Necrotizing autoimmune myopathy (NAM) is a rare and emerging entity of idiopathic inflammatory myopathy (IIM).
They have been associated with connective tissue disorders, viral infections, malignancy, anti-signal recognition particle (SRP) and anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase protein (with or without the use of statins).
Anti-SRP-associated NAM has different clinical and histological characteristics that differentiate them from other IIMs, resulting in a poor prognosis. Very few cases treated with rituximab have been published, with varying clinical response.
Here we describe a case of anti-SRP-associated NAM refractory to conventional immunosuppressants and its successful long-term management with the combination of rituximab, corticosteroids and methotrexate.
Las miopatías necrosantes autoinmunes (NAM) son unas entidades raras y emergentes de las miopatías inflamatorias idiopáticas (MII).
Su presencia se ha asociado a conectivopatías, infecciones virales, neoplasias, anticuerpos frente a la partícula de reconocimiento de la señal (anti-SRP) y a anticuerpos frente a la proteína 3-hidroxi-3-metilglutaril-coenzima A reductasa (HMG-CR) (asociada al empleo o no de estatinas).
Las NAM asociadas al anti-SRP presentan unas características clínicas y anatomopatológicas que las diferencian del resto de las MII, resultando en un peor pronóstico. Hasta el momento han sido muy pocos los casos publicados tratados con rituximab y con diferente respuesta clínica.
Presentamos el caso de una mujer de 30 años con NAM asociada al anti-SRP refractaria a tratamiento convencional inmunosupresor y que ha sido controlada a largo plazo con la combinación de rituximab, metotrexato y corticoides.
Necrotizing autoimmune myopathies (NAMs) usually present as a myopathy with marked elevation of creatine kinase (CK), characteristic histopathological findings (presence of necrotic and regenerative muscular fibres, near absence of inflammatory cell infiltrate) and poor response to standard treatment.
Anti-signal recognition particle (anti-SRP) antibodies were described for the first time in 19861 and later were described in patients diagnosed with inflammatory myopathies (IMs) with a more severe disease course. The anti-SRP antibodies are directed against a sub-unit of 54kDa of several ribonucleoproteins with GTPase activated involved in the transport of new proteins to the endoplasmatic reticulum.2–4
Rituximab is a chimeric monoclonal antibody targeted at CD20, the use of which is standard in other connective tissue disorders.5 A series of published cases of NAM exist which were treated with rituximab, with varying clinical responses.6–10
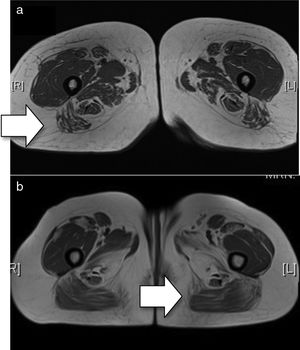
Clinical CaseA female aged 30 was diagnosed with NAM as a result of the presence of proximal myopathy, dysphagia to liquids and dysphonia of 2-month onset, elevation of muscular enzymes (CK 5.922UI/l, aldolase 84UI/l), electromyography with myopathic pattern, signs of intramuscular oedema on the magnetic resonance in shoulder and pelvic girdles (Fig. 1a) and anatomical–pathological findings of necrotic myopathy which had defied standard treatment (corticoids, inmunoglobulins, azathioprine).
(a) Baseline MRI (T1 sequence). There is minimal oedema in the distal end of both the sartorius and gracilis muscles. Compromise of both compartments, with the lateral mass more affected in the anterior compartment and the middle masses more affected in the posterior compartment. In general, there is greater posterior compromise (arrow). (b) MRI after treatment with rituximab (T1 sequence). Bilateral and symmetrical compromise is noted of both compartments with muscular atrophy observed with diffuse fatty deposits. The lateral and middle masses are more affected in the anterior compartment and the hamstring muscles are more affected in the posterior compartment, with predominance of compromise in the posterior compartment (arrow).
As a result of histological findings, anti-SRP antibodies were requested which resulted positive (IFI 1:1.280 technique). Treatment was initiated with methotrexate (up to 30mg per week). Clinical symptoms improved after 3 months with the exception of the iliacus-psoas musculature (4/5). Progressive reduction of corticoids resulted in clinical and analytical deterioration. Concomitant treatment was subsequently started with rituximab (two 1-g doses). Five months later, the patient's condition was improved. After 6 months, a new cycle of rituximab was begun to maintain therapy response.
Magnetic resonance of the muscles after 2 years showed an absence of any signs of inflammatory activity (Fig. 1b).
As methotrexate was lowered below 22.5mg per week, the patient again began to be clinically and analytically compromised, and the dose was therefore increased to regain clinical control in the last year (with the exception of iliacus-psoas musculature weakness).
DiscussionTo our knowledge, less than 30 cases of anti-SRP NAMs treated with rituximab have been published.6–10 Pinal-Fernandez et al.7 describe 17 patients treated with rituximab, which was apparently effective in 13 of them. The duration of the biological effect of the rituximab was variable (from 6 to 9 months to over more than 2 years). In our patient, it has not been administered on more occasions since mid-2012.
Two refractory cases were also published which improved with the combination of corticoids, plasmapheresis and rituximab,8 as well as another two cases treated with rituximab with favourable outcomes.9
However, a British series10 of two patients treated with rituximab presented a poor clinical response.
In our case, two cycles of rituximab were administered, leading to an exacerbation on reduction of the dose of methotexate, with favourable subsequent control when the dose was increased. The CK values of the patient were lower than that published in other series and this may be one of the reasons why a good therapeutic outcome was achieved. The imaging findings were also of note, where greater compromise of the posterior thigh compartment was noted when there were no apparent clinical signs of this.
ConclusionNAMs should be suspected in patients with atypical findings, of MII, both histopathologically (presence of necrotic muscular fibres, near absence of inflammatory cell infiltrate) and clinically (poor response to immunosuppressants).
Rituximab could be a good therapeutic option for those cases where standard treatment has failed.
Ethical LiabilitiesProtection of people and animalsThe authors declare that no experiments using human beings or animals have been carried out for this research study.
Data confidentialityThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of InterestThe authors have no conflicts of interest to declare.
Please cite this article as: Nóvoa Medina FJ, Gutiérrez Martínez J, González González Y, Romero Díaz B, Machín García S, Rosas Romero A. Tratamiento con rituximab para las miopatías necrosantes autoinmunes asociadas al anti-SRP: caso clínico y revisión de la evidencia. Reumatol Clin. 2018;14:379–381.