Autoimmune hemolytic anemia (AIHA) is a common manifestation in systemic lupus erythematosus (SLE), and is often refractory to standard treatment. Corticosteroids constitute the first-line treatment, with a rate of initial response of 70%–85%, although the response is maintained 1 year later in less than 20%.1,2 In cases of refractory AIHA, splenectomy has traditionally been the second-line treatment, with a response rate of 60%–70%, resulting in a considerable increase in the risk of severe infections.2 In cases of refractory disease or in which splenectomy is contraindicated, immunosuppressive agents such as azathioprine, cyclophosphamide, cyclosporine A and, in recent years, rituximab (anti-[cluster of differentiation] CD20 monoclonal antibody) can be employed with a good response.3,4 We report two cases of refractory AIHA secondary to SLE treated with rituximab.
Case no. 1: The patient was a 37-year-old woman who had been diagnosed with SLE 10 years ago, and was being treated with hydroxychloroquine (200mg/day). She came to the emergency department with severe weakness, arthralgia and mucocutaneous pallor. Laboratory studies revealed a hemoglobin (Hb) level of 5g/dL and a mean corpuscular volume of 81fL, and thus required a transfusion of packed red cells. We found her total bilirubin (TB) to be 2.3mg/dL at the expense of indirect bilirubin, haptoglobin of 1mg/dL, lactate dehydrogenase (LDH) of 856U/L and positivity on direct and indirect Coombs tests. The patient was diagnosed with AIHA and treatment was begun with corticosteroids (1mg/kg body weight/day) and azathioprine (50mg/12h). The disease became chronic, with corticosteroid-dependent flares.
Case no. 2: The patient was a 19-year-old woman who had recently been diagnosed with SLE and was being treated with azathioprine (50mg/day) and hydroxychloroquine (200mg/day). She came to the emergency department with a fever of 38.5°C, deterioration of her general health status, polyarthralgia and weakness. Laboratory studies revealed a Hb level of 7.5g/dL, TB of 2.16mg/dL with a predominance of indirect bilirubin and LDH was 678U/L. She was diagnosed with AIHA and was treated with corticosteroids at a dose of 1mg/kg body weight/day. She progressed favorably, but subsequently had further hemolytic crises upon corticosteroid tapering.
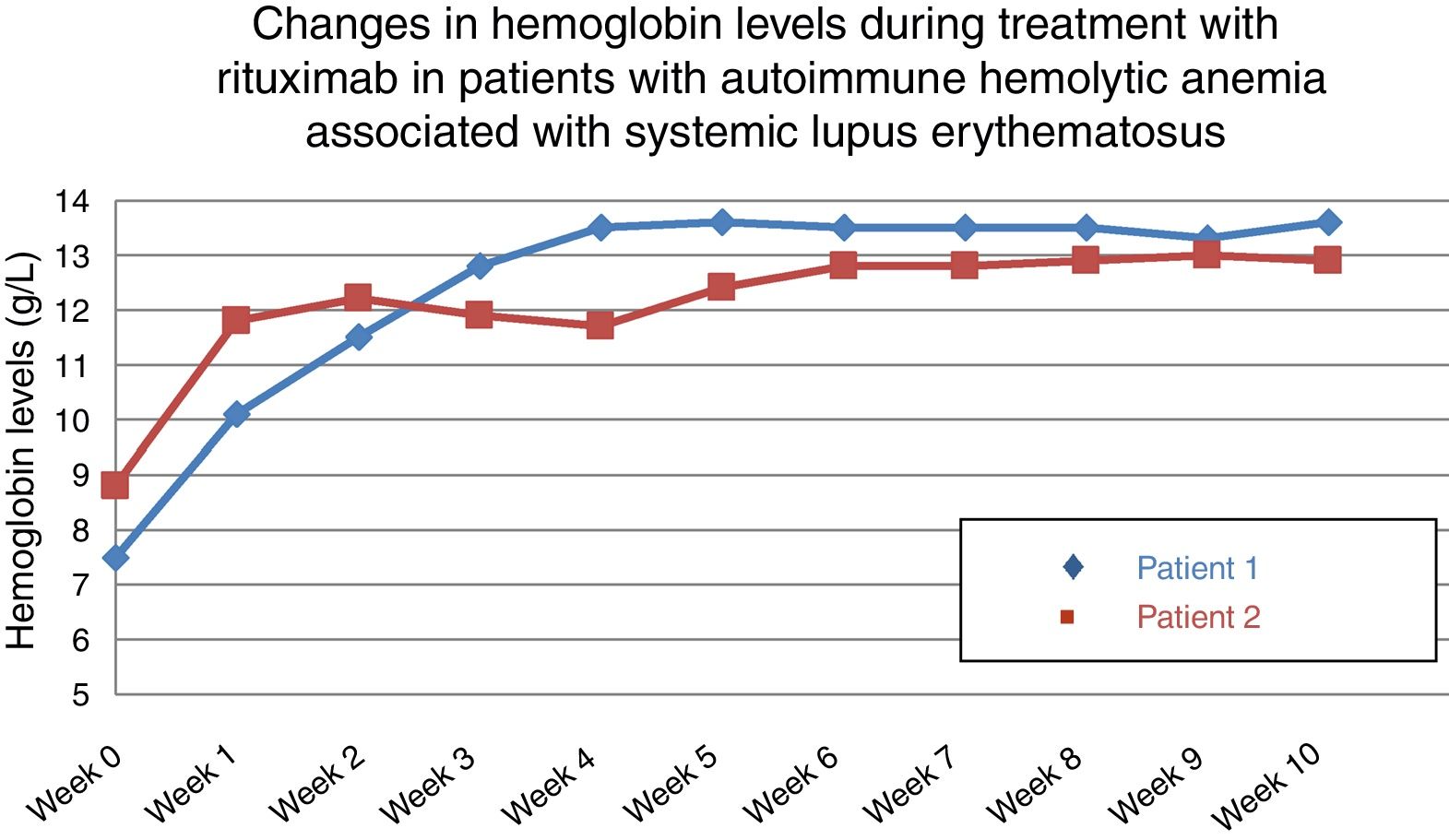
In both cases, given the persistence of relapses despite corticosteroid and immunosuppressive therapy, it was proposed to initiate treatment with rituximab at a weekly dose of 375mg/m2 for 4 weeks, and the outcome was favorable and rapid. The treatment maintained Hb levels over 12.5g/dL after 10 and 12 months, respectively (Fig. 1). In the first patient, we administered a second course of rituximab 1 year later; however, the second patient achieved a response that she continues to maintain.
The efficacy of rituximab in autoimmune hematologic disorders is probably due not only to the elimination of the pathogenic autoantibody, but to B-cell depletion as antigen-presenting cells and their conversion to producers of cytokines.5 Although the impact of rituximab as the first-line treatment of patients with AIHA is still unknown, it has been found to be effective as second-line therapy in prospective and retrospective studies,6–8 with rates of relapse-free survival that range from 64% to 100% at 36 months. Moreover, the rate of response to therapy with a combination of rituximab and corticosteroids is significantly higher than that of corticosteroid monotherapy.8,9
Autoimmune hemolytic anemia is a serious pathological condition. Thus, an early diagnosis is important, as is intensive treatment to detain the hemolytic process. In our experience, the use of rituximab resulted in a rapid and durable response that continued over time in two patients of similar characteristics. We consider, in this respect, and according to the literature reviewed, that the initiation of this drug should not be delayed.6,7,10 Given the safety and tolerability of rituximab,8,9 its use as the second-line of treatment should be recommended, instead of the utilization of immunosuppressive agents that have a greater toxicity or rather than splenectomy.8
Please cite this article as: Pavo-Blanco M, Novella-Navarro M, Cáliz-Cáliz R, Ferrer-González MA. Rituximab en la anemia hemolítica autoinmune refractaria en lupus eritematoso sistémico. Reumatol Clín. 2018;14:248–249.