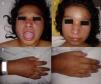
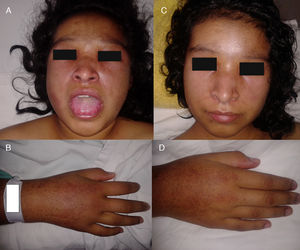
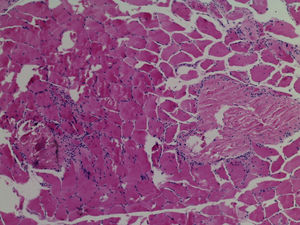
We present the case of a 13-year-old female patient with a condition of 15 days onset, unable to get up and to climb up stairs, who presented with myalgias, dysphagia to solids, and oedema of the face and arms. Physical examination showed weakness on the Daniels scale as follows: cervical 2/5, shoulder girdle 3/5, pelvic girdle 2/5, macroglossia, with oedema of the face and limbs (Fig. 1). Serous creatinine phosphokinase was at 600U/L (normal 38–174U/L), lactate dehydrogenase at 1278U/L (normal 120–300U/L), ANA negative, AST 88U/L normal (10–35U/L), ALT 31U/L (normal 7–35U/L), albumen at 4.2g/dL (normal 3.4–4.8g/dL) and urine test normal. Muscle biopsy was compatible with myositis (Fig. 2). Electromyography with myopathic pattern. Upper GI series with impaired swallowing. A final diagnosis of juvenile dermatomyositis (JDM) was made. Treatment was started with 5 methylpredinisolone pulse therapy (30mg/kg/day), and subsequently with 1mg/kg/day of prednisone, IV immunoglobulin (2g/kg/total dose) and 11 doses of cyclophosphamide monthly (1g/m2 sc), with a gradual lowering of the prednisone dose to 5mg/day. When cyclophosphamide was withdrawn treatment with methotrexate was started (15mg weekly), folic acid and chloroquine. The patient was monitored for 3 years and did not present with any further relapses.
JDM is a generalised inflammatory systemic disease associated with pathognomonic changes to the skin. Subcutaneous oedema as presentation of JDM has been rarely reported and involves an aggressive disease course. Excessive vascular permeability in the muscle and subcutaneous tissues as a result of immune complex deposits, complement activation and damage to the vascular endothelium which leads to muscle microinfarctions may contribute to the presence of oedema.1–3 Severe cases of JDM are associated with the early onset of subcutaneous oedema in limbs, related to inflammatory vasculopathy with a narrowing or complete occlusion of the lumen of medium-sized and small arteries.3 The absence of nephrotic syndrome, cardiac arrest, hypothyroidism, liver disease and malignancy strongly suggest that the oedema is related to JDM. Whilst reviewing generalised oedema-associated cases of JDM we found 19 cases and it was associated with the presence of severe muscle weakness, gastrointestinal or cutaneous ulcers and a poor response to steroids.2,4
In JDM subcutaneous oedema is an indicator of severe disease, and its presence should be a guide for more intensive treatment so as to achieve rapid and appropriate control of the condition.
Ethical LiabilitiesProtection of people and animalsThe authors declare that the procedures followed comply with the ethical standards of the Committee responsible for human experimentation, the World Medical Association and the Declaration of Helsinki.
Data confidentialityThe authors declare that they have adhered to the protocols of their centre of work on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects referred to in this article. This document remains in the possession of the corresponding author.
Conflict of InterestsThe authors have no conflict of interests to declare.
Please cite this article as: Torres Jiménez AR, Solís-Vallejo E, Céspedes-Cruz AI, Sánchez-Uribe M. Edema subcutáneo en dermatomiositis juvenil. Reumatol Clin. 2019;15:e49–e50.
The person who wrote the manuscript is Alfonso Ragnar Torres Jiménez.