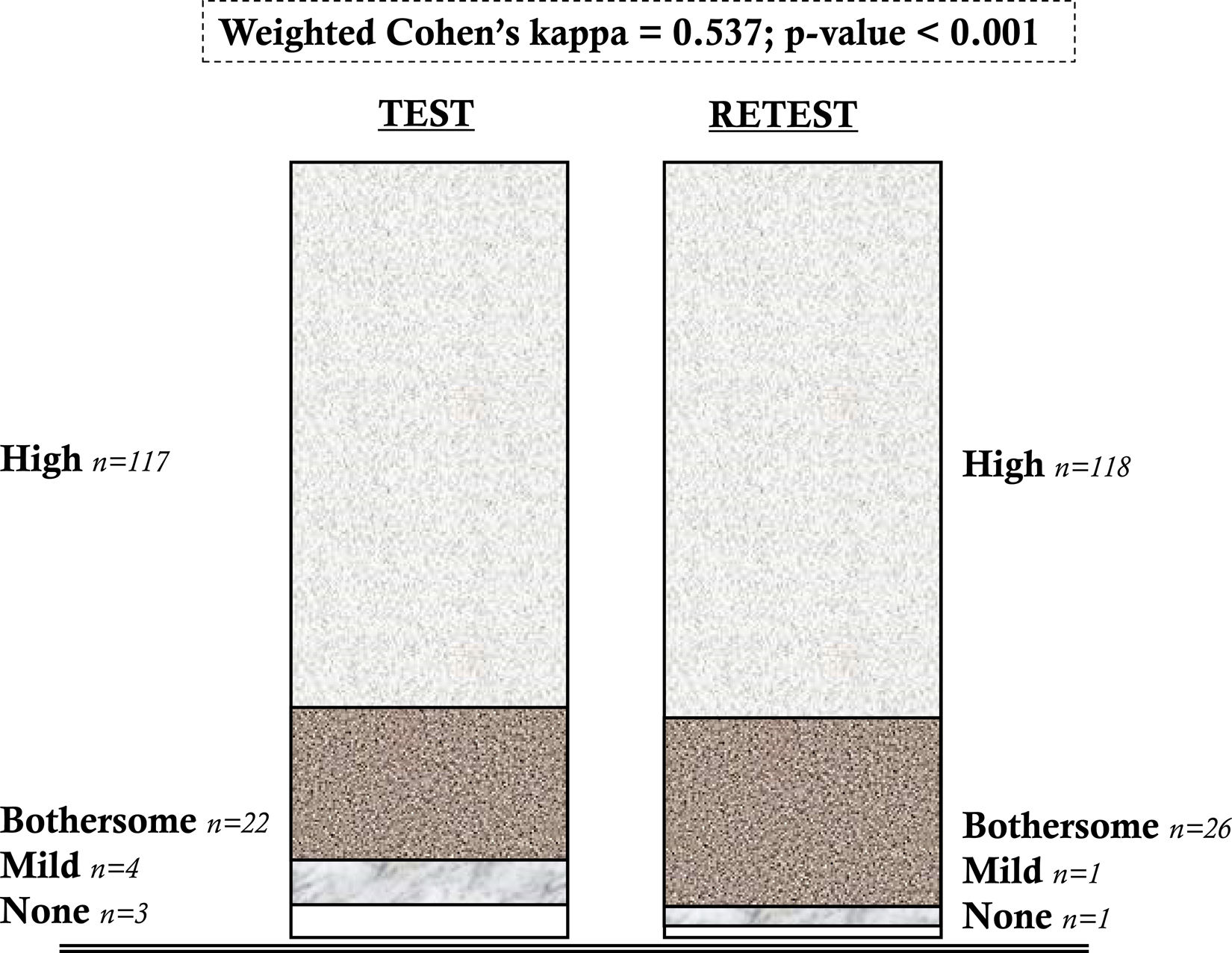
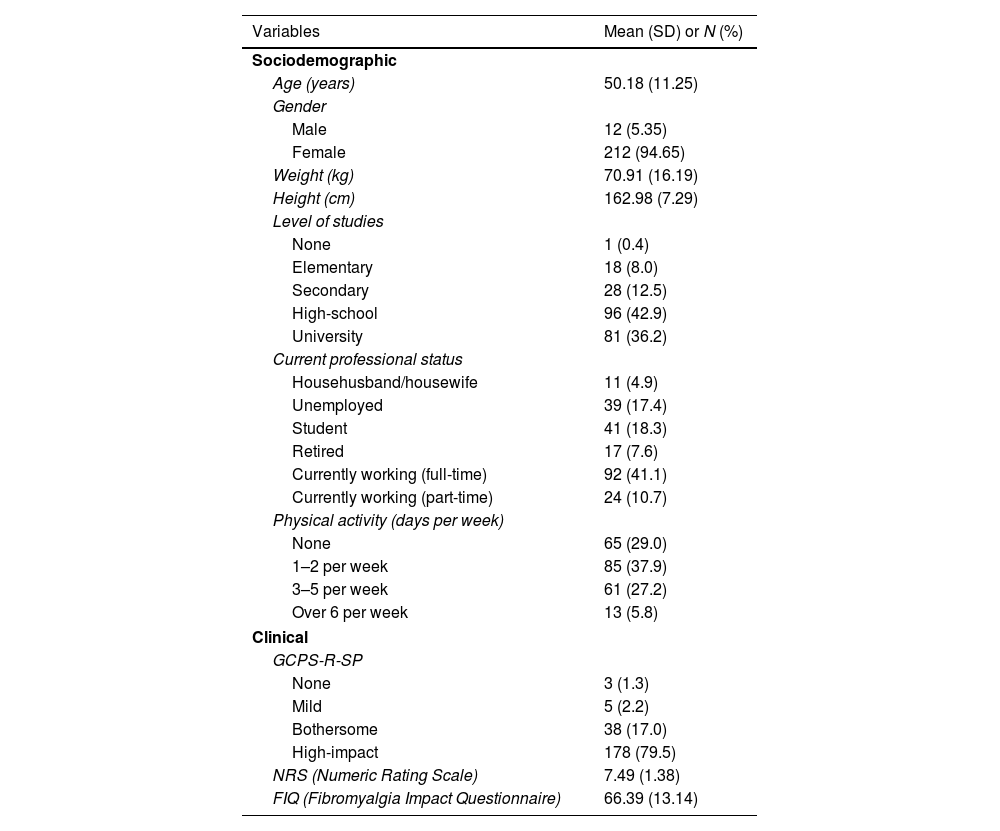
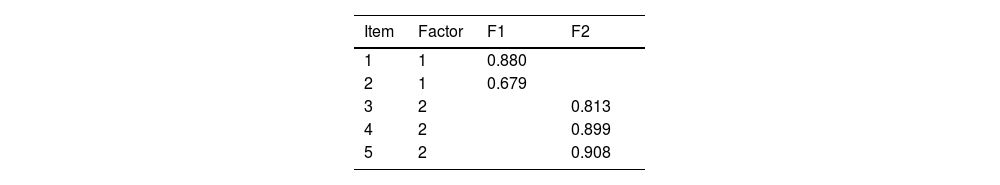
ResultsThe sample analyzed consisted of 224 subjects overall. Factor 1 displayed a Cronbach's alpha of 0.711, whilst Factor 2 had an alpha value of 0.890. The convergent validity analysis performed on the pain-intensity subscale of the instrument yielded statistically significant and strong correlation coefficients (Pearson's r=0.713; p-value<0.001). Test–retest reliability yielded weighted Cohen's Kappa scores of 0.537 (p-value<0.001).
ConclusionThe GCPS-R-SP represents a simple, easy to administrate, and clinically efficient measure with favorable psychometric properties, covering the level and the impact of chronic pain in subjects with fibromyalgia.
El dolor crónico generalizado representa una de las piedras angulares en la definición de fibromialgia. La gravedad del dolor se puede medir mediante diferentes instrumentos, entre los cuales la Escala Graduada de Dolor Crónico representa un marco sobresaliente para evaluar el dolor. Su versión revisada (GCPS-R) ha sido creada recientemente para adherirse a la nueva definición paradigmática de dolor crónico. A pesar de la relevancia e impacto clínico del GCPS-R, aún no se ha realizado su validación al español.
ObjetivosDesarrollar una adaptación transcultural al idioma español del GCPS-R en una muestra de pacientes diagnosticados con fibromialgia. Además, nos propusimos realizar un análisis psicométrico inicial del cuestionario en esta población.
MétodosSe realizó un estudio observacional, prospectivo y longitudinal entre una muestra de sujetos con fibromialgia. Se realizó la traducción y adaptación transcultural del GCPS-R y se administró la nueva versión del instrumento a pacientes con fibromialgia. La validez de constructo se evaluó mediante análisis factorial, además de la consistencia interna, la validez convergente y la fiabilidad test-retest.
ResultadosLa muestra analizada estuvo compuesta por 224 sujetos en total. El factor 1 mostró un alfa de Cronbach de 0,711, mientras que el factor 2 tuvo un valor alfa de 0,890. La validez convergente del instrumento, testada sobre la subescala de intensidad álgica, mostró correlación fuerte y estadísticamente significativa (r Pearson=0.713; p-valor<0.001). La fiabilidad test-retest resultó en puntuaciones Kappa ordinal de Cohen de 0.537 (p-valor<0.001).
ConclusiónEl instrumento GCPS-R-SP representa una medida simple, fácil de administrar y clínicamente eficiente con propiedades psicométricas favorables, que cubre el nivel y el impacto del dolor crónico en sujetos con fibromialgia.