Microscopic polyangiitis (MPA) is a systemic disease included in the Chapel Hill 2012 Classification as necrotizing vasculitis affecting capillaries, venules and arterioles. It usually expresses antineutrophil cytoplasmic antibodies (ANCA) and has a perinuclear immunofluorescence pattern and correlation with anti-myeloperoxidase (MPO) antibodies. Capillaritis with alveolar hemorrhage is the most common manifestation of lung disease. Interstitial lung disease (ILD) is uncommon, with usual interstitial pneumonia being the predominant pattern. However, other patterns such as organizing pneumonia have been described. No guidelines exist for treating patients with ILD and, currently, ANCA-associated vasculitis (AAV) is managed along the lines of small vessel vasculitis. The prognosis with this association is uncertain, with possibilities of relapse and a fatal outcome. We present a case in which ILD was the first manifestation of MPA, without alveolar hemorrhage, with subsequent renal involvement and, in which, the established treatment produced a significant clinical improvement.
La poliangeítis microscópica (PAM) es una enfermedad sistémica incluida en la clasificación de Chapel Hill 2012 como vasculitis necrosante que afecta capilares, vénulas y arteriolas. Usualmente expresa anticuerpos anti-citoplasma de neutrófilo (ANCA), con patrón perinuclear en la inmunofluorescencia, y correlación con los anticuerpos anti-mieloperoxidasa (MPO). La capilaritis con hemorragia alveolar es la manifestación más usual de afección pulmonar. La enfermedad pulmonar intersticial (EPI) es infrecuente, siendo la neumonía intersticial común el patrón predominante, sin embargo, otros patrones como la neumonía organizada han sido descritos. No existen pautas de tratamiento de los pacientes con EPI y vasculitis asociada a ANCA (VAA); actualmente son tratados con las bases de las vasculitis de vasos pequeños. El pronóstico de esta asociación es incierto, con posibilidad de recaídas y de curso fatal. A continuación se presenta un caso en el que neumopatía intersticial fue la primera manifestación de una PAM, sin hemorragia alveolar, con posterior involucro renal, y con el tratamiento instaurado presenta mejoría clínica significativa.
The patient was a 56-year-old man, an ex-smoker of 4 cigarettes/day. One month prior to being admitted to the hospital, his health status was characterized by a condition of general deterioration. He had a nonproductive cough, fever of up to 39°C – predominantly in the evening, arthralgia in elbows, knees and carpi, and right-sided chest pleuritic pain. Physical examination revealed bilateral subscapular fine crackles and proximal muscle weakness in all 4 limbs.
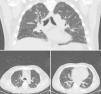
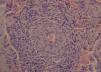
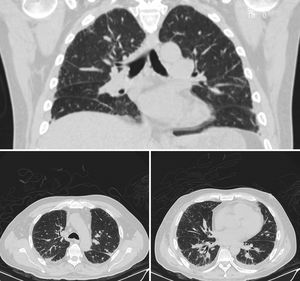
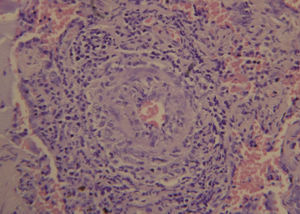
Laboratory tests showed that he had normocytic normochromic anemia. Rheumatoid factor was 65.3IU/L, C-reactive protein 290.4mg/L, total creatine kinase 3.6U/L, creatinine 2.8mg/dL, urea 86.5mg/dL and blood urea nitrogen 40mg/dL. In urine, we observed proteinuria with a level of 75mg/dL and erythrocytes at 260cells/mL, with active sediment characterized by dysmorphic erythrocytes. No evidence of bacteria in urine culture, blood culture, sputum culture or stool culture. The results of a viral panel for hepatitis C virus, hepatitis B virus and human immunodeficiency virus were negative. High-resolution computed tomography of the chest revealed interstitial involvement with a pattern of irregular septal thickening and areas of alveolar filling with air bronchogram in peripherally distributed patches (pattern of organizing pneumonia) (Fig. 1). An electromyographic study showed a pattern indicating a demyelinating disease. Bronchoalveolar lavage demonstrated nonspecific chronic and acute inflammation. Studies focusing on autoimmunity found the patient to be positive for antinuclear antibodies at a titer of 1:320 with a mitochondrial pattern, as well as for perinuclear antineutrophil cytoplasmic antibodies (p-ANCA). The specificity of anti-myeloperoxidase antibodies (anti-MPO) was positive; whereas that of other autoantibodies (anti-Jo-1, anti-Scl-70, anti-double-stranded DNA and anti-cyclic citrullinated peptide antibodies) was negative. The histopathological study of the lung specimen demonstrated small-vessel vasculitis with extensive areas of fibrosis and pulmonary emphysema (Fig. 2).
The presence of interstitial lung disease (ILD), ANCA-associated vasculitis (AAV), proteinuria and neuropathy enabled the classification of the disorder as microscopic polyangiitis (MPA). The treatment was based on pulses of methylprednisolone, azathioprine and rituximab. The outcome was satisfactory, with improvement of the symptoms and signs 4 weeks after treatment, and the absence of relapse over a 3-month period of observation.
DiscussionIn this case of MPA, the presence of early ILD results in an uncommon pattern in lung disease, rather than capillaritis with alveolar hemorrhage and the usual interstitial pneumonia.1,2 In ILD, the main pathological finding lies in the interstitial alveolar structure; however, the airways and pulmonary vasculature can also be affected.3–5
The association of MPA and AAV was described by Arulkumaran et al. in 510 patients, in 14 (2.7%) of whom it was associated with ILD; all of them were positive for p-ANCA and MPO and were diagnosed with MPA.6 Eschun et al. reported similar findings in 6 patients in which the initial manifestation was ILD, which was followed by the development of vasculitis documented by positive p-ANCA and renal biopsy. The histopathological study usually demonstrates rapidly progressive and/or necrotizing crescentic glomerulonephritis.6–8
The mechanisms of lung injury in ILD with AAV are not fully understood. However, repeated episodes of subclinical alveolar hemorrhage and/or the effects of autoantibodies on the surface of neutrophils could induce proinflammatory cytokine production and the release of free radicals resulting in alveolar epithelial injury and pulmonary fibrosis.9,10
There are no guidelines for the treatment of patients with MPA and ILD. At the present time, they are managed along the lines of small-vessel vasculitis. The prognosis of this association is uncertain, and no significant differences are observed with respect to mortality.
In conclusion, the association of ILD and MPA is not very common. However, clinicians in their diagnostic approach to ILD must take into account the possibility of underlying AAV.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflicts of InterestThe authors declare they have no conflicts of interest.
Please cite this article as: García-Nava M, Mateos-Toledo H, Guevara-Canseco APG, Infante-González CE, Reyes-Nava DA, Estrada-Castro E. Enfermedad pulmonar intersticial temprana en poliangeitis microscópica: reporte de un caso y revisión de la literatura. Reumatol Clin. 2018;14:106-108.