Chronic diseases have a great impact on the morbidity and mortality and on the health-related quality of life (HRQoL) of patients around the world. The impact of rheumatic diseases has not been fully recognized. We conducted a comparative study to evaluate the HRQoL in different chronic diseases.
ObjectivesThe aim of the present study was to assess the HRQoL and identify specific areas affected in patients with rheumatoid arthritis (RA), osteoarthritis (OA), diabetes mellitus, and end-stage renal disease, in geriatric subjects and in a control group.
Patients and methodsWe conducted a cross-sectional study, in a General Hospital in Morelia, Mexico. All patients met the classification criteria for RA, OA, diabetes mellitus, and end-stage renal disease; the geriatric subjects group was aged ≥65 years and the control group ≥30 years. Demographic characteristics were recorded, different instruments were applied: SF-36, visual analog scale for pain, patient's and physician's global assessments, Beck Depression Inventory and specific instruments (DAS-28, HAQ-Di, WOMAC, Diabetes Quality of Life [DQOL] and Kidney Disease Questionnaire of Life [KDQOL]). Biochemical measures: erythrocyte sedimentation rate, blood count, glucose, HbA1C, serum creatinine and urea.
ResultsWe evaluated 290 subjects (control group: 100; geriatric subjects: 30 and the rest of groups: 160). Differences were detected in baseline characteristics (P<.0001). The SF-36 scores were different between the control group and other groups (P=.007). The worst HRQoL was observed in the end-stage renal disease group (SD: 48.06±18.84x/SD). General health was the principal affected area in RA. Pain was higher in rheumatic diseases: OA (5.2±2.4) and RA (5.1±3). HAQ was higher in OA compared to RA (1.12±.76 vs .82±.82, respectively; P=.001). Forty-five percent of all subjects had depression.
ConclusionsThe HRQoL in RA patients is poor and comparable to that of other chronic diseases (end-stage renal disease and diabetes mellitus). Rheumatic diseases should be considered as high impact diseases and therefore should receive more attention.
Las enfermedades crónicas impactan en la morbimortalidad y en la calidad de vida relacionada con la salud (CVRS) de los pacientes a nivel mundial. El impacto de las enfermedades reumáticas no ha sido totalmente reconocido.
ObjetivosDeterminar la CVRS y evaluar áreas específicas en artritis reumatoide (AR), osteoartritis (OA), diabetes mellitus, insuficiencia renal terminal, población geriátrica y un grupo control.
Pacientes y métodosEstudio transversal, realizado en el Hospital General de Morelia. Los sujetos cumplían criterios para AR, OA, diabetes mellitus, insuficiencia renal terminal, un grupo de población geriátrica (≥65 años) y un grupo control ≥30 años. Se determinaron características sociodemográficas y se aplicaron instrumentos: SF-36, escala visual analógica de dolor, valoración global del paciente y médico, inventario para depresión de Beck, e instrumentos específicos (DAS-28, HAQ-Di, WOMAC, Diabetes Quality of Life [DQOL] y Kidney Disease Questionnaire of Life [KDQOL]). Mediciones bioquímicas: velocidad de sedimentación globular (VSG), biometría hemática (BH), glucosa, HbA1C, creatinina y urea.
ResultadosFueron evaluados 290 sujetos (un grupo control: 100, población geriátrica 30 y 160 en los demás grupos). Se detectaron diferencias (p<0,0001) en las características basales. Los puntajes del SF-36 fueron diferentes entre los grupos (p=0,007). La peor CVRS se observó en el grupo de insuficiencia renal terminal (media±DE: 48,06±18,84). En el grupo de AR la salud en general fue el área más afectada. El dolor fue mayor en las enfermedades reumáticas: OA (5,2±2,4) y AR (5,1±3). El HAQ-Di fue mayor en OA comparado con AR (1,12±0,76 vs. 0,82±0,82 respectivamente; p=0,001). El 45% de los sujetos tuvo depresión.
ConclusionesLa CVRS en pacientes con AR es mala y equiparable a lo que sucede en pacientes con enfermedades crónicas (insuficiencia renal terminal y diabetes mellitus). Las enfermedades reumáticas deben considerarse padecimientos de alto impacto y por ello merecen mayor atención.
Quality of life related to health (HRQoL) is a multidimensional concept, related to the individual impact of the disease and its treatment, as well as functional capacity and the patient's perception in social, physical and mental1 roles.
Rheumatoid arthritis (RA) and osteoarthritis (OA) are two common rheumatic diseases associated with impaired physical function and HRQoL, affecting different age groups, most of them in2 productive stages of life.
Chronic diseases impact on morbidity and quality of life of patients worldwide, and are responsible for 72% of the total burden of disease.3 It is well known that end-stage renal disease (ESRD) and diabetes mellitus (DM) have a great impact on a patient's physical, mental and emotional role.4–6
The instruments to assess quality of life can be generic and specific. Generic instruments such as the SF-36 questionnaire allow us to compare HRQoL between chronic diseases (DM and ESRD) and rheumatic diseases (RA and OA).7
The level of activity of rheumatic diseases correlates inversely with HRQoL; one of the instruments used to determine the activity in RA is the Disease Activity Score (DAS-28),8 and, for OA, the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), which allows us to determine the degree of functional compromise.
The specific instruments used for estimating HRQoL in ESRD and DM (DQOL and KDQOL SF) explore points such as satisfaction with glycemic control, dialysis sessions and drug therapy.9,10
It is well known that HRQoL is reduced in rheumatic diseases as well as metabolic diseases, and this aspect has become so important that it is one of the main primary outcomes used to determine the effectiveness of treatments in each disease.
Rheumatic diseases have not been fully recognized as having a high impact on HRQoL. No information about the comparison of the quality of life of chronic diseases and rheumatic diseases exists in our country, and for that reason, we decided to evaluate HRQoL in two rheumatic diseases (RA, OA), 2 chronic diseases with high medical and social impact (DM and ESRD), a group of geriatric patients and a control group.
Patients and MethodsThis was a cross-sectional study which included consecutive patients attending the rheumatology, internal medicine, endocrinology and nephrology clinics in Morelia, Michoacán's General Hospital “Dr. Miguel Silva”, within a one year period. The patients met the classification criteria of the American College of Rheumatology for RA or OA11,12 (hip and knee) and the criteria of the American Diabetes Society 200413 for DM (at least 2 years of evolution), and the ESRD group14 was undergoing renal function substitution therapy (hemodialysis or peritoneal dialysis). Two groups were chosen for contrast: control group subjects aged ≥30 years attending a sports group and a group of15 geriatric patients (aged ≥65years) as part of an institutional program that provides medical care and treatment for this age group.
Upon entering the study demographic and clinical characteristics were recorded. Through an interview performed by trained personnel, different HRQoL questionnaires were applied, as detailed below.
The study was approved by the local ethics committee and informed consent of all participants was obtained.
MeasurementsGenericThe SF-36 questionnaire was administered to all groups; visual analog scales were applied to assess pain (0–10, where 0 is no pain and 10 is the worst possible pain); overall assessment of patient health (GPH), on a scale of 0–10, where 0 is the worst possible health state and 10 is the best; and physician global assessment, where 0 is the worst possible health state and 10 is best. The Beck questionnaire was used to assess depression. The Health Assessment Questionnaire (HAQ-Di), in the group of OA and RA, which assesses physical disability was applied according to the following scores: 0–1, mild disability; 1–2, moderate and more than; 2, severe disability.16,17
SpecificRA: DAS-28 was measued,18 to establish the degree of disease activity, setting the following levels of activity: remission <2.6; mild activity ≤3.2; moderate activity <5.1; and severe active >5.1.
OA: the WOMAC was applied in the OA group, which is an instrument that assesses pain, function, vitality and mood, in the past 4 weeks, where scores <3 indicate no functional disability, <7 moderate disability, and >8 severe disability.19
DM: DQOL is an instrument that researches satisfaction regarding glycemic control. It consists of 46 questions divided into the following dimensions: satisfaction with treatment, disease impact, social concern, concern regarding diabetes and wellness. The score is 0–100, where a higher score reflects a better quality of life.9
ESRD: KDQOL is an instrument that assesses 11 dimensions: impact of renal disease in the patient's life, quality of social interaction, cognitive function, symptoms, stress, sexual function, sleep, social support, employment status, satisfaction, and support by health workers. The score ranges from 0 to 100, where a higher score reflects a better HRQoL.10
BiochemicalIn all groups a complete blood count was performed; in RA the erythrocyte sedimentation rate was measured; we performed HbA1C in DM as well as central glucose; and in the ESRD group we measured nitrogen products.
Statistical AnalysisDescriptive statistics were used. A comparative analysis was used to determine the quality of life, and the identification of relationships was conducted with the chi-square test for nominal variables. Quality of life of the control group was compared using the Kruskal–Wallis test. Spearman's rho was used to correlate variables. The comparison between independent groups was made with the Mann–Whitney test. The strength of association was evaluated with odds ratios and 95% confidence intervals. A value of P<.05 was considered significant. SPSS 14.0 software was used.
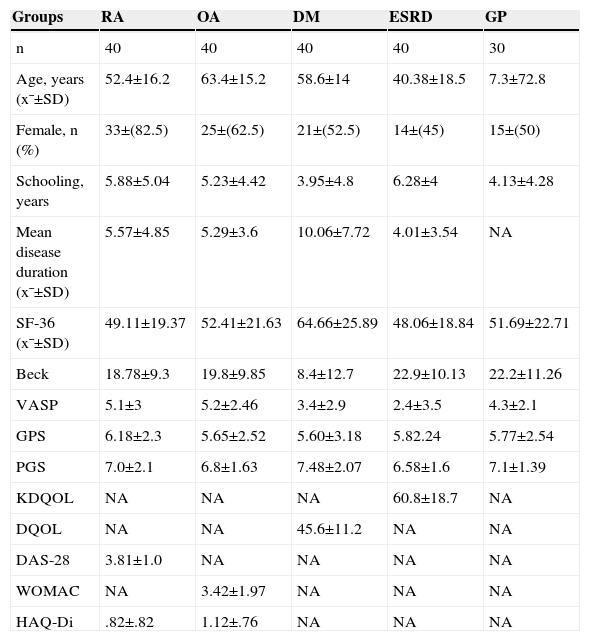
ResultsWe evaluated 290 subjects distributed as follows: 100 in the control group with an age of 53±11.2 years (mean±SD), 30 subjects in the geriatric population with 72.8±7, 3 years of age and 40 subjects in each group with different diseases (RA, OA, DM, and ESRD). The average age in the rest of the groups was as follows: RA, 52.4±16.2 years; OA, 63.4±15.2 years; DM2, 58.6±14 years; and ESRD, 40.38±18.5. Differences in baseline characteristics were detected regarding the instruments used to assess HRQoL, depression and visual analog scales (Table 1).
Demographic Characteristics, Measurement of Quality of Life, Physical Function and Disease Activity.
| Groups | RA | OA | DM | ESRD | GP |
|---|---|---|---|---|---|
| n | 40 | 40 | 40 | 40 | 30 |
| Age, years (x¯±SD) | 52.4±16.2 | 63.4±15.2 | 58.6±14 | 40.38±18.5 | 7.3±72.8 |
| Female, n (%) | 33±(82.5) | 25±(62.5) | 21±(52.5) | 14±(45) | 15±(50) |
| Schooling, years | 5.88±5.04 | 5.23±4.42 | 3.95±4.8 | 6.28±4 | 4.13±4.28 |
| Mean disease duration (x¯±SD) | 5.57±4.85 | 5.29±3.6 | 10.06±7.72 | 4.01±3.54 | NA |
| SF-36 (x¯±SD) | 49.11±19.37 | 52.41±21.63 | 64.66±25.89 | 48.06±18.84 | 51.69±22.71 |
| Beck | 18.78±9.3 | 19.8±9.85 | 8.4±12.7 | 22.9±10.13 | 22.2±11.26 |
| VASP | 5.1±3 | 5.2±2.46 | 3.4±2.9 | 2.4±3.5 | 4.3±2.1 |
| GPS | 6.18±2.3 | 5.65±2.52 | 5.60±3.18 | 5.82.24 | 5.77±2.54 |
| PGS | 7.0±2.1 | 6.8±1.63 | 7.48±2.07 | 6.58±1.6 | 7.1±1.39 |
| KDQOL | NA | NA | NA | 60.8±18.7 | NA |
| DQOL | NA | NA | 45.6±11.2 | NA | NA |
| DAS-28 | 3.81±1.0 | NA | NA | NA | NA |
| WOMAC | NA | 3.42±1.97 | NA | NA | NA |
| HAQ-Di | .82±.82 | 1.12±.76 | NA | NA | NA |
RA, rheumatoid arthritis; DAS-28, Disease Activity Score; DM, diabetes mellitus; DQOL, Diabetes Quality of Life; PGS, physician global assessment; PGS, patient global assessment; VASP, visual analog pain scale; Di-HAQ, Health Assessment Questionnaire Disability Index; ESRD, terminal renal failure; KDQOL, Kidney Disease Quality Of Life; NA, not applicable for the group; OA, osteoarthritis; GP, geriatric population; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
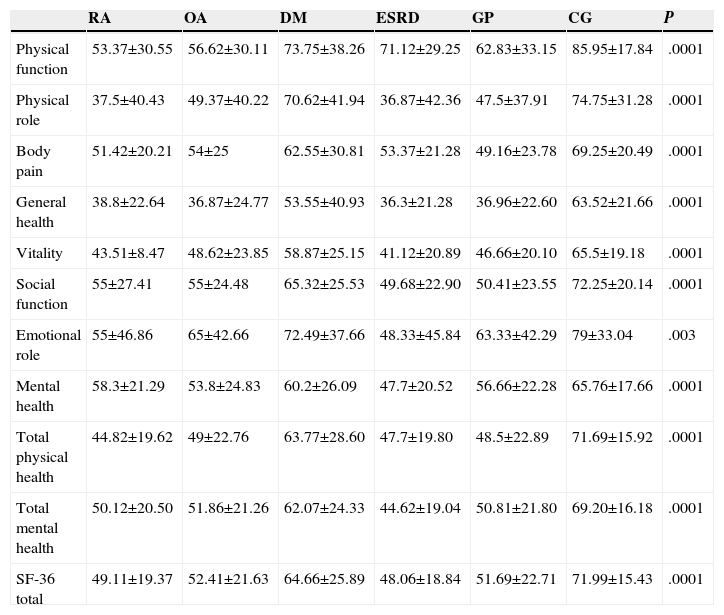
The SF-36 scores were significantly different between the control and groups, with statistical significance (P=.007). The worst HRQoL was observed in patients with ESRD (mean±SD 48.06±18.84) and the RA group, with an average score on the SF-36 questionnaire of 49.1 (SD 19.37), with these 2 groups having the worst HRQoL, taking as a control a value of 71.9 (SD 15.43/min.–max.28.5–94.9). Table 2 describes the SF-36 scores in each of its dimensions: physical function, physical role, bodily pain, general health, vitality, social function and mental health; of all the dimensions, general health was the most affected in all groups studied. The RA group had the lowest score on the general health dimension of the SF-36 (38.80 SD±22.64; 0–87 min–max) and it was statistically significant (P=.0001).
Comparison of the SF-36 Between Groups. P Values Were Obtained With Kruskal Wallis Test for Independent Samples.
| RA | OA | DM | ESRD | GP | CG | P | |
|---|---|---|---|---|---|---|---|
| Physical function | 53.37±30.55 | 56.62±30.11 | 73.75±38.26 | 71.12±29.25 | 62.83±33.15 | 85.95±17.84 | .0001 |
| Physical role | 37.5±40.43 | 49.37±40.22 | 70.62±41.94 | 36.87±42.36 | 47.5±37.91 | 74.75±31.28 | .0001 |
| Body pain | 51.42±20.21 | 54±25 | 62.55±30.81 | 53.37±21.28 | 49.16±23.78 | 69.25±20.49 | .0001 |
| General health | 38.8±22.64 | 36.87±24.77 | 53.55±40.93 | 36.3±21.28 | 36.96±22.60 | 63.52±21.66 | .0001 |
| Vitality | 43.51±8.47 | 48.62±23.85 | 58.87±25.15 | 41.12±20.89 | 46.66±20.10 | 65.5±19.18 | .0001 |
| Social function | 55±27.41 | 55±24.48 | 65.32±25.53 | 49.68±22.90 | 50.41±23.55 | 72.25±20.14 | .0001 |
| Emotional role | 55±46.86 | 65±42.66 | 72.49±37.66 | 48.33±45.84 | 63.33±42.29 | 79±33.04 | .003 |
| Mental health | 58.3±21.29 | 53.8±24.83 | 60.2±26.09 | 47.7±20.52 | 56.66±22.28 | 65.76±17.66 | .0001 |
| Total physical health | 44.82±19.62 | 49±22.76 | 63.77±28.60 | 47.7±19.80 | 48.5±22.89 | 71.69±15.92 | .0001 |
| Total mental health | 50.12±20.50 | 51.86±21.26 | 62.07±24.33 | 44.62±19.04 | 50.81±21.80 | 69.20±16.18 | .0001 |
| SF-36 total | 49.11±19.37 | 52.41±21.63 | 64.66±25.89 | 48.06±18.84 | 51.69±22.71 | 71.99±15.43 | .0001 |
RA, rheumatoid arthritis; DM, diabetes mellitus; CG, control group; ESRD, chronic renal failure; OA, osteoarthritis; GP, geriatric population.
Global health assessments were performed by the physician and the patient regarding the GPS for OA, DM2, ESRD, geriatric population and control groups, with similar scores; the best score was in the control group followed by the RA group (7.1cm±1.39 and 7cm±2, respectively) (Table 1).
The visual analog pain scale in the OA group was 5.2cm±2.46; in RA, 5.1±2.5; and after them, the geriatric population group, ESRD, DM, and finally the control group which had the lowest score for pain.
Mild to moderate depression was detected in 29% (84), and 26% (76) had severe depression. The group with major depression was the ESRD with a score of 22.23 in the questionnaire ((SD/min.–max. 11.26/4–42), P=.0001).
The HAQ-Di was higher in the OA group compared with that in the RA group, 1.12±.76 vs .82±.82 respectively (P=.001).
Differences between the groups were as follows: number of drugs used, which was higher in the ESRD group (5.20±2.45) followed by the RA group (3.53±1.1); and age, higher in the geriatric population and the OA group.
The WOMAC in OA group was 3.42 (SD/min.–max. 1.97/.1–8.7), where the pain subscale score was the highest (3.86±2.37).
DQOL assessed with HRQoL in patients with DM was 45.6 (SD 11.2), being impaired in all 5 areas assessed with this instrument: treatment satisfaction, disease impact, social concern, concern regarding diabetes and general welfare.
The causes of ESRD in the study group were DM in 21 subjects (28%) and hypertension in 21%. Time undergoing dialysis therapy was 1.66 years. The most common dialysis mode was peritoneal dialysis (67.5%/27). HRQoL assessed with KDQOL was 60.8 (SD 18.7), and the best score subscale was that supported by the medical staff.
In RA patients the ESR was 24.23mm/h (SD/min.–max. 7.6/10–48). Glycemic control rates in the DM group was 9.37% for the HbA1C (SD/min.–max. 4.15/3–29) with a fasting blood glucose of 173.43mg/dL (SD/min.–max. 64.96/77–329). Finally, in patients with ESRD, nitrogenous products were as follows: creatinine 12.44mg/dL (SD/min.–max. 4.53/1.4–21.34) and urea 208.66mg/dL (SD/min.–max.95.5/36.4–389).
The correlations that were of interest were as follows: GPS with the erythrocyte sedimentation rate in patients with RA (Spearman rho −.33, P=.03) and in the DM group and, in ESRD, correlation of depression (measured by Beck) with GPS (rho .64/P=.001 and rho=.46/P=.002 respectively). No correlations between GPS and DAS-28, HAQ, HbA1C, fasting blood glucose and creatinine were found.
DiscussionThis study describes the findings in HRQoL in 4 chronic diseases. The quality of life was low in all the groups, consistent with previous studies.20,21 Differences were seen in the perception of pain and disability, which were higher in the groups of rheumatic disease. The lowest level of HRQoL was found in the ESRD group followed by the RA and OA groups.
There are no similar studies in Mexico. A Swedish study published by Arne et al.22 has similar results. They compared HRQoL in patients with obstructive lung disease, RA, DM and a control group. They found that the group with obstructive lung disease had the lowest HRQoL and that fatigue was the most important symptom in both the obstructive pulmonary disease and the RA group. This paper also states that the impact on HRQoL in patients with RA is important and greater than that in patients with DM, because of the greater intensity of pain and functional disability in these patients.
Several studies have shown that patients with musculoskeletal disorders have poor HRQoL; worse HRQoL among rheumatic disease has been detected in patients with OA, RA, osteoporosis and fibromyalgia.23 In our study, the RA group had a poorer HRQoL when compared with the OA and control groups.
A study of HRQoL, held at the Helsinky University clinic24 where patients with rheumatic diseases are sent, found that patients with OA and chronic arthritis reported worse HRQoL scores (.81 on a scale of 0–1, using a generic instrument 15-D).
The functional capacity was accessed using traditional HAQ method25; in the RA group a mean value of .82 was found, somewhat higher than that found in a Canadian study where a HAQ of .66 was found in patients with RA undergoing disease modifying treatment, and 1.14 even in patients without disease modifying treatment.26
Krein et al.27 demonstrated that chronic pain causes diabetic patients to have limitations in the control of their own disease, and, that in patients with higher body pain and poor physical function, glycosylated hemoglobin levels are higher. In our study we found no correlation between the patient global assessment and glycosylated hemoglobin levels, but noted that the degree of depression is correlated with the general perception of the patient's health. This correlation is expected, given that depressed patients found that their quality of life scores are lower.
The impact on HRQoL cannot be attributed only to the underlying illness, influencing other variables such as comorbidity, gender and age, among others. This aspect has already been discussed in previous studies28 where it was found that RA patients have multiple comorbidities, higher scores on cardiovascular risk, and psychiatric disorders such as depression. The depression score of patients evaluated was 18.77±9.3, which places them in mild to moderate depression.
Models with these factors combined have also been observed in studies of HRQoL in DM, where personal, medical and lifestyle factors explained 29% and 14% of the variance of HRQoL and personal satisfaction, respectively, in patients with DM.29
The ESRD study group was very heterogeneous, as some of the patients were on replacement therapy with hemodialysis or peritoneal dialysis; in our country this latter method is still widely used. This point has been evaluated in previous studies, and the type of dialysis and time on dialysis definitely influence the quality of life of these patients.
A prospective observational Australian study30 conducted in 351 geriatric subjects, reported that the quality of life measured by EQ-5D was .55 (.20), and that the most affected areas were the vitality and muscle strength, which make the HRQoL score low. In our study, the lowest score was obtained in the SF-36 in the general health and physical roles; as in the Australian study decrease in physical function in our elderly subjects was probably associated with muscle wasting and changes in OA.
Our study has some limitations. First, it is a cross-sectional study, which could not detect changes over time. The results obtained may not be used for patients with severe disease activity, since patients had mild to moderate levels of activity.
This information is important for both clinicians and health authorities. These measures are needed to improve HRQoL in rheumatic diseases. The results of this study prove that the impact of rheumatic disease on HRQoL is high and is comparable to that of traditional chronic diseases. Therefore, strategies should be aimed at improving HRQoL in these diseases.
Quality of life is a widespread term that allows us to measure the impact of different diseases and the degree of control of these; rheumatic diseases definitely should be considered as high impact diseases, and therapeutic measures should focus more intensively to impact changing their progression which, if not properly modified, leads to deformity, functional limitation, and professional and daily life limitation.
Ethical ResponsibilitiesProtection of people and animalsThe authors declare that in this research no experiments was performed on humans or animals.
Data confidentialityThe authors declare that they have followed the protocols of their workplace on the publication of data from patients, and all patients included in the study have received sufficient information and gave written informed consent to participate in the study.
Right to privacy and informed consentThe authors have obtained informed consent from patients and/or subjects referred to in the article. This document is in the possession of the corresponding author.
Conflict of InterestThe authors have no conflicts of interest.
Please cite this article as: Ambriz Murillo Y, Menor Almagro R, Campos-González ID, Cardiel MH. Calidad de vida relacionada con la salud en artritis reumatoide, osteoartritis, diabetes mellitus, insuficiencia renal terminal y población geriátrica. Experiencia de un Hospital General en México. Reumatol Clin. 2015;11:68–72.