We report the case of a 51-year-old man diagnosed in 2011 with polyarticular psoriatic arthritis with nail involvement. The disease was refractory to treatment with nonsteroidal anti-inflammatory drugs (NSAID) and synthetic disease-modifying antirheumatic drugs (DMARD) (methotrexate at 25mg/week and salazopyrine at a dose of 500mg/8h). The decision was made to start biological therapy with tumor necrosis factor inhibitors (anti-TNF), following the recommendations of the European League Against Rheumatism.
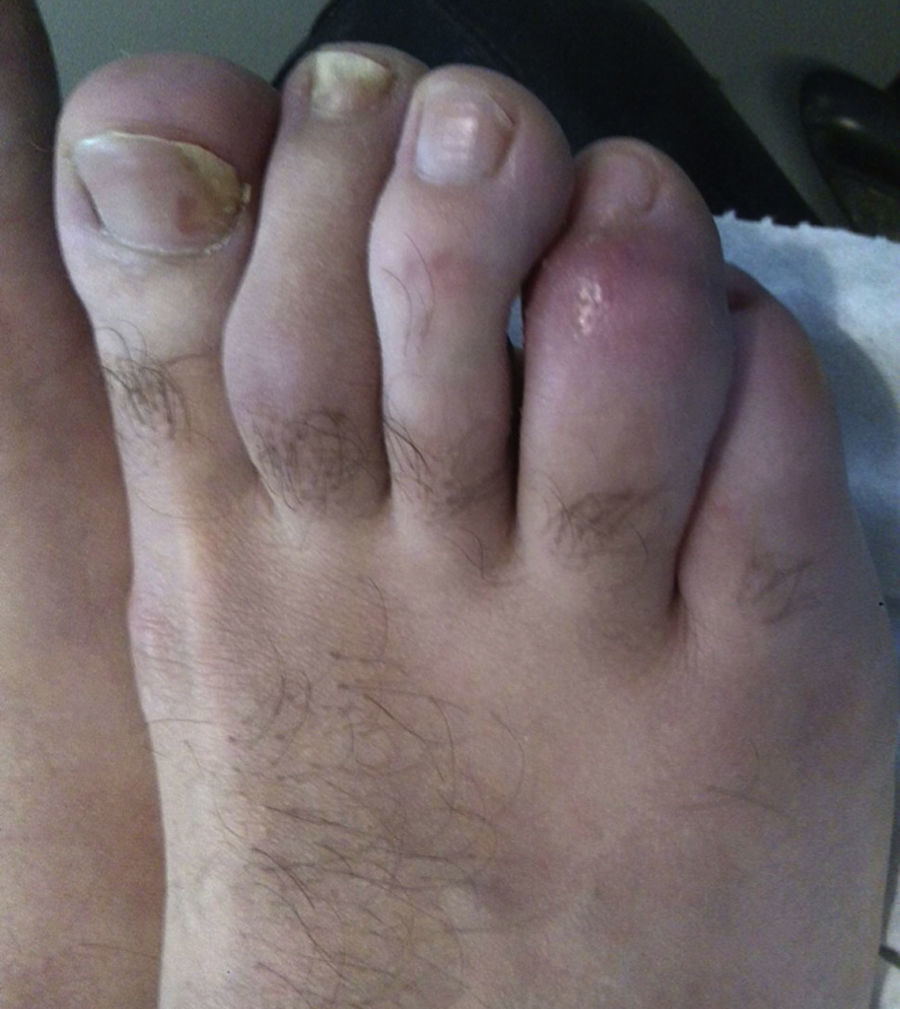
Adalimumab was chosen as the first biological agent. However, after 2 years of treatment, the activity of the disease remained moderate (Disease Activity Score—erythrocyte sedimentation rate [DAS28-ESR=3.70]; 3 tender joints and 3 swollen joints: right 4th metacarpophalangeal [MCP] joint, left 3rd MCP and left knee). In addition, he had painful dactylitis in the 4th toe of right foot (Fig. 1). Thus, treatment was begun with etanercept. Nevertheless, after 6 months of treatment, he continued to have high inflammatory activity (DAS28-ESR=5.80; 4 tender joints and 4 swollen joints: right 4th MCP, left 3rd MCP and both knees). Therefore, treatment was started with certolizumab pegol, which he took for 6 months, although it failed to control the inflammatory activity (DAS28-ESR=4.89; 4 tender joints—right 4th MCP, left 3rd MCP, left 4th proximal interphalangeal [PIP] joint and left knee—and 3 swollen joints: right 4th MCP, left 3rd MCP and left knee), and the dactylitis was not resolved.
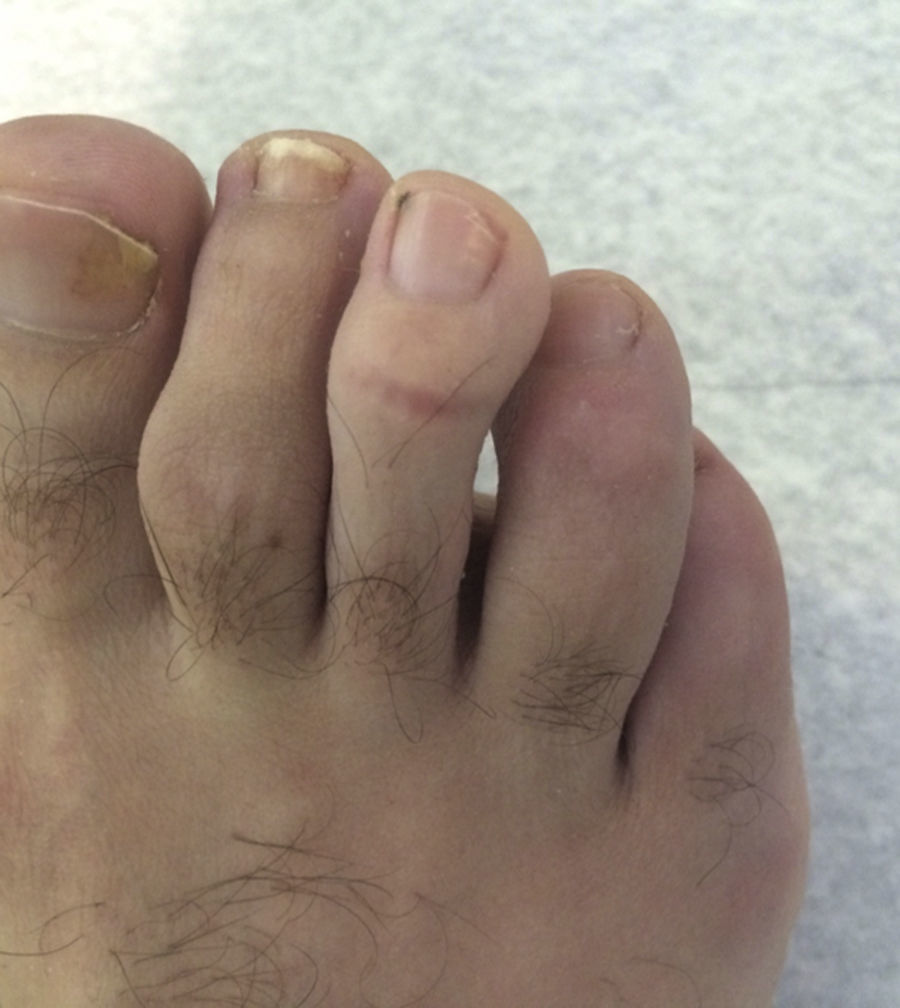
Given the therapeutic failure with 3 anti-TNF-α agents, the decision was made to initiate treatment with ustekinumab. After 2 doses of ustekinumab, he achieved complete resolution of the dactylitis, with a significant improvement in the nail involvement (Fig. 2), and better control of the joint inflammatory activity (DAS28-ESR=3.50; 2 tender joints and 2 swollen joints: left 4th PIP and left knee).
Ustekinumab is a monoclonal antibody that inhibits the p-40 subunit of interleukin (IL)-12/IL-23, that has been found to be effective in all of the domains of psoriatic arthritis.1–3
The importance of this case lies in the rapid resolution of the dactylitis with ustekinumab, despite the fact that it had been found to be refractory to treatment with 3 anti-TNF agents.4,5 This incredible improvement may corroborate the importance of IL-23 in the development of dactylitis and the relevance of its inhibition for the treatment of this domain.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflicts of InterestThe authors declare they have no conflicts of interest.
Please cite this article as: Quesada Moreno A, Martínez González O, Pérez Garrido L, Montilla Morales C. Rápida resolución de la dactilitis en un paciente con artritis psoriásica tras tratamiento con ustekinumab. Reumatol Clin. 2018;14:113–114.