This study aimed to evaluate the efficacy of secukinumab (SEC) in axial spondyloarthropathy (axSpA) in anti-TNFα naïve and anti-TNFα experienced patients. It also focused on the duration of SEC treatment and its side effects.
Patients and methodsThe patients with axSpA treated with SEC and followed up in our outpatient clinic from May 2018 through October 2021 were included in this study. All patients in the study also fulfilled the ASAS classification criteria for axSpA. Patients were separated into two groups according to whether they received prior anti-TNFα therapy. While anti-TNFα naïve patients comprised group 1, anti-TNFα experienced patients were included in group 2. Pre- and post-treatment BASDAI scores were reported and compared.
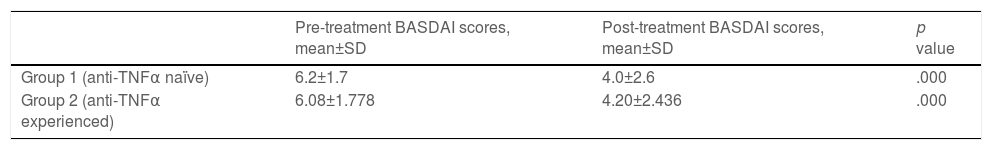
ResultsEighty-four axSpA patients (42 men; duration of the disease: 86.86±65.35 months in group 1 and 160.65±97.4 months in group 2) were treated with SEC. 45.5% of anti-TNFα naïve patients and 56.5% of anti-TNFα experienced patients were still on SEC therapy in October 2021. Duration of SEC treatment was 12.5±7.9 months in group 1 and 17.19±12 months in group 2 (p=0.098). The differences between pre-and post-treatment BASDAI scores were statistically significant in both groups (p<0.001). While patients in group 1 did not develop any adverse effects, three patients in group 2 experienced alopecia, uveitis, and recurrent pneumonia after SEC treatment.
ConclusionOur study's efficacy and safety data on the use of SEC were reassuring in both anti-TNFα naïve and anti-TNFα experienced patients. However, further studies are still needed to determine the appropriate timing to begin SEC treatment.
Este estudio tuvo como objetivo evaluar la eficacia de secukinumab (SEC) en la espondiloartropatía axial (axSpA) en pacientes sin experiencia previa con anti-TNFα y con experiencia con anti-TNFα. También se centró en la duración del tratamiento SEC y sus efectos secundarios.
Materiales y métodosSe incluyeron en este estudio los pacientes con axSpA tratados con SEC y seguidos en nuestra consulta externa desde mayo de 2018 hasta octubre de 2021. Todos los pacientes en el estudio también cumplían con los criterios de clasificación de ASAS para axSpA. Los pacientes se separaron en dos grupos según si habían recibido terapia anti-TNFα previa. Mientras que los pacientes sin tratamiento previo con anti-TNFα comprendían el grupo 1, los pacientes con experiencia con anti-TNFα se incluyeron en el grupo 2. Se informaron y compararon las puntuaciones BASDAI antes y después del tratamiento.
ResultadosOchenta y cuatro pacientes con axSpA (42 hombres; duración de la enfermedad: 86,86 ±65,35 meses en el grupo 1 y 160,65±97,4 meses en el grupo 2) fueron tratados con SEC. El 45,5% de los pacientes sin experiencia previa con anti-TNFα y el 56,5% de los pacientes experimentados con anti-TNFα seguían en tratamiento con SEC en octubre de 2021. La duración del tratamiento con SEC fue de 12,5±7,9 meses en el grupo 1 y de 17,19±12 meses en el grupo 2 (p=0,098). Las diferencias entre las puntuaciones BASDAI antes y después del tratamiento fueron estadísticamente significativas en ambos grupos (p<0,001). Mientras que los pacientes del grupo 1 no desarrollaron ningún efecto adverso, tres pacientes del grupo 2 experimentaron alopecia, uveítis y neumonía recurrente después del tratamiento con SEC.
ConclusionesLos datos de eficacia y seguridad de nuestro estudio sobre el uso de secukinumab fueron alentadores tanto en pacientes sin tratamiento previo con anti-TNFα como en pacientes experimentados con anti-TNFα. Sin embargo, aún se necesitan más estudios para determinar el momento apropiado para comenzar el tratamiento con SEC.
Spondyloarthropathies (SpA) are a group of inflammatory joint disorders that are divided into two major categories: axial SpA (axSpA) and peripheral SpA. The axSpA are further divided as radiographic-axSpA (r-axSpA), namely ankylosing spondylitis (AS), and non-radiographic-axSpA (nr-axSpA) according to the presence of radiographic sacroiliitis on plain X-ray.1,2 The disease burden is mostly similar in r-axSpA and nr-axSpA.3,4
Pharmacological intervention for SpA focuses on relieving symptoms and preventing disease progression. Nonsteroidal anti-inflammatory drugs (NSAIDs) are beneficial for most symptomatic axSpA and are generally initial therapy. However, the capability of hindering the new bone formation of NSAIDs is scarce, so their long-term use is not enough to prevent disease-related morbidity.5 Disease-modifying antirheumatic drugs (DMARDs) have led to significant improvement in the management of SpA. Conventional synthetic DMARDs are usually ineffective in treating axial manifestations; instead, they are useful for treating peripheral manifestations. The biologic DMARDs, which provide long-term control of the disease and help to prevent consequent axial immobility, are comprised of anti-TNFα and anti-IL17A agents.
Secukinumab (SEC), a fully humanized IgG1 anti-IL 17A monoclonal antibody, showed remarkable efficacy for axSpA in Measure 2 and Measure 3 trials.6,7 Europian Medicines Agency and the United States Food and Drug Administration approved SEC for AS in 2015 and 2016, respectively. According to the current Assessment of Spondyloarthritis International Society (ASAS) guidelines, SEC is recommended after the first-line therapy with anti-TNFα.8 Previous studies have concluded that SEC was more efficacious in anti-TNFα naïve patients, although the current guidelines recommend starting the treatment with anti-TNFα agents.8–11
SEC is administered subcutaneously 150mg weekly for four weeks as a loading dose and then continued monthly for maintenance.12–14 There are currently sparse real-life data on the outcomes of SEC treatment in axSpA patients. This retrospective study aimed to evaluate the efficacy of SEC for axSpA in two patient groups separated according to previous anti-TNFα exposure by using Bath Ankylosing Spondylitis Disease Activity Index (BASDAI).8 BASDAI is used commonly in clinical practice and includes patient-reported outcomes, and a score ≥4 indicates high disease activity.8,6 The duration of the treatment and side effects were also studied along with the treatment response.
Materials and methodsThe present study retrospectively evaluated patients diagnosed with AS and followed up in the outpatient rheumatology clinic of Cerrahpasa Medical Faculty from May 2018 through October 2021. Patients who used SEC during their follow-ups and fulfilled the ASAS criteria6 were included in the study. Patients who were under 18, not able to express themselves, or diagnosed with psoriatic arthritis were excluded. All patients had used non-biologic DMARDs before beginning the biologic agent. Biologic and non-biologic DMARDs were not administered to patients concurrently.
Baseline demographic characteristics (gender and age), duration of the disease, type of involvement (axial only, axial and peripheral), grade of involvement (radiographic or not), pre-treatment BASDAI scores, best BASDAI scores after SEC treatment, and side effects of the treatment were assessed.
Patients were divided into two groups according to their previous anti-TNFα exposure. Furthermore, patients in both groups were evaluated only as long as they used SEC, and treatments subsequent to SEC were not taken into account. Group 1 consisted of the patients who had not previously received anti-TNFα therapy and were called anti-TNFα naïve. Group 2 consisted of patients who had received one or more anti-TNFα agents and were called anti-TNFα experienced. Moreover, no patient received any biologic before SEC other than anti-TNFα. Patients in group 2 had been treated with at least one of the following anti-TNFα agents prior to treatment with SEC: etanercept, adalimumab, certolizumab, infliximab, and golimumab. These anti-TNFα agents were interchanged according to treatment response and development of adverse effects. SEC was started in patients who did not respond adequately under anti-TNFα or developed side effects. The decision on when to start SEC treatment was made based on expert opinion and the availability of the agents because of the absence of solid recommendations.11 Based on their SEC response, groups 1 and 2 were further divided into three subgroups: patients still on SEC therapy, primary non-responders, and secondary non-responders. Patients who remained unresponsive to treatment after a loading dose of SEC were defined as primary-nonresponders and patients who developed resistance after at least six months of consistent resolution of symptoms were defined as secondary-non-responders.9,10 All patients gave their consents for their data to be used in research upon admission. Approval was received from the independent ethics committee of Cerrahpasa Medical Faculty to look for data in the clinical charts of the patients before the initiation of the study.
Statistical analysisAll analyses were performed with the IBM SPSS Statistics for Windows, Version 21.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were indicated as numbers and percentages for categorical variables and as means and standard deviations for numerical variables. Demographic data were evaluated by Pearson's chi-square test. Within the groups, patients who were still on treatment, primary non-responders, and secondary non-responders were evaluated by the Student's t-test method. Changes in the pre- and post-treatment BASDAI scores in both groups were evaluated by paired t-test method. p<0.05 was regarded as statistically significant.
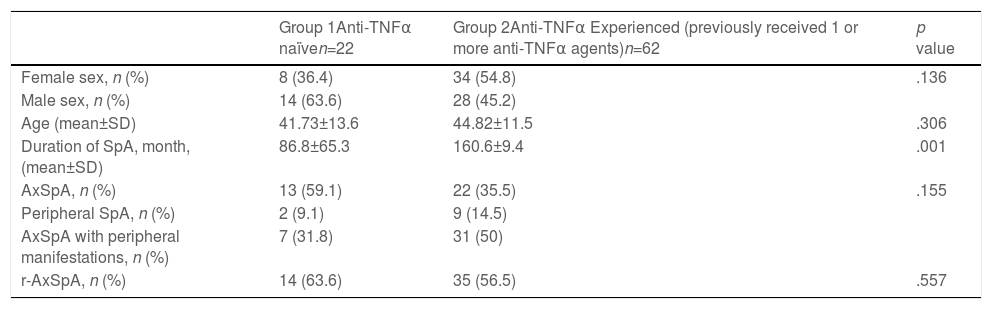
ResultsThe present study included 84 patients (mean age: 41.73±13.6 in group 1, 44.82±11.5 in group 2) with axSpA (42 males and 42 females) who received SEC at any time of their follow-ups. The mean duration of the disease was 86.86±65.35 months in group 1 and was 160.65±97.4 months in group 2 (p=0.001). The demographic and clinical characteristics of the patients are presented in Table 1.
Baseline characteristics of anti-TNFα naïve and anti-TNFα experienced SpA patients.
| Group 1Anti-TNFα naïven=22 | Group 2Anti-TNFα Experienced (previously received 1 or more anti-TNFα agents)n=62 | p value | |
|---|---|---|---|
| Female sex, n (%) | 8 (36.4) | 34 (54.8) | .136 |
| Male sex, n (%) | 14 (63.6) | 28 (45.2) | |
| Age (mean±SD) | 41.73±13.6 | 44.82±11.5 | .306 |
| Duration of SpA, month, (mean±SD) | 86.8±65.3 | 160.6±9.4 | .001 |
| AxSpA, n (%) | 13 (59.1) | 22 (35.5) | .155 |
| Peripheral SpA, n (%) | 2 (9.1) | 9 (14.5) | |
| AxSpA with peripheral manifestations, n (%) | 7 (31.8) | 31 (50) | |
| r-AxSpA, n (%) | 14 (63.6) | 35 (56.5) | .557 |
TNF, tumor necrosis factor; SpA, spondyloarthritis; Ax-SpA, axial-spondyloarthritis; r-AxSpA; radiographic axial-spondyloarthritis; nr-AxSpA, non-radiographic axial-spondyloarthritis; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; SD, standard deviation.
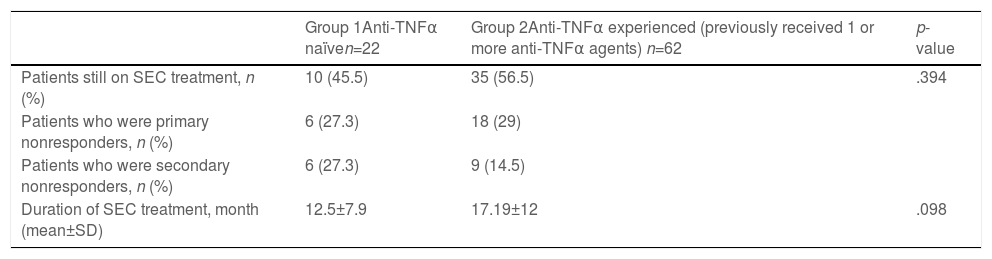
This group included 22 patients (14 males), of whom 13 had axSpA, and 9 had axSpA with peripheral manifestations (Table 1). Eight patients had no radiographic evidence of sacroiliitis, and 14 had r-axSpA (Table 1). Ten patients were still on SEC therapy and the mean duration of SEC therapy was 12.5±7.9 months (Table 2). The mean BASDAI score was 6.24±1.763 before SEC treatment, and the patient-reported best score mean was 4.07±2.649 (p<0.001) after the treatment (Table 3). Treatment cessation was due to primary unresponsiveness in 6 patients and secondary unresponsiveness in 6 patients (Table 2). No adverse events were observed in this group.
Secukinumab treatment details of anti-TNFα naïve and anti-TNFα experienced SpA patients.
| Group 1Anti-TNFα naïven=22 | Group 2Anti-TNFα experienced (previously received 1 or more anti-TNFα agents) n=62 | p-value | |
|---|---|---|---|
| Patients still on SEC treatment, n (%) | 10 (45.5) | 35 (56.5) | .394 |
| Patients who were primary nonresponders, n (%) | 6 (27.3) | 18 (29) | |
| Patients who were secondary nonresponders, n (%) | 6 (27.3) | 9 (14.5) | |
| Duration of SEC treatment, month (mean±SD) | 12.5±7.9 | 17.19±12 | .098 |
TNF, tumor necrosis factor; SD, standard deviation; SEC, Secukinumab.
Pre- and post-treatment BASDAI scores of anti-TNFα naïve and anti-TNFα experienced SpA patients.
| Pre-treatment BASDAI scores, mean±SD | Post-treatment BASDAI scores, mean±SD | p value | |
|---|---|---|---|
| Group 1 (anti-TNFα naïve) | 6.2±1.7 | 4.0±2.6 | .000 |
| Group 2 (anti-TNFα experienced) | 6.08±1.778 | 4.20±2.436 | .000 |
TNF, tumor necrosis factor; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; SD, standard deviation.
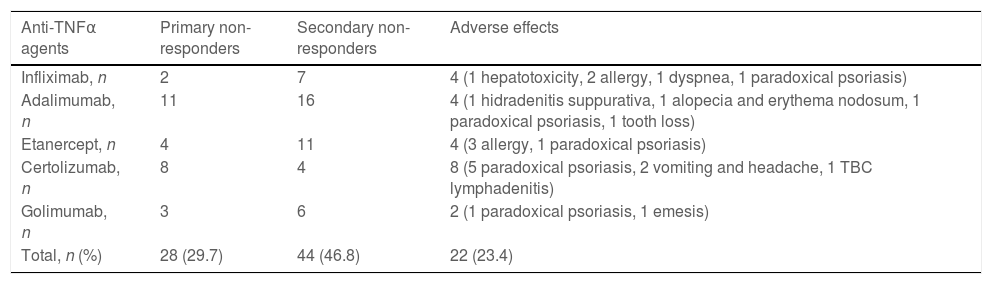
This group included 62 patients (28 males), of whom 22 had axSpA, and 40 had axSpA with peripheral manifestations (Table 1). 27 patients had no radiographic evidence of sacroiliitis, and 35 had r-axSpA. Thirty-five patients were still on SEC therapy, and the mean duration of SEC therapy was 17.19±12 months (Table 2). The mean BASDAI score was 6.08±1.778 before the initiation of SEC, and the patient-reported best score mean was 4.20±2.43 (p<0.001) after the treatment (Table 3). Primary unresponsiveness in 18 patients and secondary unresponsiveness in 9 patients caused treatment cessation (Table 2). Before the onset of SEC patients had used one or more anti-TNFα agents and in whom causes of discontinuation were of primary unresponsiveness in 28 (29.7%), secondary unresponsiveness in 44 (46.8%), and adverse effects in 22 (23.4%) (Table 4). The causes of discontinuation of anti-TNFα agents in group 2 are also given in Table 4.
Causes of discontinuation of anti-TNFα agents in group 2.
| Anti-TNFα agents | Primary non-responders | Secondary non-responders | Adverse effects |
|---|---|---|---|
| Infliximab, n | 2 | 7 | 4 (1 hepatotoxicity, 2 allergy, 1 dyspnea, 1 paradoxical psoriasis) |
| Adalimumab, n | 11 | 16 | 4 (1 hidradenitis suppurativa, 1 alopecia and erythema nodosum, 1 paradoxical psoriasis, 1 tooth loss) |
| Etanercept, n | 4 | 11 | 4 (3 allergy, 1 paradoxical psoriasis) |
| Certolizumab, n | 8 | 4 | 8 (5 paradoxical psoriasis, 2 vomiting and headache, 1 TBC lymphadenitis) |
| Golimumab, n | 3 | 6 | 2 (1 paradoxical psoriasis, 1 emesis) |
| Total, n (%) | 28 (29.7) | 44 (46.8) | 22 (23.4) |
TNF, tumor necrosis factor; TBC, tuberculosis.
A patient developed alopecia on the 26th month of SEC therapy. Therefore, SEC was switched to tofacitinib. Another patient developed uveitis confirmed by ocular examination on the 16th month of SEC therapy and SEC was changed to adalimumab. An elderly patient developed recurrent pneumonia twice within six months after the initiation of SEC therapy. For this reason, the patient was placed on hydroxychloroquine therapy without any biologic agent.
DiscussionThe use of SEC in treating axSpA is increasing, and currently, there are limited real-life data on SEC treatment outcomes in patients with axSpA. More real-life data on SEC safety profile and effectiveness will guide rheumatologists in their treatments. In the present study, we assessed the clinical characteristics of patients receiving SEC in our center and their responses to therapy. Among the study groups, which are constituted according to prior anti-TNFα exposure, there were distinctions in baseline characteristics of the patients, although most of them were not statistically significant. In anti-TNFα naïve patients, the percentage of male patients was higher (p=0.136). Disease duration was considerably longer in anti-TNFα experienced patients (p=0.001). The patient's ages were similar between the two groups (p=0.306). The percentage of axSpA with peripheral manifestations was higher in group 2 (p=0.058). In group 1, the number of r-axSpA patients was almost twice as high as the number of nr-axSpA patients.
The duration of SEC therapy was longer in group 2 (12.5±7.9 months in group 1, 17.19±12months in group 2, p=0.098). The percentage of patients still on SEC treatment was higher in group 2 (56.5%) relative to group 1 (45.5%). The percentage of primary non-responders was similar between groups. In comparison to group 1, the proportion of secondary non-responders was lower in group 2. The main reason for the cessation of anti-TNFα agents was secondary unresponsiveness (46.8%) in our patients, and adverse effects constituted the smallest percentage (23.4%). Paradoxical psoriasis as an adverse event was observed (40.9%) with each anti-TNFα agent in our study, while certolizumab was responsible for most of them. Both groups achieved statistically significant reductions in BASDAI scores after SEC treatment (p<0.001).
Compared to Measure 2 and Measure 3 trials which led to SEC approval for AS, patients in our study were of similar ages with those of Measure trials (mean age 43 vs. 42 and 43 years), had a lower proportion of anti-TNFα naïve patients (35% vs. 61% and 57%), and had lower baseline BASDAI scores (mean BASDAI 6.1 vs. 6.6 and 7.0).6,7 Unlike Measure 2 and Measure 3 trials, which included patients with r-axSpA only, our study included patients with r-axSpA and nr-axSpA both.6,7 Patients with nr-axSpA were found to respond to SEC treatment along with patients with r-axSpA in our study. In other words, our study outcomes were in line with a recent randomized-controlled study results regarding the resolution of symptoms of non-radiographic axial spondyloarthritis in patients treated with SEC.15 Williams et al. concluded that SEC was efficacious for improving mean BASDAI scores of patients when used as first-line, second-line, or third-line.16 Thus, our study outcomes were consistent with the previous studies regarding the effect of SEC on axSpA.
Even though two studies concluded that the first-line anti-TNFα retention rate was higher in biologic DMARD naïve patients, our study suggested that it was not the case for the axSpA patients on SEC therapy.17,18 Also, Michelsen et al. supported SEC use as first-line therapy for axSpA.10 However, they also emphasized the need for head-to-head studies on treatment effectiveness of anti-TNFα and anti-IL17 agents.10 A study from Italy reported no significant differences in the BASDAI scores and global retention rate between biologic-naïve patients and patients who previously failed to respond to anti-TNFα (p=0.482 and p=0.619, respectively).19 We observed that the patients using SEC in the later stages of their diseases might be treated with SEC for longer periods (p=0.098) even though there was no considerable difference in BASDAI scores between the two groups. In 2019, the American College of Rheumatology (ACR) recommended anti-TNFα agents over secukinumab as the first biologic in both r-axSpA and nr-axSpA based on greater experience with anti-TNFα agents and familiarity with their long-term safety and toxicity.11 On the other hand, our study confirmed that SEC treatment alleviated the symptoms in ax-SpA in both biologic naïve and anti-TNFα experienced patients (p<0.001).
We observed adverse effects that were reported in the literature previously in a small proportion of patients.20–22 Although inflammatory bowel disease (IBD) cases associated with SEC were reported in the literature, we did not observe new-onset IBD in follow-ups of our patients.23 Safety data in our clinic was in line with what has been reported in the previous studies.
The most important limitation of our study is that it was retrospective. However, presenting real-life data provides crucial information for our daily clinical practice. Another factor that might have impacted study results was that SEC entered the market in Turkey in 2018 even though it was approved for axSpA in 2015. Therefore, the difference in treatment durations between the two groups would probably be statistically significant if we had the opportunity to observe patients for a longer period. Also, since this study investigated patients in a single-center registry and SEC entered the market recently in Turkey, our sample size is relatively small. On the other hand, peripheral involvement might be a confounding variable because there was a higher proportion of patients with axSpA with peripheral manifestations in group 2, though the difference was not significant. Further studies that exclude the patients who received anti-TNFα should be conducted and examine whether the SEC treatment outcomes differ according to the pattern of joint involvement. Also, designing prospective studies that include more patients with multiple centers can provide more data regarding peripheral involvement rates in patients who were unresponsive to first-line anti-TNFα as biologic treatment. By merging the data of those future studies, we can figure out more clearly when to start SEC therapy and which joint involvement pattern would be most appropriate to select SEC.
ConclusionSEC, a recombinant anti-IL17A monoclonal antibody, is an alternative to anti-TNFα agents for patients with SpA. In our patients with axSpA, SEC was found to have a satisfying safety profile and efficacy in both anti-TNFα naïve and anti-TNFα experienced groups. However, there is still a scarcity of evidence regarding the timing of SEC treatment. Prospective real-life studies should be performed to guide the recommendations for SEC treatment in different stages of axSpA.
Ethics approvalApplicable, 5.10.2021-203356.
Consent to participateNot applicable.
Consent for publicationApplicable.
Availability of data and materialNot applicable.
Code availabilityNot applicable.
FundingNo funding was received.
Conflict of interestThe authors declare they have no conflicts of interest.
The authors want to thank Deniz Tuna and Tolga Tunc for their assistance in Spanish translation.