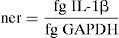The monocyte locomotion inhibitory factor (MLIF) is a heat-stable pentapeptide produced by Entamoeba histolytica in culture. This factor displays several anti-inflammatory properties (i.e., inhibition of locomotion and respiratory burst in monocytes, reduction of skin hypersensitivity and delay of mononuclear cells in human Rebuck skin windows) with inhibition of adhesion molecules, chemokines, and other genes including interleukin-1β (IL-1β). In animal models, it reduces carragenin-induced inflammation and delays the inflammatory process in murine collagen-induced arthritis (CIA).
ObjectivesTo test, in vitro, the anti-inflammatory capacity of MLIF on a promonocytic human cell line (U-937) cells and peripheral blood mononuclear cells (PBMC) from healthy subjects and from patients with rheumatoid arthritis (RA).
Material and methodsIL-1β gene expression was evaluated in cell cultures either in the presence of MLIF, lipopolysaccharide (LPS), or both. Relative gene expression and immunoreactivity of IL-1β were assayed in cells and supernatants, respectively.
ResultsAmebic peptide was able to down-regulate LPS-induced expression of IL-1β, in U-937 cells without a detectable effect upon the bioavailability of the cytokine. In similar culture conditions, MLIF was capable to down-regulate baseline and LPS-induced expression of IL-β only in PBMC from patients with RA. Peptide effect on immunoreactivity of IL-1β was not statistically significant.
ConclusionsMLIF exerts, in primed cells, exquisite anti-inflammatory properties that deserve to be explored mechanistically.
El factor inhibidor de la locomoción de monocitos (FILM) es un pentapéptido termoestable producido por Entamoeba histolytica en cultivo. Este factor presenta diversas propiedades antiinflamatorias, a saber: inhibición de la locomoción y estallido respiratorio en monocitos, abatimiento de la hipersensibilidad por contacto al dinitroclorobenceno y retraso de la quimitoaxis de células mononucleares, disminución en la expresión de moléculas de adhesión y quimiocinas entre otros genes. En ratones el FILM reduce la inflamación inducida por carragenina y retrasa el proceso inflamatorio de la artritis inducida por colágena.
ObjetivoEvaluar in vitro el efecto del FILM sobre la expresión de IL-1β en la línea celular promonocítica humana (U-937) y en células mononucleares de sangre periférica provenientes de donadores sanos y de pacientes con artritis reumatoide.
Material y MétodosSe realizaron cultivos celulares en presencia de FILM, lipopolisacárido o ambos. Después del cultivo se determinó expresión relativa e inmunoreactividad de IL-1β en los botones celulares y sobrenadantes respectivamente.
ResultadosEl péptido amibiano pudo reducir la expresión de IL-1β inducida por LPS en células U937, sin mostrar un efecto detectable sobre la biodisponibilidad de la citocina. En condiciones de cultivo similares, el FILM fue capaz de disminuir la expresión de IL-1β, basal e inducida por LPS, sólo en células mononucleares provenientes de pacientes con artritis. Su efecto sobre la inmunoreactividad de la citocina no fue significativo estadísticamente.
ConclusionesEl FILM ejerce en las células activadas propiedades antiinflamatorias exquisitas que merecen ser exploradas mecanísticamente.
Entamoeba histolytica produces a heat-stable pentapeptide (Met-Gln-Cys-Asp-Ser, MQCNS) that was originally identified by its effects in delaying mononuclear phagocytes migration to inflammation sites. This peptide was hereby denominated by the MLIF and was shown to have several anti-inflammatory properties.1In vitro, it inhibits induction of macrophage inflammatory proteins (MIP-1α, -1β), IL-1β, IL-8, and chemokine CCL1 (I-309), and the chemokine receptor 1 (CCR1) in phorbol myristate acetate (PMA)-stimulated promonocytic human cell line (U-937) cells.2,3 In umbilical vein endothelial cells (UVEC), MLIF reduces vascular cell adhesion molecule (VCAM)-1 and E-selectin expression (personal communication). In vivo it delays mononuclear cells migration to the inflammatory focus in human skin1. The peptide is able to inhibit expression of cell adhesion molecules very late antigen (VLA)-4 and VCAM-1 in a dinitrochlorobenzene (DNCB)-hypersensitivity, guinea pig model.4 In U-937 cells and in the CIA model, analysis of gene expression profiles employing microarrays reveals a regulatory capacity of MLIF on numerous inflammatory genes including IL-1β.2 In addition, the amebic peptide has exhibited a notorious anti-inflammatory effect in carragenin-induced inflammation model in BALB/c mice (personal communication). The MLIF molecular mechanisms of action are poorly understood; however, there is evidence of its involvement in nuclear factor-kappa B (NF-κB) signal transduction; MLIF appears to induce nuclear translocation of p50 homodimers, affecting a number of inflammatory transcripts.5
RA is a multisystemic chronic disease of unknown etiology. During its development, there are periods of activity and reminiscence that could be diagnosed on the basis of a battery of clinical and laboratory tests.6,7 One hallmark of RA is the inflammatory process that occurs in the joint. The resulting inflammatory response induced by pro-inflammatory cytokines (mainly tumor necrosis factor [TNF]α and IL-1β) leads to synovial fibroblast hyperplasia, increased vascularity, and inflammatory cell infiltrate.8–10 Messenger RNA (mRNA) and protein analysis of patients with RA has shown abundant expression of IL-1β and TNFα cytokines.11,12 In this work we tested the effect of anti-inflammatory peptide MLIF upon the constitutive and LPS-induced synthesis of IL-1β in U-937 and PBMC from healthy subjects and from patients with RA.
Materials and methodsCellsThe promonocytic human cell line U-937 (American Type Culture Collection, ATCC, Manassas, VA, USA) was propagated in RPMI-1640 endotoxin-free medium (In vitro, Life Technologies, Eggenstein, Germany) supplemented with 2mM l-glutamine, 1.5g/L sodium bicarbonate, 4.5g/L d-glucose, 10mM HEPES, 1.0mM sodium pyruvate and 10% heat-inactivated, endotoxin-free fetal bovine serum (FBS, Gibco BRL Life Technologies, Grand Island, NY, USA). Cells were maintained at 37°C in a humidified atmosphere with 5% CO2. PBMC were obtained from healthy individuals or patients with AR by Böyum method.13 Briefly, 4mL of diluted blood sample was carefully layered on 3mL of Ficoll-Paque PLUS (GE Heathcare Bio-Sciences Corp, Piscatawa, NJ, USA), tubes were centrifuged at 400×g for 30min at room temperature. Using a Pasteur pipette we take off the mononuclear layer and washed it with balanced salt solution. Typically recovered cells were ≥95% mononuclear cells with ≥95% viability.
CulturesTime-course experiments were carried out in triplicate groups of 4×106mL−1. U-937 cells incubated at 37°C in a humidified atmosphere with 5% CO2 during 0–10h were treated as follows: (a) in RPMI-1640 medium alone (unstimulated); (b) MLIF (50g/mL) added (per se effect); (c) LPS (10 ng/mL) added (induced expression), and (d) MLIF and LPS added simultaneously (interference). After 0.3, 1, 2, 4, 6, 8 and 10h, cells were harvested by centrifugation. All cell pellets were used for RNA isolation, and 4, 6 and 8h supernatant fluids employed for IL-1β immunoreactivity. Triplicate groups of 4×106mL−1 human PBMC were incubated during 4 and 6h without stimulus, MLIF, LPS or both. After 4h, the cell pellet (for RNA isolation) or after 6h, the supernatant fluid (for IL-1β immunoreactivity) was harvested by centrifugation. At the conclusion of all experiments, cell viability was ≥90% by trypan blue dye (Sigma Chemical Co., St. Louis, MO) exclusion. The concentration of MLIF employed in these studies was based on previous in vitro functional experiments.2,3 Simultaneous addition of MLIF to assays was employed, because pilot studies have revealed that MLIF mainly affected cytokine induction when added prior to (weak) or simultaneously (strong), but less after LPS stimulation.
Chemical substancesA single batch, 96% pure MLIF (Met-Gln-Cys-Asp-Ser, MQCNS) endotoxin-free sample was commercially obtained (American Peptide Co., Sunnyvale, CA, USA). MLIF was dissolved in sterile PBS and tested for endotoxin activity using Limulus amoebocyte lysate KTA (Charles River Endosafe Inc., Charleston, SC, USA). Endotoxin activity was ≤0.0625UIEmL/endotoxin. LPS Escherichia coli serotype 0111:B4 was obtained from Sigma.
SubjectsFemales, 35–59 years of age, were recruited from the Blood Bank (healthy donors) and Rheumatology Clinic of “Dr. Emilio Varela-Lujan” General Hospital, Mexican Institute of Social Security (IMSS) in Zacatecas City, Mexico. Patients with RA were classified as such when they fulfilled four or more American College of Rheumatology (ACR) criteria. Subjects were under Anti-inflammatory non-steroid treatment (AINES), or glucocorticoids at doses ≤10mg/day and were free of biologic therapy (anti-TNFα, anti-IL-1β or anti CD20) at least 6 months prior to the study. The study included 13 healthy subjects and 11 patients with RA. All study participants gave written informed consent to participate in the study. The procedure complies with the international ethics for human research and was approved by the Dr. Emilio Varela Luján Hospital Ethics Committee.
RNA extraction and reverse transcriptionTotal RNA was extracted using TRIzol (Invitrogen, Carlsbad, CA, USA) as described.3 RNA purity was established in 2.5% denaturing agarose gels, and the concentration determined in a NanoDrop® ND-1000 spectrophotometer (Thermo Scientific Inc., Wilmington, DE, USA). For reverse transcription, 1μg of total RNA was mixed with 1μL oligo-dT (12–18-mer, Invitrogen), 200U SuperScript™ reverse transcriptase (Invitrogen), 1× First Stand Buffer (Invitrogen), and 0.5mM mixed dNTPs (dATP, dCTP, dGTP, dTTP) (Promega, Co., Madison, WI, USA). The mixture was incubated at 42°C for 1h. Reverse transcription was heat-inactivated at 80°C for 10min and the resulting cDNA was employed for real-time polymerase chain reaction (PCR).
Real-time polymerase chain reaction analysisReal-time PCR was performed in capillaries in a 20μL reaction mix containing Light Cycler Taqman (Roche Applied Science, Salt Lake City, UT, USA), 0.2μM specific primer mix, and 0.1μM of gene-specific hydrolysis probes from the Universal Probe Library (Roche) and cDNA. PCR reaction included 45 cycles consisting of denaturation at 95°C for 10s, annealing at 60°C for 20s, and extension at 72°C for 1s. All reactions were performed in a LightCycler 2.0 instrument (Roche) and data analysis was conducted with LightCycler software 4.05 (Roche).
We obtained the relative expression of IL-1β induced by LPS, MLIF and LPS+MLIF with respect to unstimulated cells using the ΔΔCP method.14 Briefly, we copied the CP values for each sample in an Excel spreadsheet, the first delta CP was calculated from IL-1β CP minus glyceraldehyde 3-phosphate dehydrogenase (GAPDH) CP for each sample accordingly. Deltadelta CP for each condition was equal to stimulated sample delta CP minus unstimulated sample CP. Relative expression was calculated using Relative expression=2(−deltadelta CP) formula.
We considered IL-1β as target gene and GAPDH as housekeeping gene.
For comparison among study groups (healthy subjects vs patients with RA), real-time PCR calibration curves for IL-1β and GAPDH genes were produced from purified specific amplicons. In all groups we obtained normalized expression ratio (ner) of each condition (unstimulated, LPS, MLIF and LPS+MLIF) employing the formula:
Comparison among different ner was used to evaluate basal IL-1β, and responses induced by LPS and MLIF, as well as MLIF capacity to antagonize LPS response.
Enzyme-linked immunoabsorbent assaysIL-1β proteins in supernatant fluids were measured using a commercial ELISA kit DuoSet® (R & D Systems, Inc., Minneapolis, MN, USA). Plates were incubated overnight at 4°C, washed and blocked. Final development employed ExtrAvidin™ HRP and substrate 3,3′,5,5′-tetramethyl-benzidine (Sigma). Microtiter plates were read at 450nm in a BIO-RAD microplate Benchmark densitometer (Richmond, CA, USA). Results were expressed as ng/mL. Sensitivity for IL-1β was ≥2pg/mL.
Statistical analysisResults are expressed as mean ranks. Statistical significance of differences in mean values between different groups was evaluated by Kruskal–Wallis test, followed by U-Mann–Whitney test for inter-group comparisons. A value of p<0.05 was considered to be significant. All analyses were performed with SPSS software version 15.0 (SPSS Inc., Chicago, IL).
ResultsCellular response to lipopolysaccharide and the antagonist effect of monocyte locomotion inhibitory factorPrevious work utilizing U-937 demonstrated that MLIF was able to decrease PMA-induced expression of chemokines MIP-1α and MIP-1β, as well as that of cytokine IL-1β.2,3 We observed this effect only when the peptide was administered prior to or simultaneously with the activator agent. The neutralizing effect of MLIF on such an event was outstanding, if we take into account that PMA is a powerful cell activator that disrupts several cell signaling pathways. On the other hand, because MLIF is involved in the NF-κB signaling, we were interested in observing the amebic peptide effect on the production of LPS/TLR4 signaling-dependent IL-1β.
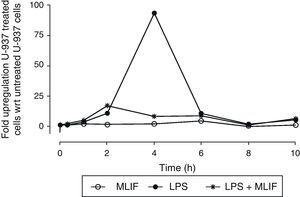
In a triplicate time-course experiment using U-937 cells, LPS-induced expression of IL-1β was followed with real-time PCR. Specific IL-1β mRNA was detected in a 1–10h window of time (Fig. 1). Maximum response to LPS peaked at 4h post-stimulation with 94±1.15 (mean±SEM) folds, and afterwards there was a symmetrically decreasing production. When MLIF was administrated simultaneously with LPS, the amebic peptide exhibited an astonishing antagonist effect at 4h (relative expression 94±1.15-fold with LPS vs 8±0.03-fold with LPS+MLIF (p<0.05)). On the other hand, amebic peptide per se showed no IL-1β regulatory capacity.
Time-course expression of IL-1β in U-937 cells. Triplicate cultures of 4×106 U-937 cells in the presence of MLIF, LPS or both were harvested at 0.3, 1, 2, 4, 6, 8 and 10h. RNA was obtained from pellets and used for real time PCR. Comparative expression was established using unstimulated cells as reference. LPS induced a strong up-regulation of IL-1β which peaks at 4h. MLIF per se did not affect IL-1β basal expression but MLIF was able to reduce significantly the stimulatory effect of LPS when amebic peptide was added simultaneously to LPS (p=0.05). Bars of SEM are <1 in all points.
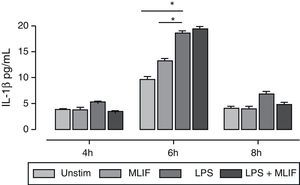
Supernatants from mentioned triplicate experiment were used for measuring IL-1β immunoreactivity released by U-937 cells stimulated with LPS, MLIF or both at three different times (4, 6, and 8h). A higher amount of IL-1β was detected after 6h of incubation where LPS-induction was maximum with 18.5±0.74pg/mL, statistical difference between LPS vs MLIF (18.5±0.4 vs 13.2±0.4), and LPS vs unstimulated cells (18.5±0.4 vs 9.6±0.5) was significant with p<0.05 for both. MLIF showed no effect on IL-1β release by LPS stimulation. At 4 and 8h IL-1β response to LPS was poor, and in most conditions immunoreactivity was near the assay detection limit (Fig. 2). There is a trend for MLIF to interfere with LPS effect at 4h and 8h but statistics showed it not to be significant. Once again, the peptide per se did not affect the production of IL-1β.
Time-course of IL-1β immunoreactivity in U-937 cells. Triplicate cultures of 4×106 U-937 cells in the presence of MLIF, LPS or both were harvested at 4, 6 and 8h by centrifugation. Supernatant fluids were employed for IL-1β immunoreactivity. In general IL-1β released to medium was low at 4 and 8h, response to LPS was higher at 6h *(p=0.05), MLIF per se had no effect over IL-1β release and did not interfere with LPS stimulation.
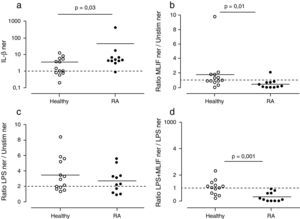
We were interested in testing the observed MLIF effects in differentiated immunocompetent cells. PBMC cultures from healthy subjects and patients with RA were used to look at basal expression of IL-1β. As expected, patients with RA exhibited higher expression of IL-1β mRNA in comparison with healthy subjects. 91% of patients with RA (10 of 11 patients) IL-1β ner was >1; meanwhile in 54% of healthy subjects (9 of 13) IL-1β ner was >1 (Fig. 3a). Statistical data showed a significantly higher expression of IL-1β in patients with RA compared with healthy donors (p=0.03).
Effect of MLIF in PBMC from healthy and patients with RA. Healthy donors (n=13) and patients with RA (n=11) PBMC were cultured during 4h in the presence of LPS, MLIF or both. qPCR analysis for IL-1β transcripts was carried out in each case and comparison between groups established by a normalized expression ratio (ner). (a) Patients with AR constitutively express more IL-1β than healthy donors. IL-1β ner between them was statistically different (p=0.03). (b) MLIF shows a down-regulatory effect on IL-1β expression in patients with RA which ner is <1 in 91% of cases (p=0.01). (c) Capacity of LPS to up-regulate IL-1β in both healthy donors and patients with RA was not significantly different. (d) MLIF was able to down-regulate LPS-induction in all patients with RA (p=0.001).
The capacity of MLIF to affect the expression of IL-1β was determined as the ratio of MLIF ner to unstimulated ner. The presence of MLIF in PBMC cultures decreased IL-1β expression (ratio <1) in 91% of patients with AR (10 of 11) and in 46% of healthy donors (6 of 13) (p=0.01) (Fig. 3b).
Ratios of LPS-induced ner to unstimulated ner were employed to evaluate LPS stimulatory capacity in PBMC cultures. We considered an increase of >2-folds in IL-1β expression as a biologically effective criterion. Studied groups responded in similar fashion to LPS stimulus; 64% (7 of 11) patients with RA and 62% (8 of 13) healthy donors responded to LPS, and no significant differences were found among them (Fig. 3c).
We considered that MLIF could antagonize with LPS when ratios of LPS+MLIF ner to LPS-induced ner were <1. Expression of IL-1β decreased in 100% (11 of 11) of patients with RA and in 38% (5 of 13) of healthy subjects (p=0.001) (Fig. 3d).
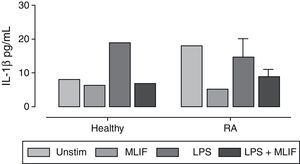
Inmunoreactivity of interleukin-1β in peripheral blood mononuclear cellsIL-1β inmunoreactivity in PBMC cultures was in general low (below the detection limit in the majority of cases). It was only possible to detect IL-1β in 36% (4 of 11) of patients with RA and in 8% (1 of 13) of healthy donors. Apparently, RA group releases more IL-1β than healthy group (18±2.1 vs 8±1.6pg/mL, respectively). However, because the low number of data, no significant differences were found between them. In the group of patients with RA, we observed a MLIF trend to diminish the amount of IL-1β spontaneously released in culture from 18.0 to 5.1pg/mL, as well as an antagonist effect on LPS-stimulation in both RA and healthy groups (from 14.6 to 9.0; and from 19.2 to 7.1pg/mL, respectively) (Fig. 4). This result is interesting but not conclusive due the low number of immunoreactive samples.
Immunoreactivity of IL-1β in PBMC from healthy donors and patients with RA IL-1β immunoreactivity was detected by ELISA in supernatant fluids of 6h cultures of PBMC from healthy donors (n=11) and patients with RA (n=13). IL-1β was not detected in most patients with RA and healthy donors. Patients with RA show higher spontaneous release of IL-1β than healthy donors, and a poor response to LPS stimulus. MLIF per se seems to down-regulate IL-1β in patients with RA, and interferes with LPS induction in both healthy donors and patients with RA. Statistics were not available because of the low number of immunoreactive samples.
Several studies have shown the MLIF modulatory capacity on inflammatory genes. The antagonist effect of the amebic peptide when administered prior to or simultaneously with LPS appears to involve it in early signaling events linked with either putative or non- putative membrane receptors.5 Data on LPS activation by means of TLR4 in U-937 cells had revealed that MLIF is able to interfere with MyD88 membrane recruitment in response to LPS, thus affecting an array of MyD88-dependent genes 5. Ongoing studies in our laboratory with respect to IL-1β synthesis utilizing U-937 cells have shown that MLIF increase the degradation of specific mRNA when induced with LPS. The use of ERK, JNK, p38 and NF-κB inhibitors have brought to light the participation of a NF-κB inhibitory dimmer induced by MLIF that is perhaps responsible for abrogating IL-1β transcription and participating in a cross-talk between NF-κB and mitogen-activated protein kinase (MAPK) signaling cascades. Differential expression profiles of U-937 stimulated with PMA demonstrated a remarkable interfering capacity of MLIF on induced transcription of IL-1β. In this report when using U-937 cells, we found that MLIF per se did not affect IL-1β expression, but that it indeed interferes with LPS in a narrow window of time. MLIF shows no capacity in regulating the LPS-induced immunoreactivity of IL-1β in U-937 cells; however, it is important to consider the poor response of these cells to LPS. Regulation of MLIF on IL-1β transcripts but not on IL-1β protein of U-937 cells could be due to genetic alterations in the cell line, lack of synchronicity in the synthesis, storage, and further secretion of mature IL-1β. Assessment of IL-1β requires the presence in the medium of inmunoreactive cytokine which is generated in a cyclic and complicated process. The immunoreactivity detected is closely related with the cell's ability to generate and to secrete caspase-1-processed IL-1β. Clearly, additional studies on the extracellular bioavailability and intracellular storage of IL-1β in stimulated cells in the presence of MLIF are required.
On the other hand, the effect of MLIF on primed cells is slightly different in immunocompetent PBMC from an inflammatory environment such as RA. In arthritic PBMC, there is an over-expression of basal IL-1β, which is in agreement with other reports.15 MLIF down-regulated IL-1β basal expression and interfered with LPS-induction. IL-1β immunoreactivity levels in PBMC culture supernatants exhibited in general a lower detection limit, but in the few subjects with detected activity MLIF acted mainly on patients with RA, decreasing spontaneous release of IL-1β and interfering with LPS-induction. An increased number of subjects would aid in clarifying this issue.
We are not certain about MLIF action only in transcription but not in translation of IL-1β. For example, we have evidence that administration of the amebic peptide in a CIA model ameliorates the inflammation process and modulates gene expression profiles (IL-1β included among other dozens of genes).16 Furthermore, MLIF is able to reduce the immunoreactivity of IL-1β and matrix metalloproteinases in joints of arthritic mice (paper in progress).
Concluding remarksMLIF was able to distinguish between healthy subjects and patients with RA, based perhaps upon a differential cell-activation degree and the signaling pathways in use. It is plausible to think that synovial cells would be a more representative entity for testing the interesting effects described in this work, and further studies are required to resolve this matter. The MLIF effect on caspase-1, translation, storage and release of mature IL-1β is unknown.
The results presented in this work provide evidence of MLIF's most exquisite anti-inflammatory properties on prime and arthritic cells and encourage its research in other inflammatory processes.
Ethical disclosuresProtection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of Data. The authors declare that no patient data appears in this article.
Right to privacy and informed consent. The authors have obtained the informed consent of the patients and /or subjects mentioned in the article. The author for correspondence is in possession of this document.
FundingThis work was supported by Fondo de Investigación en Salud of IMSS, Mexico, grant FIS/IMSS/PROT/356.
Conflicts of interestThe authors have no conflicts of interest to declare.
We wish to thank José Alvarez-Nemegyei PhD, Unidad de Investigación Médica Yucatán, IMSS, for his helpful comments and critical revision of the manuscript. The style review of the manuscript by Ms Maggie Brunner is greatly appreciated.