Diagnosis of lupus nephritis (LN) is usually based on renal biopsy, which is an invasive technique that involves multiple risks. Therefore, different biomarkers have emerged as alternatives for the diagnosis of LN. Nonetheless, studies regarding urinary biomarkers in Latin American patients are limited. The objective of this study was to assess the diagnostic value of urinary transferrin and ceruloplasmin to differentiate patients who have renal involvement from those who do not.
Materials and methodsSystemic lupus erythematosus (SLE) patients that met the revised American College of Rheumatology (ACR) classification criteria were recruited. Patients with another autoimmune disease, active infection (urinary tract or systemic infection), renal replacement therapy, human immunodeficiency virus infection or pregnancy were excluded. A urine sample was collected from each patient. LN was diagnosed according to ACR criteria. The activity and chronicity of LN were measured using the Austin indices. Urinary transferrin and ceruloplasmin levels were measured using commercial enzyme-linked immunosorbent assay (ELISA) kits. Mann–Whitney U test and Student's t-test were used to compare data. Spearman's rank correlation was used to determine associations. Lastly, receiver operating characteristic (ROC) curves were created.
ResultsThe study involved 120 SLE patients. In all, 85% were female, 76% mestizo, the mean age was 32.8±12.1years and mean systemic lupus erythematosus disease activity index (SLEDAI) was 8.4±8.9; 64% had renal involvement. Urinary levels of the two biomarkers were significantly higher in patients with LN compared to those without LN. Similarly, urinary levels of both biomarkers were significantly higher in patients with active LN compared to those with inactive LN. Furthermore, urinary transferrin levels were significantly higher in Afro-Latin American patients. On the other hand, urinary transferrin levels correlated with SLEDAI and proteinuria, and transferrin and ceruloplasmin levels correlated with each other. The diagnostic value of ROC curves for these urinary biomarkers for LN were good.
ConclusionsIn our cohort of SLE patients, we found that transferrin and ceruloplasmin were potential biomarkers for LN, and can even differentiate active LN.
El diagnóstico de la nefritis lúpica (NL) se suele hacer con la biopsia renal, que es una técnica invasiva que conlleva múltiples riesgos. Por lo tanto, han surgido diferentes biomarcadores en orina como posibles alternativas para el diagnóstico de la NL. Sin embargo, los estudios de biomarcadores en orina de pacientes latinoamericanos con lupus eritematoso sistémico (LES) son escasos; por lo tanto, el objetivo del presente estudio fue determinar el valor diagnóstico de la transferrina (TF) y la ceruloplasmina (CP) en orina, para diferenciar los pacientes que tienen compromiso renal de aquellos que no.
Materiales y métodosSe incluyeron prospectivamente pacientes con diagnóstico de LES de acuerdo a los criterios del American College of Rheumatology (ACR). Se excluyeron los pacientes con otra enfermedad autoinmune concomitante, infección activa (de vías urinarias o sistémica), terapia de reemplazo renal, infección por virus de la inmunodeficiencia humana y embarazo. A cada paciente se le tomó una muestra de orina. El diagnóstico de NL se realizó mediante los criterios ACR para la definición de NL. La actividad y la cronicidad de la NL en la biopsia renal fueron medidas con el índice de Austin. La determinación de los niveles de TF y CP se realizó con kits comerciales de ELISA. Se utilizó la prueba t de Student y la prueba U de Mann Whitney para comparar los datos. Para determinar las asociaciones entre las variables se utilizaron los coeficientes de correlación de Spearman. Por último, se construyeron curvas ROC.
ResultadosSe incluyeron 120 pacientes con LES, de los cuales el 85% fueron de sexo femenino. El 76% fueron de raza mestiza. Presentaron una edad media de 32,8±12,1años, y una media del SLEDAI de 8,4±8,9, y un 64% presentaron compromiso renal. Los niveles de ambos biomarcadores fueron significativamente mayores en pacientes con NL comparados con aquellos sin NL. De igual manera, los niveles de ambos biomarcadores fueron significativamente mayores en pacientes con NL activa comparados con aquellos con NL inactiva. Los niveles de TF fueron significativamente mayores en pacientes afro-latinoamericanos. Por otro lado, las concentraciones de TF se correlacionaron con el SLEDAI y el rango de proteinuria, y las concentraciones de TF y CP se correlacionaron entre sí. Las curvas ROC para ambos biomarcadores mostraron un buen valor diagnóstico de la NL.
ConclusionesEn nuestra cohorte de pacientes con LES encontramos que la TF y la CP son potenciales biomarcadores para el diagnóstico de la NL e, incluso, de la actividad de la NL.
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease involving multiple organs. Although it can affect both sexes, there is high incidence in females, especially of childbearing age, with a ratio of 9 women for every man suffering the disease.1 The kidney is one of the organs involved, lupus nephritis (LN) has been documented in up to 50% of patients, and varies in severity and aggressiveness according to race, since Hispanic or African American patients have higher rates of terminal chronic renal disease.1,2 The benchmark technique for diagnosing LN is currently renal biopsy, and subsequent histopathological analysis of the sample.3 However, it is an invasive method, which is not without risk. Therefore, in the past decade multiple blood and urinary biomarkers have been investigated as candidates for non-invasive diagnosis of LN, and to differentiate its activity; these include urinary transferrin (TF) and ceruloplasmin (CP).4
TF is a protein that is a member of the transferrin superfamily: on average it weighs 76–81kDa, it can bind to two atoms of ferric iron, and plays a major role regulating multiple processes, because most iron circulates freely to the tissues to supply their needs bound to FR, the most dynamic portion of iron in the organism, and adapts to the needs of the tissues; this is due to its capture mediated by receptors, especially TF1 (TfR1).5 TF is also filtered in the glomerulus and then recaptured by TfR1, which is expressed in the apical membrane of the cells of the proximal, distal and collecting tubule, representing a mechanism by which the tubular cells can obtain the iron necessary for their metabolism.6
CP is a protein of 122kDa that carries most circulating copper; its function is iron oxidase, and this function is directly related to TF, since CP is associated with oxidising ferrous iron to ferric iron so that it can then bind to TF.7 In SLE it has been it has been shown that the levels of molecules associated with iron metabolism, such as ferritin, may be increased, because this can be induced by pro-inflammatory cytokines such as IL-6, TNF-α and IL-1a.8
As mentioned above, diagnostic alternatives to renal biopsy need to be found, due to the risks it involves. Moreover, there are few studies of urinary biomarkers in Latin American patients.9
The possible role of urinary concentrations of TF and CP as biomarkers of LN activity have been documented by Suzuki et al.,10 reporting significantly higher levels of TF and CP in patients with greater renal activity.
For the above reasons, we planned to measure concentrations of TF and CP in urinary samples from a Columbian population with SLE to analyse the correlations between these biomarkers and measures of activity of the disease, and to determine the discriminatory value of these biomarkers in LN.
Materials and methodsPatientsOne hundred and twenty patients were included prospectively, diagnosed with SLE according to the revised American College of Rheumatology (ACR) classification criteria (1082/1997)11 in the San Vicente Fundación University Hospital, between March 2015 and March 2016. Patients with another concomitant autoimmune disease, active infection (urinary tract or systemic), kidney replacement therapy, human immunodeficiency virus and pregnancy were excluded. All the patients gave their informed consent prior to taking the sample, and this research study was approved by the bioethical committee of the San Vicente Fundación University Hospital.
The disease activity was scored with the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI); similarly, renal activity was measured with the renal SLEDAI, the damage caused by the disease was measured using the ACR (SLICC) damage index. LN was diagnosed according to the ACR criteria to define LN. Renal SLEDAI was measured using the following criteria: presence of cylinders in the urinary sediment, haematuria (>5 red blood cells/HPF), pyuria (>5 leukocytes/HPF), and proteinuria >0.5 in 24h of recent onset, or increased proteinuria >0.5g in 24h. Active disease was defined as overall SLEDAI ≥4. Clinically inactive LN was defined by the presence of the following criteria: 24h proteinuria ≤500mg/dl, in active urinary sediment (<5 red blood cells/HPF), no red cell cylinders, no leukocyturia (<5 leukocytes/HPF) and stable serum creatinine.
The information of each renal biopsy was reported using the standard classification of the International Society of Nephrology/Renal Pathology Society (ISM/RPS).12 The biopsy activity index and biopsy chronicity index (BAI and BCI, respectively) were also calculated according the National Institute of Health scores. The activity and chronicity of the LN were measured using the Austin index.13 The items evaluated in the renal biopsy were, for the activity index, endocapillary hypercellularity, leukocytic infiltration, hyaline subendothelial deposits, fibrinoid necrosis/karyorrhexis, epithelial crescents and interstitial inflammation. For the chronicity index they were glomerulosclerosis, fibrous crescents, tubular atrophy and interstitial fibrosis.
SamplesA urinary sample was taken from each patient in the morning. This sample was centrifuged at 1200g for 20min; then filtered with 0.25μm membranes to eliminate cellular detritus, and then were stored at −70°C until they were analysed.
Testing for antibodies and complementThe ANA antibodies were measured using the immunofluorescence technique: double-stranded anti-DNA antibodies by indirect immunofluorescence on the protozoan Arithidia luciliae. Anti-Ro, anti-La, anti-Sm, anti-RNP, anti-C1q and anticardiolipin antibodies were measured by ELISA, with commercial kits.
Complement level testing was carried out using nephelometry techniques.
Determination of proteinuria and creatinine clearanceProteinuria was determined from 24h urine collection using the kinetic alkaline picrate technique. Creatinine clearance was measured with the turbidimetric technique.
Determination of transferrin and ceruloplasmin levelsUrinary TF and CP were measured with commercial ELISA kits (Assaypro, USA), following the manufacturer's instructions. The laboratory staff were blinded to the patients” clinical and histopathological information. Briefly, this procedure consisted of incubating the samples, in dishes sensitised with monoclonal antibody against TF and human CP, then incubated with a biotinylated secondary antibody and an antibody conjugated with streptavidin peroxidase. Finally, the chromogenic substrate was added, the reaction with hydrochloric acid was stopped and read at a wavelength of 450nm with correction of 570nm. Initially a test was carried out on 5 samples in duplicate at different dilutions (1:4 [suggested by the manufacturer of the kit], 1:20 and 1:100 for TF; 1:16 [suggested by the manufacturer], 1:50, 1:100, 1:150 and 1:200 for CP). The TF determinations were made based on these tests with dilutions of 1:4, 1:20 and 1:100. For the CP determinations dilutions of 1:16 and 1:150 were used. In both tests a calibration curve with an R2 of 0.997 for TF and 0.9958 for CP was obtained.
Statistical analysisMultiple variables were dichotomised for analysis. The differences between the means in the variables with normal distribution were analysed using the parametric Student's t-test, and the variables with a non-normal distribution were analysed using the Mann–Whitney U test. An analysis of the concentrations of the biomarkers was done according to the BAI and BCI indices. These variables were dichotomised in two categories (BAI ≥7, BCI ≥4), as associated variables with a worse prognosis. In addition, an analysis was carried out of the subgroups according to race, analysing TF and CP concentrations among the mestizo patients and the African Americans. The correlations between the biomarker concentrations, SLEDAI and the range of proteinuria were analysed using Spearman's correlation coefficients.
ROC curves were constructed for CP and TF to evaluate their diagnostic yield for kidney involvement. The areas under the curve were considered exceptional (1–0.9), excellent (0.9–0.81), good (0.8–0.71), fair (0.7–0.61), and poor (0.6–0.5). SPSS (IBM SPSS version 23.0, Chicago, IL, USA) was used for the statistical analysis and GraphPad Prism (GraphPad Software version 7.0, San Diego, CA, USA).
ResultsThe information about the patients is summarised in Table 1. Of 120 patients, 85% were female; 76% mestizo, and with a mean SLEDAI of 8.4±8.9. In addition, 64.4% had kidney involvement, with mean proteinuria over 24h of 20,241±2695.7mg/dl. The use of prednisolone, cyclophosphamide, and mycophenolate mofetil was significantly higher in the patients with LN compared to those without LN. On the other hand, the patients with LN had significantly higher serosa involvement compared to the patients with no kidney involvement (44.1% vs 15.4%, P<.002). The mean time of duration of the LN at the time the biomarkers were measured was 3.3±2.6 years.
Clinical, serological and demographic features.
| Total SLE patients (n=120) | Lupus nephritis (n=76) | Non-lupus nephritis (n=44) | pa | |
|---|---|---|---|---|
| Demographic features | ||||
| Female (%) | 87 | 85 | 93 | NS |
| Mean age (years±SD) | 32.8±12.1 | 29.3±9.4 | 38.8±14.1 | 0.011 |
| Mean duration of disease (years±SD) | 7.3±6.9 | 7.1±5.8 | 7.7±8.4 | 0.038 |
| Mean SLEDAI (score±SD) | 8.4±8.9 | 11.2±9.3 | 3.2±5.2 | <0.001 |
| Race | ||||
| Mestizo (%) | 90 (76.7) | 52 (70.0) | 36 (87.0) | 0.039 |
| Afro-Latin American (%) | 25 (21.6) | 21 (28.0) | 4 (10.5) | 0.024 |
| Clinical features | ||||
| Skin involvement (%) | 83.9 | 85.7 | 81.0 | NS |
| joint involvement (%) | 80.7 | 79.2 | 83.3 | NS |
| Haematological involvement (%) | 84.4 | 85.5 | 82.5 | NS |
| Serositis (%) | 33.6 | 44.1 | 15.4 | 0.002 |
| Kidney involvement (%) | 64.4 | 100 | 0 | <0.001 |
| Mean 24h proteinuria (mg/dl±SD) | 2024.1±2695.7 | 2310.4±2779.7 | 105.9±62.3 | 0.001 |
| Pharmacological therapy | ||||
| Prednisolone (%), on occasion | 93.8 | 98.6 | 84.2 | 0.003 |
| Antimalarials (%), on occasion | 94.4 | 94.3 | 94.7 | NS |
| Cyclophosphamide (%), on occasion | 35.8 | 48.7 | 14.3 | <0.001 |
| Mycophenolate mofetil (%), on occasion | 38.4 | 55.6 | 8.3 | <0.001 |
| Rituximab (%), on occasion | 14.7 | 18.6 | 8.3 | NS |
| Serology | ||||
| Hypocomplementaemia (%), on occasion | 74.5 | 84.5 | 61.1 | 0.016 |
| Hypocomplementaemia C3 (%)b | 57/100 (57) | 44/69 (64) | 13/31 (41) | 0.04 |
| Hypocomplementaemia C4 (%)b | 65/100 (65) | 51/69 (74) | 14/31 (45) | 0.005 |
| Anti-ds DNA antibodies (%), on occasion | 69.1 | 77.1 | 55.0 | 0.001 |
| Elevated anti-ds DNA antibodies (%)b | 72/105 (69) | 64/75 (85) | 8/30 (26) | <0.0001 |
Of the total of 76 patients with LN, 55 (72%) had a diagnosis confirmed by biopsy. One patient was classified as class II, and 5 patients as class II, 36 (65.5%) patients had proliferative forms (16 class IIl patients and 20 class IV patients). Finally, 13 (23.6%) patients had class V LN.
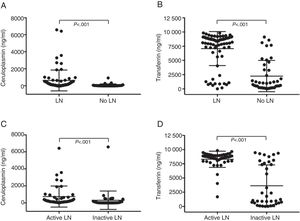
Urinary biomarkersThe urinary concentrations of CP were significantly higher in patients with LN compared to patients without LN (630.73±1232.23 vs 63.02±160.89ng/ml, P<.05). In addition, the TF levels were also significantly higher in the patients with LN compared to the patients without LN (7083.28±2997.33 vs 2244.24±2743.38ng/ml, P<.05) (Fig. 1A and B). On the other hand, it was assessed whether the levels of CP and TF correlated with LN activity, comparing the levels of the patients with active nephritis with patients without LN activity. The concentrations of CP were significantly greater in patients with active LN than those with inactive LN (742.35±1236.81 vs 309.38±1081ng/ml, P<.05); similarly, TF levels were significantly higher in the patients with active LN (8294±1427.06 vs 3627.82±3677.19ng/ml, P<.05) (Fig. 1C and D).
Urinary concentrations of CP and TF according to the presence of LN and the activity of LN. (A) Urinary CP concentrations of patients with LN and without LN. (B) Urinary concentrations of TF of patients with LN or without LN (C) Urinary concentrations of CP with active LN and inactive LN. (D) Urinary concentrations of TF of patients with active LN and inactive LN.
No statistically significant differences were found of levels of CP or TF among the different classes of LN (data not presented).
Concentrations of TF and CP were analysed according to the presence of a BAI≥7 and a BCI≥4. Concentrations of CP were significantly higher in patients with a BAI≥7; among the specific items of the Austin index, the concentrations of CP were significantly higher in patients with epithelial crescents (Table 2). There were no significant differences in levels of TF with respect to the BAI, BCI and the different items of the Austin index.
Association between TF and CP concentrations and the different items of the activity and chronicity indices.
| Biomarker | TF (ng/ml) | P | CP (ng/ml) | P | ||
|---|---|---|---|---|---|---|
| Index item | ||||||
| AI | Endocapillary hypercellularity | Yes | 7530.89±2511.25 | NS | 396.53±115.5 | NS |
| No | 7514.37±2507.61 | 239.92±98.6 | ||||
| Leukocytic infiltration | Yes | 8726.71±516.04 | NS | 390.30±460.93 | NS | |
| No | 8254.69±1001.53 | 344.81±341.43 | ||||
| Subendothelial hyaline deposits | Yes | 8451.02±749.17 | NS | 244.92±278.20 | NS | |
| No | 8540.31±931.27 | 546.01±492.38 | ||||
| Fibrinoid necrosis/karyorrhexis | Yes | 7767.24±2570.58 | NS | 394.45±495.88 | NS | |
| No | 7475.64±2497.17 | 346.18±314.90 | ||||
| Epithelial crescents | Yes | 7359.85±2787.00 | NS | 493.69±485.86 | .017 | |
| No | 7672.91±2339.33 | 228.50±219.80 | ||||
| Interstitial inflammation | Yes | 7525.91±2416.58 | NS | 400.31±447.99 | NS | |
| No | 7550.36±2738.26 | 208.60±210.12 | ||||
| CI | Glomerulosclerosis | Yes | 2447.88±3638.75 | NS | 351.89±428.13 | NS |
| No | 1671.20±452.21 | 353.48±392.87 | ||||
| Fibrous crescents | Yes | 6931.40±2901.89 | NS | 125.02±79.33 | .025 | |
| No | 7675.04±2431.31 | 443.93±427.64 | ||||
| Tubular atrophy | Yes | 7026.66±2676.27 | NS | 291.01±339.5 | NS | |
| No | 7777.75±2418.58 | 423.41±442.26 | ||||
| Interstitial fibrosis | Yes | 6871.04±2768.08 | NS | 291.01±339.5 | NS | |
| No | 7807.57±2376.67 | 423.41±442.26 | ||||
AI: activity index; CI: chronicity index.
In the analysis by race, we found that the patients of Afro-Latin American origin had significantly higher concentrations of urinary TF (6638.8±3250 vs 5017.5±3778.8ng/ml, P=.01) than the mestizo patients, but we found no significant differences with regard to concentrations of CP (4843±709 vs 4189±1123.1ng/ml, P=.57). In patients with NL, concentrations of urinary TF were also significantly higher in Afro-Latin Americans than in mestizos, but there were no differences in CP concentrations (data not presented).
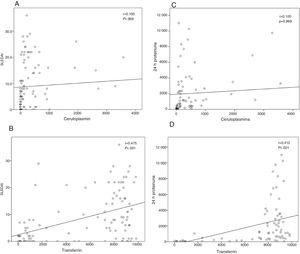
Correlation between biomarkers and clinical characteristicsA statistically significant positive correlation was found between concentrations of TF and SLEDAI (rho=0.475, P<.001) and the range of proteinuria (rho=0.412, P<.001). In addition, the concentrations of TF and CP correlated positively with each other, a finding that was statistically significant (rho=0.345, P<.001). On the other hand, there was no correlation between concentrations of CP and the range of proteinuria or the SLEDAI (Fig. 2A–D). A statistically significant positive correlation was also found between the modified SLEDAI without including the kidney domain scores and the urinary TF concentrations (rho=0.253, P<.05) (data not presented).
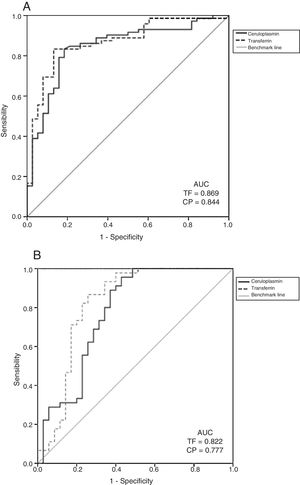
The ROC curves for discriminating LN determined an excellent diagnostic value for CP (AUC=0.84, 95% CI: 0.76–0.92), and TF (AUC=0.86, 95% CI: 0.79–0.93) (Fig. 3A). On the other hand, both biomarkers performed well for diagnosing active LN, both TF (AUC=0.82, 95%CI: 0.66–0.88) and CP (AUC=0.77, 95% CI: 0.71–0.92) (Fig. 3B).
DiscussionIn this study, we found concentrations of TF and CP that were significantly higher in patients with LN compared to those without LN. Similarly, the concentrations of both biomarkers were significantly higher in patients with active LN than those without kidney activity. Furthermore, a positive correlation was found between concentrations of TF, SLEDAI and 24h proteinuria. We also found a positive correlation between concentrations of CP and concentrations of TF. Regarding the performance of these biomarkers, we found excellent diagnostic value for both in determining the presence of LN; they also had good discriminatory power (TF and CP) in determining LN activity.
These results are in line with those of some previous studies.10,14 In a multi-centre, initial validation study of a panel of biomarkers for the diagnosis of active LN undertaken in the United States and Canada on a principally African American and Caucasian population, it was found the concentrations of TF and CP were found to be higher in the patients with active LN.10 On the other hand, another international validation study of a panel of biomarkers found that concentrations of TF and CP and other biomarkers were significantly higher in patients with active LN compared to patients without LN; in this study, CP, TF and other biomarkers strongly correlated with each other. This panel of biomarkers had an excellent AUC of 0.952.14 Another study, performed in Cincinnati by Brunner et al.,15 on a cohort of 76 patients, of whom 46% were African Americans and 43% Caucasians analysed the association between urinary biomarkers and the histological characteristics of the kidney biopsy for LN, and found that TF and CP, among other biomarkers, differentiated between high and low BAI scores (≥7). We replicated this finding in our study, since CP concentrations were significantly higher in the patients with BAI≥7. Likewise, a significant correlation between CP concentration and the kidney SLEDAI (K-SLEDAI) was found. CP formed part of a panel of activity predictor variables ≥7 in the kidney biopsy, with an AUC of 0.85. On the other hand, TF formed part of a predictive panel for membranous LN (class V), with an AUC of 0.75. In this study we found no differential levels between classes of LN and concentrations of urinary TF and CP.
In the same manner, it has been reported that CP and TF concentrations, in combination with other biomarkers, are higher in patients with LN who respond to therapy, compared to non-responders.16 Given the nature of our study, the relationship between clinical response and biomarker concentrations was not possible to establish.
As already established in other diseases in which kidney damage occurs – such as Fanconi's syndrome, where urinary TF concentrations have been found to be elevated,6 high concentrations of TF and CP in LN might be due to the fact that the cells suffer a degree of hypoxia that stimulates the expression of iron response elements and iron regulatory proteins (IRE and IRP, respectively), and in turn to increased iron uptake through TF, and thanks to the ferro-oxidase action of CP.
This study has some limitations. As we mention above, the cross-sectional design of the study did not enable us to evaluate the behaviour of biomarkers over time or their relationship with treatment. Likewise, we did not have a healthy control group to perform the comparative analysis. Furthermore, our study was undertaken in a university referral hospital with a cohort of patients with highly complex SLE, as shown by the high activity indices, which mean our results cannot be extrapolated to patients with milder forms of SLE. Finally, although this study was performed in a highly complex centre, not all the patients had access to a kidney biopsy, either because it was not considered appropriate by the clinician, or due to administrative difficulties.
On the other hand, some of the strengths of our study were that it is the first to study concentrations of TF and CP in mestizo and Afro-Latin American patients, reporting differences in the concentration of the biomarkers between subgroups, the strict selection process of patients with SLE, discounting patients with concomitant autoimmune disorders, or active infectious processes, and its systematic process of gathering, storing and processing the urine samples.
In conclusion, in our study we found that TF and CP are biomarkers with the potential to differentiate LN, and even to serve as markers for it, which could be a non-invasive alternative to kidney biopsy for some selected patients, or for patients with no access to kidney biopsy, although these results still have to be validated prospectively.
As future perspectives, we expect to validate these results in patients with SLE throughout Latin America, in collaboration with the Latin American Lupus Study Group (GLADEL).
Conflict of interestsThe authors have no conflicts of interest to declare.
This study was financed by Colciencias (call 656 of 2014), Francisco José de Caldas Patrimonio autónomo Fondo Nacional de Financiamiento para la ciencia, la tecnología y la innovación (Autonomous National Heritage Fund for Financing Science, Technology and Innovation), and by the sustainability programme of the University of Antioquia.
Please cite this article as: Urrego T, Ortiz-Reyes B, Vanegas-García AL, Muñoz CH, González LA, Vásquez G, et al. Transferrina y ceruloplasmina en orina de pacientes con lupus eritematoso sistémico. ¿Son útiles para diferenciar pacientes con nefritis lúpica? Reumatol Clín. 2020;16:17–23.