The aim of this study was to analyze which are the main factors that could influence the result of a CT guided biopsy in vertebral osteomyelitis (VO) patients.
MethodsA single center retrospective observational study was performed including adult patients who had been diagnosed with VO and undergone CT guided needle biopsy from January 2010 to January 2020. Demographical features, concurrent diseases, laboratory findings, microbiological diagnosis, radiological data, medical complications, antibiotic exposure were compiled. Multivariate analysis was performed with a logistic regression comparing the patients depending on the culture result.
ResultsSeventy-seven patients were included in the study. Baseline characteristics were comparable between groups. Sample culture was positive in 43 cases (56%). Microorganism isolated were gram+(72%), gram−(14%), mycobacteria (7%) and fungi (7%). Delay in the procedure, antibiotic exposure and blood culture positivity were also similar among both groups. The biopsy results were not influenced by the CRP value, the presence of fever nor antibiotic exposure. The longer duration of back pain was associated to a lower probability of a positive culture.
ConclusionsIn conclusion, our study displays an acceptable reliability of CT guided needle biopsy in VO patients, even in cases under antibiotic treatment. The presence of fever or CRP values did not predict a positive culture. Delay in diagnosis could impact negatively on culture yield.
El propósito del presente estudio es analizar qué factores pueden influir en el resultado del cultivo de las muestras obtenidas por punción guiada por TC en pacientes con osteomielitis vertebral.
MétodosSe realizó un estudio en un único centro, retrospectivo y observacional en pacientes diagnosticados de osteomielitis vertebral, que fueron subsidiarios de punción-biopsia entre enero de 2010 y enero de 2020. Se recogieron para su análisis, variables demográficas, comorbilidades, resultados de laboratorio, radiología, el tratamiento previo con antibióticos y la demora previa a la realización de la técnica. Se realizó un análisis multivariante mediante regresión logística.
ResultadosSe incluyó a un total de 77 pacientes que fueron sometidos a la técnica. Sus características basales fueron similares. El cultivo fue positivo en 43 casos (56%). Los microorganismos aislados fueron gram + (72%), gram – (14%), micobacterias (7%) y hongos (7%). El retraso en la ejecución de la técnica y el tratamiento previo con antibióticos fue similar en ambos grupos. Ni el valor de PCR, la presencia de fiebre ni la antibioterapia tuvieron influencia en el resultado del cultivo. Se observó que una mayor duración del dolor lumbar se relacionó con una menor probabilidad de obtener un resultado positivo en el cultivo.
ConclusionesIncluso bajo exposición antibiótica, la punción asistida por TC mostró una rentabilidad aceptable. La presencia de fiebre o valores elevados de PCR no fueron predictivos de positividad del cultivo. El retraso diagnóstico sí podría impactar negativamente en la rentabilidad diagnóstica del cultivo procedente de la biopsia.
Vertebral Osteomyelitis (VO) is an infectious disease of the vertebral body which can involve the intervertebral disc space.1 The clinical manifestations, laboratory markers and radiological findings may be sometimes unspecific, thus its diagnosis turns frequently into a challenge.2,3 Therefore, current ISDA (Infectious Diseases Society of America) guidelines suggest that a proper evaluation of a patient should include a thorough neurological examination, baseline CRP and ESR determination, a spine imaging technique (preferred MRI) and bacterial blood cultures. Moreover it is recommended, if possible, to withhold the antibiotic therapy until a microbiological diagnosis could be established. However, if blood cultures do not yield the causative microorganism, an image-guided aspiration biopsy is encouraged.3 The most frequent microorganism involved is Staphylococcus aureus in more than half of the cases but other causes could be seen such as other bacteria species or even fungi or mycobacteria.4,5
Biopsy material could be obtained via open procedure (open biopsy) or by percutaneous access (usually assisted by CT or fluoroscope). There are several studies reporting both techniques yield, which had shown a wide range of results. Some of them had shown low positivity rates with CT guided biopsy (30–50%) and higher with open biopsy,6–8 however other groups highlighted the importance of withholding antibiotics prior to the procedure,9–12 fact that needs to be further explored because other studies have failed in finding any relationship between the antibiotic exposure and the technique result. Regardless, though open biopsy seems to be more reliable, percutaneous procedures such as CT guided biopsy are preferred1 because they are safer, less invasive, require shorter hospital stays and in most cases, general anesthesia is not required.13
Since there is still controversy around this topic,6 the aim of this study was to analyze which are the main factors that could influence in the result of a CT guided biopsy in vertebral osteomyelitis patients.
Patients and methodsStudy design and populationA single center retrospective observational study was performed. Inclusion criteria was adult patients who had been diagnosed with VO from January 2010 to January 2020. Diagnosis of VO, according to other studies was established based on the combination of the clinical presentation with either a definitive bacteriologic diagnosis or pathological and/or imaging studies.2–12 Patients were admitted to different wards, so medical and surgical approach was not standardized. Patients who did not undergone CT guided biopsy or had a postsurgical infection were excluded. Coagulase-negative staphylococcus was considered a true pathogen when it was isolated from biopsy or when it was detected in two or more blood cultures.
Clinical cases were search through the clinical history management software (Orion Clinic, Everis Group). Terms searched were “vertebral osteomyelitis”, “discitis” and “spondylodiscitis”.
Demographical features, concurrent diseases, laboratory findings, microbiological diagnosis, radiological data, medical complications, antibiotic exposure were compiled.
Variable definitionImmunosuppressed patients were considered those who had a rheumatic or inflammatory bowel disease undertaking immunomodulatory drugs, solid organ transplantation receptors, or in Human Immunodeficiency Virus (HIV) patients. Neurological deficit included paresthesia, limb weakness, radiculopathy, sensory loss and/or paraparesis. Clinical and radiological history of lumbar stenosis or disc herniation was considered as prior spine pathology. Chronic Kidney Disease was considered when glomerular filtration rate (GFR) was below 30mL/min, thus, pertaining to groups 4 and 5 of the CKD classification.
Radiological presence of epidural abscess, paravertebral abscess, cord compression and vertebral destruction was determined by a musculoskeletal specialized radiologist. Infection location was defined by the most superior segment involved.
Procedure descriptionStandard procedure at our center is performed by Musculoskeletal Specialized Radiologist under local anesthesia and CT control. Abscess sample is collected with Acecut® needle (18G) with coaxial technique, trying to obtain at least 3 samples. For discal space, Ostycut® needle (13.5G–15G) is used. For detection of mycobacteria, culture and real time PCR was performed (Seegene kit). If possible, samples were sent for histopathology, anaerobic, aerobic, mycobacteria and fungal cultures.5
Statistical analysisContinuous variables were summarized using mean (SD, standard deviation) and median (1st, 3rd quartiles). Categorical variables were summarized using absolute and relative frequencies (%). To assess the association between risk factors a logistic regression was adjusted including CRP at admission, the presence of fever, back pain span and antibiotic exposure comparing the patients depending on the culture result. All statistical analyses were performed using software R (version 3.5.0). P values were set based on number of analyses.
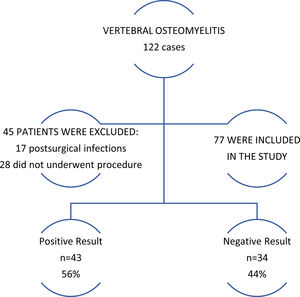
ResultsStudy patientsOverall, from a total of 122 vertebral osteomyelitis cases found, 77 (63%) were included in the study (Fig. 1). Patients were divided into groups depending on the culture result. Baseline characteristics are shown below (Table 1).
Demographic and baseline characteristics.
| Characteristic | Biopsy culture positive (56%) | Biopsy culture negative (44%) |
|---|---|---|
| n=43 | n=34 | |
| Age – years (ds) | 60 (16.6) | 61 (14.2) |
| Male sex – number (%) | 33 (77) | 21 (62) |
| Comorbidities – number (%) | ||
| High blood pressure | 21 (49) | 18 (59) |
| Diabetes Mellitus | 12 (28) | 5 (15) |
| Chronic kidney disease | 7 (16) | 7 (21) |
| Liver cirrhosis | 7 (16) | 8 (24) |
| Immunosuppression– number (%) | 9 (21) | 8 (24) |
| Prior spine pathology – number (%) | 20 (47) | 19 (56) |
| Symptoms and signs at diagnosis | ||
| Back pain – number (%) | 43 (100) | 33 (97) |
| Time since pain started – median | 30 (20.5, 60) | 55 (22.5, 90) |
| Fever – number (%) | 15 (38) | 12 (36) |
| Time since fever started - median | 0 (0, 1) | 0 (0, 6) |
| Acute paraparesis – number (%) | 5 (12) | 7 (21) |
| Underlying endocarditis – number (%) | 0 (0) | 2 (6) |
| Acute phase reactants | ||
| CRP – mg/L median | 88.8 (48.9, 124) | 73.8 (23.1, 128) |
| ESR – mm/h mean | 69.5 (32.1) | 59.5 (24.3) |
| Infection location – number (%) | ||
| Cervical | 5 (12) | 4 (12) |
| Dorsal | 19 (44) | 14 (41) |
| Lumbo-sacral | 19 (44) | 16 (47) |
| Abscess – number (%) | 42 (98) | 32 (94) |
| Paravertebral | 41 (95) | 32 (94) |
| Epidural | 29 (67) | 21 (62) |
| Vertebral destruction – number (%) | 29 (67) | 19 (56) |
| Cord compression – number (%) | 15 (35) | 17 (50) |
Mean values are expressed: Mean (SD)/n(%). Median values are expressed: Median (1st, 3rd Q.).
Most patients were diagnosed with MRI (65% and 85% respectively between groups), followed by CT (33% and 15% respectively). In 1 case in the biopsy positive group, 18F-FDG PET/CT was the diagnosis imaging tool.
InterventionDelay in the procedure, antibiotic exposure and blood culture positivity were similar among both groups (Table 2).
Procedure related variables.
| Biopsy culture positive (56%) | Biopsy culture negative (44%) | |
|---|---|---|
| n=43 | n=34 | |
| Blood culture (positive) – number (%) | 14 (42%) | 13 (41%) |
| Time since admission to procedure – median days | 6 (3, 11) | 6.5 (3, 18.5) |
| Antibiotic exposure – number (%) | 19 (49%) | 18 (58%) |
| Antibiotic exposure prior procedure – days | 0 (0, 6) | 4 (0, 11) |
Sample culture was positive in 43 cases (56%) as seen in Fig. 1. Microorganism isolated included gram+(72%), gram−(14%), mycobacteria (7%) and fungi (7%). Detailed data of microbiological findings are provided in Table 3. Seventeen patients had a positive result on the biopsy and blood cultures. In 14 of these 17 cases (83%) it was isolated the same organism on the blood and biopsy culture (see notations below Table 3). Microbiological diagnosis in biopsy negative group was established according to blood cultures and was possible in 16 patients (47% of the biopsy negative group).
Microorganisms isolated from biopsies.
| Microorganism isolated | n=43 |
|---|---|
| Gram positive species – number (%) | 31 (72) |
| Staphylococcus aureus MS | 7 |
| Staphylococcus epidermidisa | 12 |
| Staphylococcus hominis | 1 |
| Streptococcus mitis | 1 |
| Streptococcus anginosus | 2 |
| Streptococcus beta hemol grupo B | 1 |
| Streptococcus gallolyticus spp.b | 1 |
| Streptococcus sanguinisc | 1 |
| Corynebacterium spp. | 1 |
| Staphylococcus capitisd | 2 |
| Enterococcus spp. | 1 |
| Propionibacterium acnes | 1 |
| Gram negative species – number (%) | 6 (14) |
| Pseudomona aeruginosae | 3 |
| Brevundimonas spp. | 1 |
| Escherichia colif | 2 |
| Fungi – number (%) | 3 (7) |
| Candida aurisg | 2 |
| Candida albicansh | 1 |
| Mycobacteria – number (%) | 3 (7) |
| Micobacterium tuberculosis | 3 |
Multivariate analysis was performed with a logistic regression including CRP at admission, fever, back pain span and recent antibiotic treatment. The biopsy cultures were not influenced by the CRP value (CI 95% [0.994–1.006] p=0.927), the presence of fever (CI 95% [0.518–5.533] p=0.41) nor antibiotic exposure (CI 95% [0.116–1.355] p=0.155). The longer duration of the back pain was associated to a lower probability of a positive biopsy (CI 95% [0.977–0.998] p=0.042).
Patients received antibiotic treatment during the next 84 (56, 112) days in biopsy positive group and 84 (56, 120) days in negative group. Fifteen patients required surgery (6 in positive group and 9 in the negative group, 14% and 26% respectively). Sixteen patients were readmitted after discharge (16 in positive group and 10 in the negative group, 37% and 29% respectively). Six patients died due to the infection (4 cases) or related complications (2 cases) (3 in positive group and 3 in the negative group, 7% and 9% respectively).
Immunosuppresed patientsA total of 17 patients were immunosuppressed. Results of these population are commented separately due to its special characteristics. Immunosuppressed patients represented the 22% of the total sample and were balanced between both groups, whom exhibited similar baseline characteristics than non-immunosuppressed patients (see Table 1). Acute phase markers seemed to be slightly inferior in negative biopsy group. Positive biopsy group may have more abscess formation (100% of the positive group and 87% on the negative group, but epidural abscess was detected in 100% of the patients of the positive group and in the 50% of the negative group). Delay in procedure performance was slightly inferior in immunosuppressed patients (mean 5.2 days). Gram positive predominated on cultures (8 cases) and in the other case, C. auris was detected. In 4 cases (44%), blood and biopsy cultures isolated the same microorganism. Two patients were splenectomized and in one case Staphylococcus epidermidis was isolated on the blood culture (during a febrile episode) and in the other no potential causative organism was detected (the biopsy was performed under antibiotic exposure). On both cases, the biopsy was negative with a delay of 3 and 4 days respectively.
DiscussionIn this study including VO patients, the focus has been placed on the biopsy that is needed in many patients, in an attempt of elucidating the causative microorganism so as to provide a targeted antibiotherapy, and thus, reducing side effects and improving the efficacy.1,7 In particular CT guided needle biopsy had been analyzed in a population with demographic and clinical comorbidities distribution similar to other studies.2,6,7 The population of the study had been split in two groups attending to the result of the culture of the sample obtained from de biopsy.
In this cohort a higher presence of immunosuppressed patients could be noted, due to the characteristics of the center, which is a university hospital. The results of that subset of patients have been displayed separately. In general, the distribution of cardiovascular risk factors and other clinical variables has been fair between both groups. Although there are some differences on the radiological variables (more prevalence of epidural abscess and cord compression) and also, the acute phase markers were slightly greater on the positive result group. In any case, extravertebral extension could not act as a factor of poor prognosis.4 This fact would be explored on further studies but seems to be logical due to a presumed more aggressiveness of the infection on immunocompromised host. Moreover, other studies found correlation between higher CRP value and greater chance of a positive result.7–9
The signs and symptoms collected seems to be in agreement with other series3,4,7 with the expected high prevalence of back pain, presented in almost all patients and fever in just 1 out 3 patients, approximately in both groups. As seen in other works, delay in diagnosis is still a problem in VO patients2,4 who usually waits at least a month since the beginning of the pain, until diagnosis. Additionally, in some cases this period is increased dramatically. Further strategies should be set up in order to reduce this term and prevent future complications.14
Infectious endocarditis is a frequent association with VO,15,16 but in this study were scarcely represented because in those patients the causative microorganism is more frequent detected through the blood cultures.
Acute phase markers (CRP and ESR) were elevated in both groups. Numerically, values were higher in biopsy positive groups, as seen in other studies7–9 but statistical signification was not reach. Radiological variables were comparable in both groups. A slight difference was noted in cord compression (higher in negative group) probably related to a greater presence of patients with acute paraparesis at diagnosis in this group. This result might be linked to the higher presence of further surgical approach detected in the negative culture group.
As it has been pointed out in several studies delay in diagnosis is probably the main issue in VO.2,17 Moreover, since antibiotic withholding is widely recommended,1 these biopsies should be performed as soon as possible. In this study a median delay of 6 days was detected on both groups, with few differences in the interquartile range (Table 2). Marschall et al. achieved a 65% of positivity with a median delay of 3 days.10 Better mean values in immunosuppressed patients have been noted, but overall, this should be improved and analyzed (for instance, creating a specific circuit).
Microorganisms isolated had a distribution comparable to another studies.2,18 Antibiotic exposure was registered in half of the cases on the positive group and in the 58% of the negative group. The higher prevalence of patients with acute paraparesis at diagnosis could be a reasonable cause. In addition, more antibiotic exposure is presumed to reduce the culture yield,11 but it was not detected on our study. This is a matter of controversy, but there are other several studies that concluded the same.12,17 A possible explanation of the lack or the small influence of antibiotic exposure on the culture result could be that intervertebral space is avascular. A high correlation between blood cultures and biopsy had been noted, so as it is had been previously recommended on the guidelines and other works,1 CT-guided biopsy could be avoided in most cases.
Overall positivity (56%) was on the average of the studies published which vary from 21% to 89%.4,7–9,19–21 The biopsy result was not modified by prior CRP values, fever as clinical presentation nor antibiotic exposure. The longer the length of painful period before diagnosis was related to a lower chance of obtaining a positive result on culture. This result could be explained by a greater aggressiveness of pyogenic organisms that perhaps congregate in the lesser time span instead of non-pyogenic agents and seems logical that the delay in diagnosis makes a negative impact on the technique reliability. Other studies has been performed in order to detect predictive factors of positivity, such as a recent study found that using larger diameter biopsy needle increases the likelihood of causative microorganism.19 Other factor detected related to higher chance of positivity has been the attenuation value on CT scan.20
In order to point out the limitations of the study, since this is a retrospective study, clinical cases may be missed during the search process. Moreover, patients were admitted to different wards, and that hence on variability in patient management. Concerning the procedure, adding more variables would provide more information such as areas specifically punctured, specific antimicrobial treatment received prior to the procedure or number of samples taken at each procedure. During statistical analysis some difficulties were found, such a huge variability in days of antimicrobial therapy received prior the procedure, finally solved dichotomizing in “exposure: yes/no” and reducing the statistical power. The inclusion of mycobacteria and fungi might influence the clinical picture and the yield of the CT-guided biopsy.
In conclusion our study displays an acceptable reliability of CT guided needle biopsy in VO patients, even in cases under antibiotic treatment. The presence of fever or elevated CRP values did not predict a positive culture in our study. A positive blood culture would provide a microbiological diagnosis in most cases. Delay in diagnosis could impact negatively in culture yield. Therefore, improving diagnostic strategies in general and shortening the span from VO diagnosis to the procedure performance in particular, may enhance not only the culture yield reliability but also the patient prognosis.
FundingNone funding source was required.
Conflict of interestNone.
To our partners from the Rheumatology department, the Infectious disease Unit and the Musculoskeletal Unit of the Radiology department. Also to Mrs. Paula Giménez for her effort and valuable help.
The authors are grateful for the collaboration with English language revision to Mr. Paul Godfrey.
All authors have seen and approved the manuscript, contributed significantly to the work, and also that the manuscript has not been previously published nor is not being considered for publication elsewhere.