We present the case of a 71-year-old male with a history of hypertension in treatment with 20mg/day of enalapril, as well as hyperuricemia, with episodes of monoarthritis of the left foot, without a diagnosis or follow up by Rheumatology.
The patient was admitted due to pain and swelling on the second finger of the right hand and the fourth proximal interphalangeal joint of the left hand, without fever or other clinical data.
Physical examination showed Heberden's nodules on both hands, with signs of arthritis on the second distal interphalangeal joint of the right hand, with an accompanying tophus, as well as on the fourth proximal interphalangeal joint of the left hand.
On laboratory tests he presented uric aid of 7.20mg/dl, with a normal blood count, chemistry and acute phase reactants (ESR: 14mm/h, CRP: 0.49mg/dl).
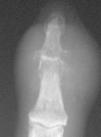
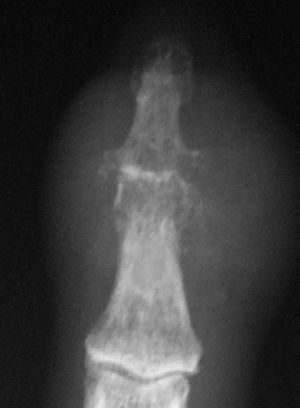
On hand x ray there was evidence of degenerative changes on metacarpophalangeal, interphalangeal, and carpometacarpal joints. The distal interpalangeal joint of the second finger presented soft tissue swelling, with a reduction in joint space and large erosions (Fig. 1). Ecocardiography and serial blood cultures were negative.
Arthrocenthesis of the swollen joint was performed, finding abundant intracellular and extracellular monosodium urate (MSU) crystals under polarized light microscopy, confirming the diagnosis of chronic tophaceous gout.
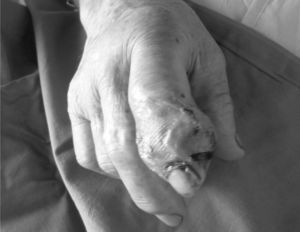
In addition, surgical drainage of the abscess (Fig. 2), obtaining a milky fluid. Samples for culture showed growth of Staphylococcus lugdunensis (S. lugdunensis), respondent to betalactamics, macrolides, and quinolones. Treatment with IV cloxacylin was started, lasting 2 weeks (2g/4h).
Progression was favorable, with clinical improvement and culture negativity after onset of treatment with cloxacylin, completed afterward with ciprofloxacyn, 750mg/12h, for 2 weeks as an outpatient.
DiscussionThe association between rheumatoid arthritis and septic arthritis is well known,1 but what is not so well documented is the coexistence of gout and septic arthritis, although there is an increasing number of published clinical cases and case series. In the study by Wallace et al.,2 on preliminary classification criteria for gout, 4% of patients with septic arthritis had coexisting gout (tophi were identified in most cases). In another study by Shah et al.,3 265 synovial fluid aspirations were analyzed, of which 183 (69%) presented monosodium urate crystals, 81 (30.6%) calcium pyrophosphate crystals, and 1 case (0.4%) where both types of crystals were isolated. The percentage of association of microcrystalline arthritis and septic arthritis was 1.5% (4 cases, of which 3 had pyrophosphate crystals and only 1 case of monosodium urate crystals).
In both situations, it is clinically manifested mainly by joint swelling and pain and it is not always easy to differentiate if the arthritis is due to this or other pathology, especially if there is no clinical evidence of infection. Delay in diagnosis of septic arthritis can cause significant joint destruction.4 It is important that, even when we make a diagnosis of gout by viewing monosodium urate crystals under polarized light microscopy, the synovial fluid samples are sent for culture and Gram staining,5 especially in cases of tophaceous or severe gout.
S. lugdunensis was described in 1988 by Freney et al.,6 and it is a coagulase negative staphylococcus, similar to S. aureus. Its incidence has increased in recent years thanks to its correct microbiological identification. It is usually isolated from skin and soft tissue infections, but has also been reported in cases of native valve endocarditis, breast abscesses, osteomyelitis, peritonitis, lymphangitis, bacteremia, and catheter associated infections.7 It is associated with recurrent infections of prosthetic joints and after procedures such as arthroscopy.8S. lugdunensis mainly cause infections in immunocompromised patients.7 Among its virulence factors, the ability of binding to the extracellular matrix protein, fibronectin and fibrinogen stand out.
The sensitivity of ECN to antibiotics depends on the species and origin, whether hospital or community-acquired strain. In our environment, S. lugdunensis has a good sensitivity to beta-lactams. Given the isolation of an ECN and in particular S. lugdunensis, we must be alert to their role as a more aggressive pathogen in immunosuppressed patients.
The focus of our case lies in the importance of synovial fluid samples sent for culture, even when viewing crystals, and the rare isolation of S. lugdunensis in a patient without immune system compromise or previous surgical procedure.
Please cite this article as: Carrasco Cubero C, De la Iglesia Salgado JL. No todo es gota. Reumatol Clin. 2012;8(1):46–7.